- University Vending Machine Offers Morning-After Pill (drugs.com)
The morning-after pill is being sold in a vending machine at the University of California, Davis and many people support this type of availability...Along with condoms and pregnancy tests, the Wellness-To-Go Machine in a study lounge also dispenses the Plan B pill for $30 a box…It took economics major Parteek Singh nearly two years to get the vending machine into the lounge... Singh is getting calls from student across the country who want the same type of vending machine. "I want to see this on every college campus,"...
- FDA sends warnings letters to companies making cancer cure claims (drugstorenews.com)
The Food and Drug Administration...posted warning letters addressed to 14 U.S.-based companies allegedly illegally selling more than 65 products that fraudulently claim to prevent, diagnose, treat or cure cancer. The products are marketed and sold without FDA approval, most commonly on websites and social media platforms...Consumers should not use these or similar unproven products because they may be unsafe and could prevent a person from seeking an appropriate and potentially life-saving cancer diagnosis or treatment…The illegally sold products cited in the warning letters...include a variety of product types, such as pills, topical creams, ointments, oils, drops, syrups, teas and diagnostics (such as thermography devices). They include products marketed for use by humans or pets that make illegal, unproven claims regarding preventing, reversing or curing cancer; killing/inhibiting cancer cells or tumors; or other similar anti-cancer claims.
- NASH: The next untapped pharma market gives investors many options (reuters.com)
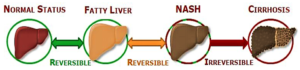
Large drugmakers with piles of cash are on the hunt for promising medicines being developed by small companies to treat NASH (non-alcoholic steatohepatitis), a progressive fatty liver disease poised to become the leading cause of liver transplants by 2020...The eventual market for the complex disease...is forecast to be $20 billion to $35 billion as populations with fatty diets increasingly fall victim to a condition with no approved treatments...Estimates for the prevalence of NASH in nations with fatty diets range from 5 to 20 percent of the population with up to 15 million potentially affected in the United States alone...Driven by the obesity and diabetes epidemics, the disease guarantees an enormous pool of patients for decades, making it a prime target for deals for promising therapies for NASH and its consequences - advanced fibrosis and liver-destroying cirrhosis…Drugmakers are taking a wide range of approaches to treat the complex disease, given multiple health issues among NASH patients that contribute to the liver damage, such as heart disease and diabetes...There are drugs targeting inflammation to prevent or reduce fibrotic scarring. Some address lipid regulation to reduce liver fat, while others attempt to directly halt or reverse fibrosis. And some companies are testing diabetes treatments to assess their ability to also improve NASH...The big sea change...is a fairly rapid acceptance of the fact that we're going to be seeking combination therapies since it's a disease that involves multiple pathways...whatever the mechanism is, it needs to yield decreased fibrosis...noting that progressing fibrosis is what ultimately causes serious health consequences...
- Clinical trial enrollment gets harder as patient populations shrink (biopharmadive.com)
...Enrollment is challenging across nearly all clinical trials. But for the rare disease variety, it’s often akin to finding a needle in a haystack — or in some cases a field of haystacks, if the illness is particularly uncommon. Though sponsors have over the years crafted electronic tools to resolve geographical barriers and reimbursement programs to alleviate financial burdens on patients, many of the studies’ inherent complications have yet to be fully remedied...In the most extreme cases, though, recruitment woes can squelch trials before they start or sap investor faith in a candidate...
Understanding the disease...Before researchers consider a patient for a clinical trial, they first need to determine whether he or she actually has the disease under investigation...having clinicians who understand a rare disease well enough to properly diagnose it — a resource often concentrated at universities or specialized research centers — can be a limiting factor…
Getting patients there -...identifying possible participants for a trial is the first hurdle in the race to enrollment, the next is getting them to sign on...
Making it all worth it -...Despite the immense preparation needed to enroll and conduct orphan drugs trials, many don’t have standardized endpoints...the right ones that would show the transformative nature of the therapy...
- In the debate over rising drug prices, both drugmakers and PBMs claim innocence (cnbc.com)
The battle over rising drug prices has become a full-blown he-said-she-said...Drug companies have pointed the finger at middlemen in the health-care system, saying they not only benefit from rising drug prices but contribute to their increases...Those middlemen — namely, pharmacy benefits managers (or PBMs) — have said the only parties responsible for drugs' list prices are the manufacturers...So who's right?...In a study released today, the PBM lobbying group, the Pharmaceutical Care Management Association, says an analysis it commissioned revealed no correlation between drug prices and rebates paid back to PBMs...The drug industry's lobby, the Pharmaceutical Research and Manufacturers of America...disagrees...
- U.S. Army can’t add a pricing safeguard to Sanofi’s Zika vaccine license, official says (fiercepharma.com)
Despite pushback from intellectual property activists...and high-profile politicians, a U.S. Army official dismissed concerns about a pending license transfer for a Zika vaccine candidate to pharma giant Sanofi...Responding to criticism of the proposed deal, the U.S. Army’s director of medical technology transfer at the Medical Research and Materiel Command, Barry Datlof, wrote...that the deal will comply with U.S. laws and will "be in the best interest of the U.S. government and the public…Originally developed by Army scientists, the immunization went into phase 1 testing with National Institutes of Health support. Sanofi joined the effort to prepare for phase 2 trials and to create a clinical development and regulatory strategy...KEI (Knowledge Ecology International )...started protesting the proposed license soon after the U.S. government disclosed the plan...Highlighting the extensive government funding for the program—$43 million so far, with the potential for more—KEI asked for pricing safeguards and argued that the license transfer would "not be legal" because it isn’t necessary to motivate Sanofi to develop the vaccine...Datlof said it’s not feasible for the Army to "define, implement and enforce 'affordable prices' or to set price controls for a potential vaccine that will require great investment and face high risk of failure."...the Army still plans to issue the license...
- GSK must pay $3 million in generic Paxil suicide lawsuit: U.S. jury (finance.yahoo.com)
GlaxoSmithKline must pay $3 million to a woman who sued the drug company over the death of her husband, a lawyer who committed suicide after taking a generic version of the antidepressant Paxil…The jury's award followed a trial in federal court...over the death of Stewart Dolin...who jumped in front of an oncoming commuter train in 2010 after taking a generic equivalent of GSK's Paxil...GSK maintains that because it did not manufacture or market the medicine ingested by Mr. Dolin, it should not be liable," GSK said. "Additionally, the Paxil label provided complete and adequate warnings during the time period relevant to this lawsuit...Dolin's lawyers had requested $39 million. They alleged GSK had evidence paroxetine increases the risk of suicide by older users by as much as 670 percent, yet failed to include that on the warning label...
- Meet the lawyer trying to pry drug pricing secrets out of Big Pharma (statnews.com)
He has a private jet, a pedigree of winning billion-dollar settlements, and the (sometimes grudging) respect of his adversaries. Now, he wants to become pharma’s latest headache...Class-action attorney Steve Berman is coming after a drug industry he says is "gouging" the American consumer. And his suits have the potential to crack the lid on the black box of drug pricing, shedding light on a secretive process that has sparked an escalating blame game between drug makers and the many middlemen in the US health care system...Berman sees the drug pricing system as a Rube Goldberg machine for extracting money from patients: Pharma sets a high price for a given medication, and then promises a big, undisclosed rebate to the pharmacy benefit managers who control which drugs get covered by insurers. As prices go up, so too do the secret rebates. Berman’s conclusion: The big guys get richer, and the patients pay the price...So he’s suing...
- 10 Reasons Merck Could Be the World’s Most Perfect Stock (fool.com)
The average investor has more than 5,000 publicly traded stocks on reputable U.S. exchanges to choose from. With that much hay in the barn, finding that needle can be quite difficult...But if you're looking for the world's most perfect stock, perhaps your search might end with pharmaceutical giant Merck. Since Jan. 1970, including splits, shares of Merck have risen by a brisk 4,320%! Though it might appear that its best days are in the rearview mirror after such large gains, there are plenty of reasons to believe it still has ample upside opportunity...Here are 10 reasons Merck just might be the world's most perfect stock.
- A leading cancer immunotherapy
- A long-tail hepatitis C opportunity
- Biosimilars
- Bolt-on acquisitions
- A premier pharmaceutical pipeline
- Tight cost controls
- Strong pricing power
- Steady growth in animal health
- Shareholder yield
- Valuation
- Canada Cannot Be America’s Drugstore (realclearhealth.com)
The drug importation debate, sparked by Sen. Bernie Sanders’ introduction of legislation that would allow Americans and pharmacies to import medicines not approved by the FDA from foreign countries through and including Canada, is largely focused on one key question: Are medications imported from outside the FDA’s jurisdiction safe for consumers?...Clear and compelling evidence tells us the answer is no...we haven’t even begun to address concerns from the Canadian side of this issue...Canada does not have the drug supply nor the drug distribution system to meet even a small level of U.S. demand. Additionally, it is highly unlikely that the pharmaceutical industry will increase the supply necessary for Canada to become a major exporter...Wouldn’t U.S.-based pharmaceutical companies simply increase their supply to Canada in order to meet this increased demand? The answer is likely "no," as it’s not a practical market-based response...If drug importation is legalized, the U.S. and Canadian pharmaceutical markets essentially become one. It would not be in the fiduciary or long-term interests of any pharmaceutical company to increase its supply to Canada at artificially low prices set by government fiat, which would only reduce demand for medications sold in the United States at market-based prices...If Canadian pharmacies can’t get the drugs from U.S. manufacturers to meet both domestic needs and export demands, they will turn to other countries to secure these supplies...drugs will instead flow from countries like Russia and India, where counterfeiting is ubiquitous..