- Genentech urges broader adoption of value-based payment models (mmm-online.com)
Researchers involved in an outcomes-based pilot between Genentech and Priority Health, a Michigan-based health insurer, argued that broader industry participation is needed for these agreements to be successful...In a study...the drugmaker and the insurer detailed the terms and challenges of the value-based program for Avastin…They also urged other organizations to continue to engage in value-based contracts, despite their inherent complexity...There are several reasons why these kinds of contracts, usually involving drugmakers taking on some degree of financial risk and payers promising prescription volume in turn for drugs that yield better outcomes for patients, have been gaining in popularity…
- The U.S. health system as a whole has been moving away from a volume-based, or fee-for-service, system to a value-based model…
- Healthcare decision makers are challenging pharmaceutical manufacturers to demonstrate the value of their medicines, not just in terms of clinical efficacy but also in terms of economic and quality-of-life outcomes…
- ...what five qualities make for a successful contract? Leadership commitment, the choice of drug (outcomes should be able to be proved within a year), agreed-upon definitions and metrics, finding a way to track data in the simplest way possible, and figuring out to address the government rules about pricing…
- Report: Hanmi’s failure to report patient death in olmutinib study broke South Korea’s medical laws (endpts.com)
A Korean patient taking Hanmi’s cancer drug olmutinib died from a rare case of Stevens-Johnson syndrome a full 14 months before it was reported to the country’s health authorities, triggering a scramble that ultimately caused its partner Boehringer Ingelheim to abruptly withdraw from their $730 million partnership on the drug…Korean officials determined that Hanmi broke two medical laws related to monitoring and reporting clinical trials…the case dates back to the death in July 2015, when a patient taking olmutinib and two other drugs died from the lethal skin condition...the case dates back to the death in July 2015, when a patient taking olmutinib and two other drugs died from the lethal skin condition. The physicians involved in the case...reported the case to a monitoring agency...it was not reported as an unexpected serious adverse event...Hanmi and the monitoring agency finally reported the death to health officials in September, 2016...only after Hanmi said it first became aware of the incident...The case spurred accusations...that the influential Hanmi...had intentionally delayed reporting the death so it could get the drug approved in Korea.
- Health Canada to increase transparency (cmaj.ca)
Health Canada plans to increase transparency into its regulatory process for drugs and medical devices by publicly releasing clinical information submitted by companies who receive market authorization...Opening access to clinical information used to support the authorization has widespread health system benefits and can help Canadians make informed decisions about their health…Under the proposed transparency policy, the information will be kept confidential during the regulatory process but disclosed if the drug or medical device is approved for sale. According to Health Canada, this information is "far more comprehensive" than data available in sources such as medical journals and clinical trial registries...Companies will be able to request, however, that certain parts of the clinical information they submit be exempt from public release...Health Canada states that this transparency initiative will increase public confidence in the drug and medical device review process; help patients and health care providers make more informed decisions about drugs and medical devices; avoid duplication in clinical trials; and make clinical trial data available for secondary analysis to advance medical science...
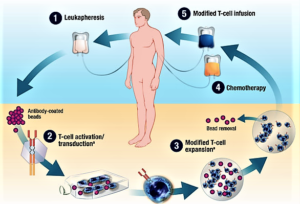
- The First CAR-T Drugs Have Left the Gate (fool.com)
Investors should keep an eye on this promising way to treat cancer...For all the talk about biotechs being nimble, it's a big pharma that looks like it'll be the first company to launch a chimeric antigen receptor T-cell (CAR-T) product...Novartis announced last week that the Food and Drug Administration accepted its application to market tisagenlecleucel-T...in patients with B-cell acute lymphoblastic leukemia who are relapsed and refractory to other therapies...A few days later, Kite Pharma completed its application for axicabtagene ciloleucel...Kite's application could be accepted early, putting it less than two months behind Novartis…Since CAR-T therapies are personalized treatments that have to be made individually for each patient, they're likely to be expensive to produce and therefore require a premium price. The first company to get a CAR-T therapy approved will set the price, which later companies may have to match unless they can justify a higher price with higher efficacy...With prices that will probably exceed those of current cancer treatments, investors should expect some pushback from insurers. One way Novarits and Kite can get around the cost issue is by offering money-back guarantees...Kite's and Novartis' CAR-T therapies are just the tip of the iceberg for this new way to treat cancer...
- Is prescription pet food better for your pet – or for the companies that make it? (sacbee.com)
A North Carolina law firm is one of the primary drivers of a class action lawsuit that alleges that prescription dog and cat food is a marketing scheme devised by pet food companies to pump up their profits...At the heart of it, the world’s largest pet food manufacturers are requiring that certain pet foods be sold by prescription even though there is no legal requirement for that prescription…requiring a prescription from a veterinarian misleads consumers, providing cover that enables pet food companies to charge excessive prices...Prescription pet food contains no drug or other ingredient not also common in non-prescription pet food...Defendants in the civil lawsuit...include the companies behind four brands of prescription pet foods that dominate the market: Hill’s Prescription Diet, Purina Pro Plan Veterinary Diets, Royal Canin Veterinary Diet and Iams Veterinary Formula...the lawsuit argues, the companies “control the sale of prescription pet food from manufacture to veterinarian to retail, which has allowed their deception and price-fixing conspiracy to be implemented and perpetuated with minimal risk of detection or defection.”...prescription pet foods “have not been evaluated by the FDA for safety, efficacy, or nutritional adequacy.”...Prescription pet food accounts for about 5 percent of the $24 billion in pet food sold in the U.S. each year...or more than $1 billion a year...Pet food...“is the only food on the planet that’s allowed to make a health claim” by the FDA...
- University of California files appeal over CRISPR patents (reuters.com)
CRISPR that favored the Broad Institute…Jennifer Doudna of the University of California...and Emmanuelle Charpentier of the University of Vienna were first to apply for patent in 2012 after discovering how the primitive bacterial system...could be used to edit genomes in simple pieces of DNA…A team at the Broad Institute led by...Feng Zhang applied for a separate patent six months later, but paid for a fast-track review process, which landed them the first CRISPR patent in 2014. The Broad's patents were for showing that the CRISPR system could be used to edit more advanced, eukaryotic cells, including animal and human cells...In its...decision, an appeals board...determined that the Broad's CRISPR patents "did not interfere" with those awarded to the UC because they were sufficiently different, allowing them to stand...In the appeal...UC is seeking a reversal of the decision, which ended before actually determining who invented the use of CRISPR in eukaryotic cells. Major commercial applications of CRISPR are expected to be in eukaryotic cells...geneticist George Church said he expects the disputes will end in cross-licensing...I'm not that interested in the details of who pays who what. We're all going to do very well, including the patients. That was evident from the very beginning…
- IBM receives patent that could accelerate discovery of more effective and safer drugs (drugstorenews.com)
IBM...announced its scientists have been granted a patent ("Method and system for exploring the associations between drug side-effects and therapeutic indications") on machine learning models to predict therapeutic indications and side effects from various drug information sources, which could accelerate discovery of more effective and safer drugs...IBM Research has implemented a cognitive association engine to identify significant linkages between predicted therapeutic indications and side effects, and a visual analytics system to support the interactive exploration of these associations...this approach could help researchers in pharmaceutical companies to generate hypotheses for drug discovery...strongly correlated disease-side-effect pairs identified by the patented invention could be beneficial for drug discovery in many ways. One could use the side-effect information to repurpose existing treatments (e.g. drugs causing postural hypotension could be potential candidates for treating hypertension). If a new drug is being designed for a disease that is strongly correlated with severe side effects, then special attention could be paid to controlling the formulation and dosing of the drug in the clinical trials to prevent serious safety issues...Lack of efficacy and adverse side effects are two of the primary reasons a drug fails clinical trials, each accounting for around 30% of failures...Computational models and machine learning methods that can derive useful insights from large amounts of data on drugs and diseases from various sources hold great promise for...improving the drug discovery process.
- WFI System Meets European Requirements (pharmtech.com)
Bosch’s new system for the production of water for injection, which is used in solutions for injection and infusion, uses membrane processing and ultrafiltration. Previously, it was only possible to produce WFI for the European market using distillation...the revised European Pharmacopeia monograph allows, for the first time, WFI to be obtained using other comparable processes...Pharmatec designed a WFI production unit using membrane processes that meets the requirements...from the European Medicines Agency...The generation unit employs cold membrane processing methods (i.e., reverse osmosis and electrodeionization), with an additional ultrafiltration step...Monitoring all the processes in the production chain is essential to guaranteeing the microbial quality of the water, which is why we fitted the unit with various sensors and measuring devices to monitor the water quality throughout the process and to check the individual steps...
- Maryland lawmakers approve bill to fight drug price-gouging (reuters.com)
Maryland lawmakers have passed a first-in-the-nation measure that lets the state attorney general sue generic drug makers that sharply raise prices in a move aimed at fighting what legislators call "price-gouging."...The bill overwhelmingly was approved by the Democratic-controlled legislature on Monday and hailed by Maryland Attorney General Brian Frosh as a way to check sharply higher prices for crucial generic drugs…Governor Larry Hogan, a Republican, has not said whether he would sign the legislation...The bill allows Maryland's state authority on Medicaid...to let the attorney general's office know when it sees patients being charged an "unconscionable increase" for essential generic drugs...The attorney general could then seek an explanation from the manufacturer and sue to protect consumers, with a fine of up to $10,000 for each violation. A judge could order the company to reverse its price increase.
- Nevada coalition wants drugmakers to reimburse diabetics (reviewjournal.com)
Nevadans dismayed by soaring drug prices told lawmakers...the state should take unprecedented action to curb costs...A coalition of hotel and casino owners, union leaders and Democratic lawmakers are attempting to mandate pharmaceutical companies refund people for overpriced American insulin...The bill targets diabetes medications...would require drugmakers to reimburse Nevada patients and insurers for what they pay above the highest price in other developed countries for the same prescriptions. Drugmakers would also have to reimburse people if American insulin prices increase more than inflation would suggest they should...Senate Bill 265 would require manufacturers to disclose the costs of all research, materials, manufacturing and administrative expenses that go into producing drugs for diabetics. Additionally, it would force them to notify government health agencies and insurance companies 90 days before they increase prices above the preceding year’s inflation rate...Nevada’s proposal would also have the state license all pharmaceutical sales representatives...Implementing that program in Nevada would cost about $350,000 and upkeep would cost $125,000 annually after that, according to the state Department of Health and Human Services...