- Harvard Pilgrim bets on value-based drug payments with new deals (biopharmadive.com)
As high pharmaceutical prices strain budgets of payers and patients alike, Harvard Pilgrim is betting so-called pay-for-performance deals will help lower drug costs and improve patient outcomes...The...health insurer announced...it had inked two separate agreements with Amgen and Eli Lilly that tie the costs of Amgen's Enbrel (etanercept) and Lilly's Forteo (teriparatide) with either performance or patient adherence...a number of large drugmakers have begun pushing value- or outcomes-based payments as a potential solution to blowback on rising drug prices. Under these types of deals, insurers and other payers would pay less for drugs that fail to meet specified benchmarks. Drugmakers, on the other hand, would be rewarded for innovative medicines that improve outcomes.
- A Call For The FDA To Raise The Bar For Clinical Trials Is Flawed (forbes.com)
As the new Trump administration begins to take shape, there are concerns that new leadership at the FDA will call for less bureaucracy and red tape, thereby lowering the bar for new drug approvals. The view is that onerous regulations are preventing life-saving medications from getting to those who most need them. Lowering the standards for drug approvals may indeed allow for earlier patient access to drugs but...liberalization of current regulations would be a mistake. Such actions will unleash drugs that will ultimately prove to be ineffective and, in some cases, harmful rather than beneficial...the specter of deregulation, comes a call to increase the requirements for allowing early clinical trials of exploratory new medicines...Trials of ineffective therapies place burdens on society...Drug development soaks up financial and research resources...expenses wasted on ineffective therapies are often passed on to healthcare systems in the form of higher drug prices. The biopharmaceutical industry is testing “duds” and wasting precious resources because of unreasonable hope for a major commercial payoff...The biopharmaceutical industry is already heavily regulated. Current standards shouldn’t be lowered...
- Pharma lobby launches crusade against Cuomo’s ‘flawed’ drug pricing plan in NY (fiercepharma.com)
Pharma has heard enough of New York Governor Andrew Cuomo’s ideas for lowering drug prices. Just a month after the governor unveiled his plan, the industry’s trade group is rolling out a counteroffensive urging state officials to “Prescribe Real Solutions”...Gov. Cuomo’s plan would create a board to rule on “fair prices” for certain expensive drugs. Then, the state would mandate that Medicaid refuse to pay more than that for those meds...Cuomo's proposal also factors into private market sales. If a pharma company sold into New York at a higher price than the board’s "fair price," the state would collect a surcharge...PhRMA is having none of that. The group’s Prescribe Real Solutions website asks the public to “demand real solutions” from their lawmakers...PhRMA’s deputy vice president for state advocacy in New York, maintains that Cuomo's proposal "does nothing to improve patient access to medicines, bring life-saving drugs to the market or create jobs" in the state..."This is not the time for unworkable proposals that would create new bureaucracies without actually addressing any of the factors that impact the prices paid by patients,"...
- Pharma Lobby May Boot Company That Introduced High-Priced Drug (bloomberg.com)
The Washington lobbying group for the pharmaceutical industry may kick out a drugmaker that introduced a high-priced version of an older medicine that had been available much more cheaply...Pharmaceutical Research and Manufacturers of America...is reviewing its membership criteria after Marathon Pharmaceuticals LLC...said it would introduce a drug for a rare, deadly muscle disease at the cost of $89,000 a year. A version of the drug had been available for about $1,000 a year overseas and many U.S. patients were allowed to import it...Marathon’s...recent actions are not consistent with the mission of our organization...The leadership of the PhRMA board of directors has begun a comprehensive review of our membership criteria to ensure we are focused on representing research-based biopharmaceutical companies that take significant risks to bring new treatments and cures to patients…
- Gilead snaps up Sarepta’s priority review voucher for $125M (biopharmadive.com)
Hoping to speed up future regulatory review for its drug candidates, Gilead has agreed to buy a priority review voucher from Sarepta Therapeutics for $125 million...Sarepta had obtained the voucher — a transferrable credit designed to speed review by the Food and Drug Administration by four months — for winning approval of its disease-modifying treatment for Duchenne muscular dystrophy last year...Sale of the voucher, albeit for a lower price than some had expected, will bolster Sarepta's cash position and help fund clinical development of Sarepta's pipeline and manufacturing scale-up…For Gilead, the voucher could be used to speed review for one of its several late-stage candidates. The $125 million is a relative bargain as well, compared to both the recent prices paid by Sanofi and AbbVie as well as to Gilead's large cash hoard.
- Cheaper drugs from Canada? Pharma despises the idea, but top senators are pushing HHS chief to try it (fiercepharma.com)
Instead of being gouged on the prices for old drugs, Americans could import certain meds from Canada if a bipartisan group of senators get their way...Sens. Klobuchar, Chuck Grassley, and John McCain...are pushing President Donald Trump’s new Department of Health and Human Services Secretary, Tom Price, to authorize cheap drug imports under strict circumstances...Their bill, the Safe and Affordable Drugs from Canada Act, would require the FDA to set up a “personal importation program” to allow individuals to import 90-day supplies of their meds from Canadian pharmacies...To qualify for importation, the senators' proposal would require products to be off patent or no longer marketed in the U.S. by the original developer. They'd need to be free of direct competition and to have seen a “significant, unexplained” price hike. The imported product would also need to be produced by a reputable company. The bill additionally calls for fast-track FDA reviews of competing drugs...Imported drugs wouldn’t “be subject to the U.S. Food and Drug Administration’s robust safety requirements, and there would be no way to trace the country of origin for the imported products...
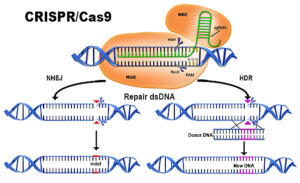
- Broad Institute notches win in CRISPR patent battle (biopharmadive.com)
In a hotly anticipated judgment, the U.S. patent office...ruled that key patents held by the Broad Institute of MIT and Harvard on the gene-editing technology CRISPR-Cas9 do not overlap with competing claims from the University of California...UC may now receive a patent covering broader claims on the revolutionary tech...the Patent Trial and Appeal Board ruled the claims of UC and the Broad to be patentably distinct subject matter, leading the three judges to declare a judgment of non-interference...Put into layman's terms, the claims of the UC and the Broad are distinct enough so as to be separately patentable and therefore don't overlap with each other. This effectively supports the Broad's existing patents, while sending the UC's pending patent application back to the initial patent examiner...UC with a patent on use of CRISPR in all cells and the Broad with a patent for use of CRISPR specifically in eukaryotic cells...If that were to happen, biotechs operating in the space could end up having to license the tech from both UC and the Broad.
- Trump is promising big changes at the FDA — here’s how drugs are approved today (businessinsider.com)
At least one of President Trump's possible picks to head the Food and Drug Administration has a radical idea for when drugs should come to market...Jim O'Neill, managing director at Mithril Capital, has said that he is in favor of approving drugs that are proven to be safe, even before they're shown to be actually effective...Regardless of who Trump picks in the end, his interest in cutting regulation at the FDA is clear..."We're going to be cutting regulations at a level that nobody's ever seen before," Trump said in a meeting with pharma executives...As it exists right now, the FDA approval process can be a long and expensive…
- Genentech lobbyist’s life and death makes pharma waves (fiercepharma.com)
A Genentech lobbyist who generated enormous sales for the company allegedly kept millions in consulting payments for himself and ended his own life after authorities and the company began looking into the charges...During his time lobbying for Roche and its Genentech unit, Evan Morris’ tactics boosted the company's top line by hundreds of millions of dollars, with the company's flu drug Tamiflu and cancer treatment Avastin among the products that benefited...Under investigation for embezzlement in July 2015, Morris committed suicide at a Virginia golf course...Morris’ lavish life, ...featured expensive houses, wines, influential contacts and more, before it came quickly crashing down. An obituary for Morris simply says he “resided in Alexandria, VA.” He was born in 1977 and died on July 9, 2015, the obituary says.
- Pfizer, Flynn appeal against UK regulator’s £89.4m fine for epilepsy drug price hike (manufacturing.pharmaceutical-business-review.com)
Pfizer and Flynn Pharma have appealed against the £89.4m ($111m) fine imposed by the UK Competition and Markets Authority (CMA) for charging excessive and unfair prices for an anti-epilepsy drug...Pfizer has been fined £84.2m($104m) and Flynn Pharma £5.2m ($6.4) after they increased prices by as much as 2,600% in September 2012...The companies have separately filed appeals in the Competition Appeal Tribunal...Both Pfizer and Flynn accused the UK watchdog of ignoring market realities when it ruled that the price Flynn paid to acquire the anti-epilepsy drug from Pfizer and the price it charged from the National Health Service (NHS) were both excessive...The CMA had stated that owing to the unreasonable price increase of the critical drug, the NHS’ annual expenditure to buy the phenytoin sodium capsules from Flynn had shot up to around £50m ($62m) in 2013 from the 2012 figure of £2m ($2.4).