- The top 10 drug launches of 2017 (fiercepharma.com)
- After an unusually slow year for new drug approvals—the FDA greenlighted just 22 meds in 2016—it remains to be seen whether drugmakers can do much better in 2017. One thing’s for sure, though: No matter what total the industry tallies up this year, the crop will bring some would-be blockbusters and market disrupters.
At the top of the list...is Ocrevus (ocrelizumab), the Roche multiple sclerosis drug that’s promising to shake things up in more ways than one...
Sanofi and Regeneron hot-shot Dupixent (dupilumab) could make a big splash in severe atopic dermatitis, assuming payers don’t get in the companies’ way.
Ditto for Biogen's Spinraza, which in December became the first FDA-approved product to treat spinal muscular atrophy—but whose high sticker could raise eyebrows at a time when U.S. President Donald Trump is threatening pricing action.
Tesaro and Neurocrine are looking for their first-ever FDA approvals, in breast cancer pill niraparib and tardive dyskinesia therapy Ingrezza, respectively.
...Kite Pharma is aiming to get the first-ever CAR-T cancer drug to market, with a candidate, KTE-C19, that the oncology community will be watching closely as the next big thing in immunotherapies.
Novartis’ ribociclib...which aims to challenge Pfizer’s Ibrance in the CDK 4/6 breast cancer space.
...semaglutide, Novo Nordisk's weekly GLP-1 drug, a would-be successor to the company's blockbuster Victoza…
- After an unusually slow year for new drug approvals—the FDA greenlighted just 22 meds in 2016—it remains to be seen whether drugmakers can do much better in 2017. One thing’s for sure, though: No matter what total the industry tallies up this year, the crop will bring some would-be blockbusters and market disrupters.
- Contentious flu vaccine policies at hospitals are based on flawed research, study says (statnews.com)
It’s an edict that comes out every autumn in many hospitals: If health care workers don’t get a flu shot, they will face consequences. Some make vaccinations a condition of employment. Others require unvaccinated staff to wear surgical masks near patients for weeks during flu season...But a new study is calling into question the scientific evidence underpinning these increasingly common hospital policies — and could fuel challenges to the contentious orders...The study...concludes that the research used to justify mandatory flu shots for health sector workers is flawed, and that the policies cannot plausibly produce the benefits that had widely been assumed...the bottom line of our paper is to say there is no valid scientific evidence, even now, underpinning enforced health care worker immunizations...trying to mandate that health care workers take the flu vaccine is well-intentioned, but is taking away resources and the focus on what our main priority needs to be, which is getting a better influenza vaccine...
- Financial ties between researchers and the pharmaceutical industry might distort clinical trial results (pharmaceutical-journal.com)
Analyses of data from 195 clinical trials show that financial ties between researchers and the pharmaceutical industry were associated with a 3.57 greater odds of a positive trial outcome...Clinical trials involving principal investigators with financial ties to the pharmaceutical industry are more likely to report positive results...The researchers...say their findings — which held even after accounting for the influence of study sponsorship — suggest that financial ties could be leading to bias in the clinical evidence base...Given the importance of industry and academic collaboration in advancing the development of new treatments, more thought needs to be given to the roles that investigators, policy makers, and journal editors can play in ensuring the credibility of the evidence base...
- Why biosimilars could be market disruptors (biopharmadive.com)
Just as generic drugs revolutionized the small molecule space, biosimilars are poised to have a major impact on the development and commercialization of biologics. These highly similar, but not necessarily interchangeable drugs, have been part of the European drug market for years, but are just starting to emerge in the U.S. While still early days, the developing biosimilars market is one to watch.
- Inherited pay-for-delay penalties are getting expensive for ‘cash-strapped’ Teva (fiercepharma.com)
Teva is eager to start moving in the right direction after a particularly rocky 2016. Problem is, it’s still paying for pay-for-delay decisions made by its products’ previous owners...the generics giant last week agreed to a $225 million settlement with a group that picked up Bayer antibiotic Cipro...Also...the FTC refiled charges against Watson and former parent Actavis, claiming they illegally blocked a lower-cost generic version of Endo’s Lidoderm after entering into a pay-for-delay pact with Endo...All things considered, “the cumulative sum of fines is getting noticeable”—especially for “cash-strapped” Teva...The Israeli company earlier this month walked down its previously outlined 2017 guidance by more than $1 billion after new 2016 launches didn’t hit their marks. And some analysts think Teva’s new revenue forecast—a range of $23.8 billion to $24.5 billion--may still be too high...
- Trump’s Crusade on Drug Pricing Puts Both Parties on the Spot (bloomberg.com)
Donald Trump has a chance to rally his core supporters as well as left-wing Democrats, wrapping himself in the populist flag to take on the politically powerful drug industry...He is vowing to keep a campaign pledge to push legislation allowing Medicare to negotiate prescription drug prices, a practice currently prohibited by law. Proponents say this would reduce drug prices and Medicare costs for the federal government. Medicare pays for about 29 percent of prescription drugs in the U.S. and would have considerable leverage...If Trump goes all out on this issue, it will be near impossible for….Democrats to side with the industry over a Republican president whom they accuse of representing the interests of the rich.
- ISMP Report Questions Safety of Hepatitis C Drugs (pharmacytimes.com)
A recently-published Institute for Safe Medication Practices report details updated concerns about newer drugs used to treat hepatitis C virus. The report, which is published in ISMP’s QuarterWatch, highlights concerns about the safety of HCV drugs in light of findings that point to associated liver failure and injury...The FDA designated 46 different drugs in development as breakthroughs last year, and although some of these medications have resulted in cure in an estimated 9 out of 10 patients treated, ISMP has identified hundreds of patients that reported antiviral failure...Liver failure occurred more frequently in men (55%) and in patients with a median age of 61 years old. Thirty one percent of cases resulted in death...Although the report questions the safety of these drugs, experts argue that the findings are inconclusive and prescribers should not be influenced by the results...These new data raise more questions than they resolve about the adverse effects of direct-acting antiviral drugs...A better understanding of what is occurring in hundreds of additional liver failure cases should be a priority for further investigation...
- Is the PCSK9 patent fight giving Amgen’s Repatha a boost? Script numbers say so (fiercepharma.com)
As Sanofi and Regeneron scramble to keep their PCSK9 cholesterol drug Praluent on the market, Amgen’s rival drug Repatha already appears to be chipping away at its market share...For the week of January 20, Repatha’s prescription total hit 3,231, ahead of Praluent’s 2,859...The Praluent partners are of course embroiled in a patent fight against Amgen, which claims that Sanofi and Regeneron knowingly stepped on its patents in developing Praluent. A district court jury sided with Amgen, and the judge in the case granted Amgen’s request for an injunction that would push Praluent off the market...Sanofi and Regeneron hope that the threat is short-lived; they’re trying to persuade the U.S. Circuit Court of Appeals in Washington, D.C., to put off the injunction while their patent appeal is heard. In their request for a stay, the two companies argued that they have a good chance of winning their appeal and that pulling Praluent in the meantime would do great damage to Regeneron.
- Walgreens, Rite Aid amend deal to $7 per share, extend end date (drugstorenews.com)
Walgreens Boots Alliance and Rite Aid on Monday morning extended their previously announced definitive merger agreement under which Walgreens Boots Alliance will acquire all outstanding shares of Rite Aid...The retail pharmacy operations also restructured a new deal that would value Rite Aid at between about $6.8 billion and $7.4 billion, depending on required store divestitures, down from an initial acquisition cost of $9.4 billion...Under the terms of the amendment, the parties have agreed to reduce the price for each share of Rite Aid common stock to be paid by Walgreens Boots Alliance...price will be a maximum of $7 per share and a minimum of $6.50 per share...Walgreens Boots Alliance will be required to divest up to 1,200 Rite Aid stores and certain additional related assets if required to obtain regulatory approval...
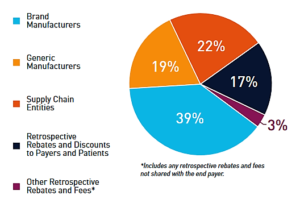
- PhRMA-backed report analyzes ‘complex process’ of drug pricing (biopharmadive.com)
The PhRMA-backed report made the case that drugmakers are not solely to blame for ballooning treatment prices, which have come under intense scrutiny from state and federal legislatures, industry executives and research organizations...As competition in the pharmaceutical marketplace has increased in recent years, brand manufacturers have been making larger payments for market access to their medicines...Government-mandated discounts and fees have also increased over the last five years. Many of these discounts are not plainly visible, leading to misperceptions about the relative share of gross and net drug expenditures realized by brand manufacturers...each player has taken home large chunks of the drug sales revenue as well...non-manufacturers, which include wholesalers, PBMs, health plans and pharmacies, snag 42% of gross drug spending — which doesn't take into account any type of discount — from the initial point-of-sale payment for a drug by a payer or patient...Branded drugmakers take home 39% of those gross expenditures, with generics manufacturers gobbling up 19%...