- Senate Aims to Stop Firms From ‘Buying Up Drugs and Jacking Up Prices’ (nytimes.com)
On the heels of headline-grabbing price spikes on prescription drugs, a bipartisan Senate report on Wednesday will call on Congress to take action to prevent huge, unjustified cost increases on decades-old prescription medicines that have no competition...The Senate Special Committee on Aging, reporting the results of a yearlong investigation, said that some drug companies behaved like hedge funds because of the influence of “activist investors.” These companies, the committee said, have developed a “business model that harms patients, taxpayers and the U.S. health care system.”...Our report does not recommend that the government get into the business of setting prices for prescription drugs...We think that would have a harmful impact on the pipeline of innovative drugs...I don’t think we can ignore the market failures that have occurred. The answer is to figure out how we can revitalize the market so that generic drug producers have incentives to compete with companies that are buying up drugs and jacking up prices to make quick, exorbitant profits...
- Looking For Bargains, Many Americans Buy Medicines Abroad (npr.org)
As drug prices have spiraled upward, tens of millions of generally law-abiding Americans have committed an illegal act in response: They have bought prescriptions medicines outside the U.S. and imported them...It's no secret that some Americans regularly buy prescription drugs on the Internet or while traveling abroad. But the popularity of the approach is underscored by the results of a Kaiser Family Foundation poll conducted in November...Eight percent of respondents said they or someone in their household had imported a drug at some point...The Kaiser poll queried a nationally representative sample of 1,202 adults...The Food and Drug Administration has cautioned that many online pharmacies aren't what they seem to be. An international crackdown in 2014 found that many packages of medicines purportedly from Australia, Canada, New Zealand and the U.K. contained drugs from other countries, including India, China and Laos...imported medications could be inferior or expired. Some could be counterfeits. But many medicines purchased from another country are the same as the ones patients buy in the U.S...When purchased outside the country, many prescription medicines cost half or less than they do in the U.S...it is generally illegal for Americans to import drugs into the U.S. for personal use. The law isn't rigorously enforced, in part, because it is difficult to monitor the entry of medicine in suitcases and small packages...in 2015 the FDA implemented a rule that would give government border inspectors expanded authority to destroy drugs imported for personal use at their point of entry.
- Five Industry Trends for U.S. Drug Wholesalers in 2017 (drugchannels.net)
Here are five significant industry trends that will substantially impact the drug wholesaling industry in the coming years.
- U.S. pharmaceutical spending – and drug prices – are still growing. - We project that in 2016, for the first time, the Big Three wholesalers’ combined drug distribution revenues will exceed $400 billion.
- The consolidation of pharmacy and provider markets shows no sign of slowing - Mergers and acquisitions among pharmacies and payers are pressuring wholesaler margins, especially as the acquiring companies consolidate buying power.
- Payers and manufacturers are narrowing drug channels, pressuring wholesalers’ profitability - Narrow pharmacy networks – either preferred or limited models – are now a widely accepted element of pharmacy benefit design.
- Large pharmacies have entered into multifaceted partnerships with wholesalers – This...consolidation is creating a channel supply alignment among the largest wholesalers, largest PBMs and largest retail chains.
- Biosimilars are unlikely to provide superior profits to wholesalers. - Competition between a biologic drug and biosimilar is therefore likely to resemble brand-to-brand competition rather than brand-to-generic competition.
- Merck wins $2.54 billion in hepatitis C drug trial against Gilead (reuters.com)
Merck & Co...was awarded $2.54 billion in royalties by a federal jury in a patent lawsuit against Gilead Sciences Inc over Gilead's blockbuster hepatitis C drugs Sovaldi and Harvoni...The jury in Delaware reached the verdict following a nearly two-week trial, finding that a patent acquired by Merck in 2014 on hepatitis C treatments was valid...Harvoni and Sovaldi have drawn attention for their breakthrough success in curing hepatitis C in more than 90 percent of patients, and for their high cost...Harvoni's list price is $1,125 per pill and $94,500 for a 12-week regimen. Foster City, California-based Gilead, one of the world's largest biotechnology companies, made nearly $20 billion on the two drugs in 2015.
- NPAC advocates separation of medical and recreational cannabis (drugstorenews.com)
The Neighbourhood Pharmacy Association of Canada...applauded the federal government's Task Force on Marijuana Legalization and Regulation for recognizing the need to preserve the system of medical cannabis separate from the recreational system...the Task Force clearly noted Neighbourhood Pharmacies' proposition that medical cannabis is a serious drug and that Canadians expect that strength and packaging standards applied to prescription medications be applied to cannabis...We're looking forward to meeting with provincial pharmacy regulatory authorities, the pharmacy community and lawmakers to shape retail rules...We will continue to advocate for patient centric regulations that bring those using medical cannabis into the circle of care…
- What do new FDA letters dinging ‘misleading’ pharma TV ads mean for the industry? (fiercepharma.com)
FDA admonishment of Celgene and Sanofi for DTC TV ads has industry insiders checking the tea leaves for what the letters might signal. The upshot? Possibly an end to overly happy and energetic lifestyle pharma ads...The main complaint in both untitled letters from FDA drug marketing enforcer OPDP (Office of Prescription Drug Promotion) —to Celgene for oral psoriasis treatment Otezla and Sanofi for diabetes drug Toujeo—is distracting music and visuals that interfere with consumers fully understanding the risk information. Both ads have been running since this summer...These commercials are not much different from many other upbeat DTC ads with music...If they are cracking down on these two, then they have decided to look at all of these type of ads. FDA is very subjective about these kind of violations...Sanofi told FiercePharma in an interview last week that it is working to pull the Toujeo TV ads, while Celgene said more broadly that it will work with the FDA to address its concern.
- NACDS hails rule relaxing access to pharmacy rewards (chaindrugreview.com)
The Department of Health and Human Services is giving pharmacies more flexibility to extend their reward and loyalty programs to beneficiaries in government programs such as Medicare and Medicaid…it will allow government program beneficiaries to enjoy access to programs voluntarily implemented by pharmacies, which reduce health care costs, improve quality and promote patient health...This is a very positive step forward for pharmacy patient care...and of the expanded innovative programs that pharmacies provide...Programs vary in their designs but often include cost savings or other rewards for filling prescriptions or engaging in other health-related activities, such as health screenings, medication adherence programs or healthy lifestyle behaviors…
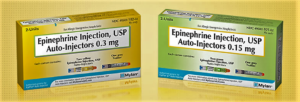
- Mylan launches first generic EpiPen as state AGs sue generics makers for price collusion (drugdeliverybusiness.com)
Mylan launched its 1st generic EpiPen emergency allergy treatment 1 day after attorney generals from 20 states filed a civil complaint against Mylan, Teva Pharmaceuticals, and 4 other generic drug makers. The complaint alleges that the companies co-conspired in informal gatherings, calls and text messages to fix the price for glyburide...and doxycycline hyclate...The generic EpiPen will sell for $300 per two-pack, which is a 50% discount compared to the price of the brand name device...Mylan CEO Heather Bresch said in a statement...“Unfortunately, families will continue to face sticker shock for medications and may be forced to make difficult choices until the pharmaceutical pricing system is reformed to address the increasing shift of costs directly to consumers.”...The 4 other companies targeted by the complaint are Heritage Pharmaceuticals, Aurobindo Pharma, Citron Pharma and Mayne Pharma...
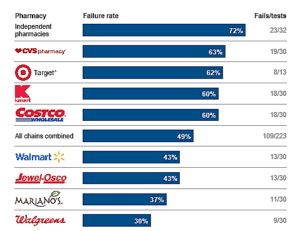
- Pharmacies miss half of dangerous drug combinations (chicagotribune.com)
In the largest and most comprehensive study of its kind, the Tribune tested 255 pharmacies to see how often stores would dispense dangerous drug pairs without warning patients. Fifty-two percent of the pharmacies sold the medications without mentioning the potential interaction, striking evidence of an industry-wide failure that places millions of consumers at risk...CVS...had the highest failure rate of any chain in the Tribune tests, dispensing the medications with no warning 63 percent of the time. Walgreens...had the lowest failure rate at 30 percent — but that's still missing nearly 1 in 3 interactions... There is a very high sense of urgency to pursue this issue and get to the root cause...test after test, other pharmacists dispensed dangerous drug pairs at a fast-food pace, with little attention paid to customers. They failed to catch combinations that could trigger a stroke, result in kidney failure, deprive the body of oxygen or lead to unexpected pregnancy with a risk of birth defects...The Tribune study...exposes fundamental flaws in the pharmacy industry. Safety laws are not being followed, computer alert systems designed to flag drug interactions either don't work or are ignored, and some pharmacies emphasize fast service over patient safety. Several chain pharmacists, in interviews, described assembly-line conditions in which staff hurried to fill hundreds of prescriptions a day.
- Hospital Impact: Drug companies win, patient safety loses with 21st Century Cures Act (fiercehealthcare.com)
The 21st Century Cures Act passed the House and Senate with overwhelming bipartisan support and has now been signed into law...On its surface, there seems to be little controversy in supporting more funding to the National Institutes of Health for innovative research, more expeditious approval of promising drugs by the Federal Drug Administration, and better approaches to combat the opioid abuse epidemic...However, there are a number of troubling provisions in this potential law of which the public should be aware...To outline the advantages and concerns, I created a series of winners and losers from which readers can draw their own conclusions:
- Winners
Drug companies: It shouldn’t surprise anyone that the pharmaceutical industry is ecstatic over the law, as the bar for approval of drugs under development is made significantly lower by reducing the rigor required for approval of new drugs by the FDA. Traditionally, rigorous double-blinded peer review studies are required prior to consideration and approval. - Losers
The FDA: Already stymied with deteriorating labs, under staffing and somewhat compromised approval processes for new drugs and medical devices, the FDA is not expected to fare better under the new law...Lowering the bar for the rapid and expeditious approval of pharmaceutical and medical device products with expansion of off label uses will only further undermine the FDA’s ability to protect the public from potential harm...Evidence-based medicine: The law allows for the use of “real-world evidence”—which may include anecdotal studies or observations, patient or professional opinion, as well as non-validated or scientifically measured outcomes—to justify the approval of a new treatment product...pharmaceutical companies will only be required to submit a “data summary” for new pharmacological agents or new indications to existing products, which will make the introduction of new products far easier.
- Winners