- FDA issues guidelines for female libido pills after learning some hard lessons (statnews.com)
After a two-year wait, the US Food and Drug Administration finally issued new guidance for companies that want to develop drugs to bolster female libidos. But the details suggest the agency has belatedly learned some hard-fought lessons following complaints that the controversial Addyi pill did not warrant approval last year...The 15-page draft guidance...offers a typical how-to for companies, but also points to certain steps that Sprout Pharmaceuticals did not follow as part of its Addyi marketing application...The drug, which is now sold by Valeant Pharmaceuticals, was approved despite debate over its safety and effectiveness, and the extent to which medicines should be used to treat female sexual dysfunction...the FDA is locking the barn door after the horse got out. So now, the agency is telling other companies to do some things that Sprout didn’t do...they’ve made it harder to get a drug approved but they have extended helping hands in numerous places...unless [a company] really looks hard for subgroups [of patients], the whole effort is hopeless...
- Treat pharma as a utility, and 10 other ways states can lower drug prices (statnews.com)
...a group of state health policy makers is offering some novel — and also some familiar — suggestions...These include regulating the pharmaceutical industry as a utility, allowing states to operate as pharmacy benefit managers and waiving some provisions of the Medicaid program...the National Academy for State Health Policy also recommends that states pursue laws that require more transparency from drug makers. And the organization floated some timeworn proposals, such as importing medicines from Canada and prosecuting drug makers for violating consumer protection laws that protect against predatory pricing monopolies..."The proposals in this paper require more dialogue, debate, development, and experimentation. These policy proposals may not be appropriate for all states or agencies, nor for every pharmaceutical product," the working group wrote. "But states need to act and this paper presents a toolbox of options to consider."... the Pharmaceutical Research & Manufacturers of America, the industry trade group. A spokeswoman wrote us to say that "many of the solutions proposed by the Work Group would harm patients and limit their access to life-saving treatments. We hope to begin a dialogue with the National Academy for State Health Policy on solutions that will help states better manage and predict costs while preserving patient access...
- Huge Valeant price hike on lead poisoning drug sparks anger (statnews.com)
A drug used to treat lead poisoning is causing a toxic reaction among hospitals and poison control centers after Valeant Pharmaceuticals jacked up the price more than 2,700 percent in a single year...At issue is a decades-old, intravenous treatment for severe and life-threatening cases of lead poisoning, which occur infrequently, but generally require supplies to be on hand...Calcium EDTA, Valeant acquired the drug in 2013…By December 2014...increases took the price to $26,927 (from $950)...Ever since, poison control specialists have been angry, especially since there are few viable options...Their reaction is a case study in the exasperation doctors and hospitals feel at the multitude of price hikes of all sizes that they see month after month...create frustration and squeeze budgets behind the scenes...There’s no justification for the astronomical price increases...which limit availability of the drug to children with life-threatening lead poisoning...
- Manufacturer of cosmetic and pharmaceutical products expands into Dayton (nnbw.com)
Cosmetic Enterprises, Ltd., a growing manufacturer of cosmetic and pharmaceutical products, is planning to expand its operations into Dayton...CEL, headquartered in Pacoima, Calif., purchased a vacant 60,000-square-foot building along with an adjacent 18 acres for potential development..."During our due diligence we reviewed many different regions and states and found Nevada to be manufacturing friendly that is centrally located to our existing customer base…Cosmetic Enterprises is a great addition to Lyon County...Not only are they creating the kind of higher paying jobs that help boost the region, they will be great corporate citizens...CEL produces cosmetic and skin care products, such as shampoo, moisturizers, skin enhancing lotion and skin protecting products, using botanicals and other raw materials...
- Inside big pharma’s fight to block recreational marijuana (theguardian.com)
Pharma and alcohol companies have been quietly bankrolling the opposition to legal marijuana, raising questions about threats to market share...Marijuana legalization will unleash misery on Arizona, according to a wave of television ads that started rolling out across the state last month...the surprise lies in who is backing them. In August, the pharmaceutical company Insys Therapeutics also cited concerns for child safety when, with a $500,000 contribution, it became the largest donor to Arizona’s anti-legalization drive. But their stated concerns have raised a few eyebrows across the state. Insys manufactures Subsys, a prescription painkiller derived from fentanyl…critics say, the Insys contribution in Arizona is a ploy to protect market share. And it mirrors other large donations to anti-marijuana campaigns by pharmaceutical and alcohol companies that fear the growing clout of legal marijuana...Research conducted...shows that medical cannabis patients are substituting cannabis for pharmaceuticals at a very high rate, and for alcohol at a pretty high rate as well...Opiate overdoses dropped by roughly 25% in states that have legalized medical marijuana compared to states that have prohibited sales of the plant, according to a 2014 study from the Journal of the American Medical Association. The study implies that people could be using medical marijuana to treat their pain rather than opioid painkillers, or they’re taking lower doses...
- Those risk disclosures on drug websites? People don’t read them, study says (fiercepharma.com)
Consumers don’t always read the risk information on branded drug websites--even though they say they do, according to new eye-tracking research from the University of Tennessee. Risk disclosures are a hot-button issue in pharma marketing, with some critics accusing brands of downplaying the risks and marketers contending that they follow the rules laid out by the FDA...The key finding? Even though 80% of the participants said they read half or more of the website information, they actually read much less than that and had limited recall of the drug’s risks. Further, the study found consumers focused on the drug’s benefit and generally ignored the risks... Mariea Hoy, who fielded the research...presented the findings to the FTC and has shared the same information with the FDA, also offered some suggestions on how to get people to read more drug risk information, Pharma companies should try to identify why people aren’t reading the risks, she said. Are they trying to avoid negative information? Do they think they already know the risks? Do they discount the risks for themselves, with the it-won’t-happen-to-me mindset known as "optimum bias?"...
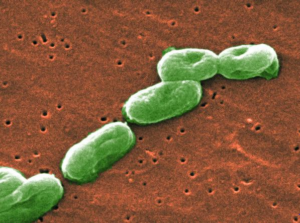
- Feds blame multistate B. cepacia outbreak on PharmaTech plant’s water system (fiercepharma.com)
After a months-long investigation, federal officials have nailed down the source of a Burkholderia cepacia outbreak that made its way into several states and infected dozens of patients...the Centers for Disease Control and Prevention and the FDA said they had detected the B. cepacia bacteria in the water system at Florida-based CMO PharmaTech. The company had produced 10 contaminated lots of constipation drug docusate sodium before 6 distributors shipped the meds around the country. In total, 60 people in 8 states became infected...The outbreak started in late June in ventilated cystic fibrosis patients. During the investigation, CDC officials said infections could be life-threatening in patients with compromised immune systems or lung conditions.
- Chicago’s proposal to license pharma sales reps is “bad morality, bad economics,” critic says (bizjournals.com)
Pharmaceutical sales representatives working in Chicago might soon have to carry a special license to operate within the city limits...city officials have proposed an ordinance that would require pharmaceutical reps to record and report to the city the number of health care professionals they've contacted, the types of drugs promoted, any samples provided, and if doctors were paid for their time...The ordinance might also require sales reps reps to keep a list of names of doctors they've contacted and potentially give those names to the city if requested. The new licensing requirements are part of a broader effort to fight prescription opioid abuse…The proposed pharmaceutical sales rep license would likely carry a $750 annual fee, the Tribune reported. In addition to the new reporting requirements, sales reps also would have to take city-certified training classes on prescription drug abuse, ethics and marketing standards...The...ordinance...would generate another stream in the flow of occupational licensing fees that go into city coffers...The city is looking for money, if I had to guess…
- Pfizer’s Lyrica patent appeal fails in U.K., endangering bid to protect $5B med (fiercepharma.com)
Pfizer’s divide-and-conquer approach to Lyrica in the U.K. just hit a wall. The Court of Appeal upheld a ruling that struck down key patent claims on Lyrica and cleared Actavis’ generic of infringing it...The...case centered on a “carve-out” approval for Actavis’ Lyrica generic, a type of regulatory nod that branded drugmakers see as a threat...drugmakers want to prolong their monopoly access to patients, and they use follow-up patents to extend their protection past the time when IP coverage expires on the original compounds...some recent “skinny” regulatory nods--from the FDA as well as international regulatory agencies--that clear generics only for particular indications have complicated those efforts, because they give generics makers an entreé onto the market while so-called method-of-use patents remain in effect...The patent at issue in this case covered Lyrica’s use as a pain treatment; the patent on pregabalin itself, the active ingredient...had already expired. These days, Lyrica is used more often for pain than for its original indication as a seizure drug...The company hopes to now take its fight to the U.K. Supreme Court...Pfizer maintains its strong belief in the validity and importance...of the patent…
- Rising drug prices are making hospitals feel ill (statnews.com)
While much of the attention over prescription drug prices is focused on consumers, a survey released on Tuesday finds that hospitals are also spending much more than in the past. Between 2013 and 2015, the average annual drug spending for patients who stay in community hospitals increased by of 23.4 percent...And on a per admission basis, hospital spending on drugs jumped nearly 39 percent, to $990...the increase in prices outpaced reimbursement rates from payers, retail spending on medicines, and the pharmaceutical price inflation...the survey found that more than 90 percent of the hospitals surveyed reported that recent price hikes for inpatient drugs had a moderate or severe effect on managing costs...The furor has put the pharmaceutical industry on the defensive as many drug makers are scrambling to justify their price hikes. Some drug makers are pointing fingers at pharmacy benefits managers, and the convoluted pricing system in which rebates that are paid to win coveted placement on lists of drugs for which insurance coverage is provided...The drugs that are increasing in price generally are not new and are off patent. There is no competition, so hospitals are forced to pay in order to make the best patient care decisions...These price increases are unsustainable for hospitals who are taking care of sicker and sicker patients…