- MD Labs is part of a new industry called pharmacogenetics in Reno (nnbw.com)
Not everyone reacts to the same medication in the same way...Benedryl makes some people drowsy and others wired. Antidepressants have no affect on 38 percent of patients...Some people are more susceptible to addiction from pain medications while others get pain relief without getting hooked...MD Labs is part of a new industry called pharmacogenetics that uses genetics to map specific genes involved in the metabolism of and response to specific drugs...Ruttledge and Denis Grizelj, co-founder and CEO, began MD Labs in 2011 as a toxicology testing facility for physicians nationwide. They expanded to pharmacogenetic testing in 2014, with the development of their proprietary genetic test Rxight, which maps genes that affect more than 200 medications...Because it’s genetically based, Rxight is a once-in-a-lifetime test...Preemptive testing makes the patient’s genetically-based profile of drug reactions available to doctors and pharmacists before illness strikes and before a drug is prescribed...MD Labs contracts with Saint Mary’s Health to offer the testing and consultation at both Saint Mary’s Regional Medical Center and Saint Mary’s Medical Group Primary Care Northwest Reno...Pharmacists at those locations are Rxight Certified to administer the test and provide consultation...Nationwide the company has 100-110 employees total, which includes about 55 people in the Reno office plus a sales force in offices in Chicago and Pennsylvania...MD Labs owners see a future in which pharmacogenetics testing is routine.
- Humana, UnitedHealthcare join new value-based prescription drug model (modernhealthcare.com)
Humana, UnitedHealthcare, WellCare, Blue Cross and Blue Shield of Florida, Blue Cross and Blue Shield Northern Plains Alliance and CVS Health are among those participating in the Part D model that gives insurers financial incentives when they offer innovative programs that encourage seniors to take their medications...The experiment comes out of the Affordable Care Act…Evidence suggests that medication therapy services currently offered by Part D plans don't improve quality or reduce unnecessary medical expenditures. That's likely due to misaligned financial incentives and regulatory constraints…The focused approach is critical Medicare Advantage plans currently have more of an incentive than stand-alone Part D plans to work with patients to take their medications because they are on the hook for hospitalizations…The Medicare Advantage value-based insurance design model will begin Jan. 1, 2017, and run for five years…
- SEC charges former Puma biotech exec with $1.1 million in insider trading (statnews.com)
In the latest instance of alleged insider trading in the pharmaceutical industry, a former Puma Biotechnology executive was charged with illegally making more than $1.1 million by taking advantage of confidential information about clinical trials for a cancer drug...Robert Gadimian, who was senior director of regulatory affairs, bought and sold Puma stock after learning about favorable study results for a medicine that was being tested to treat breast cancer…This alleged episode of insider trading is only the latest instance involving the pharmaceutical industry or those working with drug makers. The issue has increasingly raised concerns in connection with clinical trial work, as well as deal-making and the drug approval process, which some fear can be distorted by such activities.
- Many ex-FDA drug reviewers take ‘revolving door’ to pharma industry (medcitynews.com)Future jobs of FDA’s haematology-oncology reviewers (bmj.com)
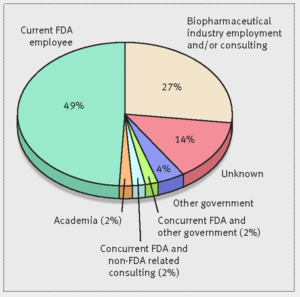
More than a quarter of the Food and Drug Administration employees who approved cancer and hematology drugs from 2001 through 2010 left the agency and now work or consult for pharmaceutical companies…Dr. Vinay Prasad, a hematologist-oncologist and assistant professor at Oregon Health and Science University, sought to understand the so-called "revolving door" between the FDA and the pharmaceutical industry, which he said is often discussed but hadn’t been quantified...Going to work for industry after leaving the FDA is not inherently bad, but it does raise some questions...If you know in the back of your mind that your career goal may be to someday work on the other side of the table, I wonder whether that changes the way you regulate...Are you more likely to give [companies] the benefit of the doubt? Are you less likely to beat them up hard over [using bad comparisons in drug studies]?...
- To stop FDA inspector, workers at Japanese drug maker formed a human barricade (statnews.com)
Someone at Nippon Fine Chemical must have been very nervous when an investigator from the US Food and Drug Administration arrived last year...In an unusual display of chutzpah, the drug and ingredients maker refused to allow the investigator to inspect its quality control laboratory at its facility in Hyogo, Japan. Employees literally formed a "shoulder-to-shoulder" barricade to prevent the FDA employee from entering…the FDA investigator reviewed customer complaints that Nippon drugs contained glass, hair, cardboard, metal — and even a black spider. But Nippon employees refused to provide the investigator with copies of documents that detailed customer complaints. The FDA letter noted that Nippon uses the same equipment and processes for drugs sold in the Japanese and US markets...As a result of these foibles, the FDA declared Nippon products to be adulterated and issued an import alert two months ago, an action that means Nippon products can be detained from entering the US...Nippon could not be reached for comment...
- Pharma industry should take a page from airline social media mavens, consultant says (fiercepharma.com)
Airlines are known for their quick and effective social media responses, often helping stranded and annoyed passengers in real time. But while pharma companies can’t solve problems instantly with a reroute or seat change, they can definitely look to airline’s social media savvy for inspiration, one industry consultant says...Pharma companies need to get over the mindset that social media is too difficult to do in the regulated industry…You have a social media manager who is actively monitoring public forums for complaints and seeking to address those complaints as quickly as possible...actionable intelligence...or...acting on the data to provide a benefit of some kind...becoming aware of discussions on social media…a "treasure trove of valuable patient insights"--and analyzing what percentage could be acted upon to the mutual benefit of the brand and the patient...
- Buyers remorse or fraud? Teva and Mexican brothers slug it out over sour deal (statnews.com)
Drug makers are constantly foraging for deals to bulk up their pipelines, but not all go according to plan...Consider the nasty spat between Teva Pharmaceuticals and two Mexican brothers, Fernando and Leopoldo Espinosa...they have filed dueling lawsuits after their $2.3 billion deal went sour…At issue was a move made last year by Teva...to expand in Mexico by purchasing Rimsa, one of the country’s largest independent pharmaceutical manufacturers, from the Espinosa brothers. At the time, Teva hailed its acquisition...as a "significant platform for growth" in the second-largest market in Latin America...Teva claims the brothers engaged in chicanery, ploys, and misrepresentations about their business in order to walk away with huge profits, according to its lawsuit...Rimsa was "was engaged in a years-long scheme to sell defective and unlawful products and to conceal those violations from Mexican regulators...Teva lawsuit goes on to claim that Rimsa submitted fraudulent information to regulators about ingredients suppliers and lied about laboratory tests, including the stability tests that must ensure the stated shelf life of each product was accurate and that the product would remain stable, and, therefore, safe and effective…
- Mylan to pay $465 million to settle claims of shortchanging Medicaid (statnews.com)
Mylan reached a $465 million settlement on Friday with the US Department of Justice and other government agencies over accusations that it shortchanged Medicaid...The move comes after a month of controversy over charges that Mylan improperly classified its EpiPen allergy device in reports to the Medicaid Drug Rebate Program. Under this program, companies must accurately report — and pay — a rebate on drugs paid for by the agency...Mylan, however, had been reporting EpiPen as a generic product for nearly a decade. And this is an important distinction, because classifications are used to determine the size of rebates that companies pay Medicaid. Rebates, which companies pay in exchange for having their products covered, are lower for generics — 13 percent versus 23 percent for a brand-name product.
- The Patent Truth: The Access to Medicines Debate (realclearhealth.com)
Bill Gates and Abraham Lincoln are in agreement. Gates, whose foundation funds the development and distribution of new medicines in the developing world, said the U.S. system is better than most other systems one can imagine…. The drug companies are turning out miracles, and we need their R&D budgets to stay strong. They need to see the opportunity." It turns out that Mr. Gates is in good company. Lincoln, the only U.S. President to hold a patent, called the IP system, "the fuel of interest which fires man’s genius."...At the United Nation’s General Assembly...a group called the High Level Panel on Access to Medicines was working hard to extinguish that source of miracles. The UN’s HLP issued a report with recommendations that would disrupt medical innovation and potentially lower the health status of impoverished communities throughout the world. The report’s fundamental premise is that millions of people in low and middle-income countries have been denied access to medicines because patents have increased their prices...The UN’s HLP ignores...progress, and the evidence that innovation and its intellectual property laws can exist alongside the delivery of medicines and health services to people in need...Governments need to be aware of attempts by UN agencies to put the HLP recommendations into programs....Replacing the current system’s proven accomplishments with an untested system would endanger the astounding improvement in health conditions of the poor over the past half-century. Over time, it might turn a miracle into a tragedy...
- European regulators appeal rulings that prevent release of drug data (statnews.com)
Once again, drug makers and European regulators are clashing over disclosing information...the European Medicines Agency is appealing two different rulings by the General Court of the European Union that prevent it from releasing data to third parties…
- ... first order blocked release of a case study report for Translarna...which is sold by PTC Therapeutics...Until PTC objected, the EMA planned to provide access to the report in response to a request, albeit with redactions that agency officials maintain are in accordance with their own regulations…
- ...second order blocked the release of three toxicity studies for Bravecto...sold by Merck’s animal health unit, which also objected to disclosure.
Our approach to transparency has been welcomed by many of our stakeholders, and these court cases are a good opportunity to test our rules on making available to the general public the documents on which EMA’s scientific opinions on medicines are based...The episode was closely watched because it arose as the pharmaceutical industry faced growing disclosure pressure following scandals over safety or effectiveness data that were not publicly shared. Drug makers have argued that disclosing certain data may compromise trade secrets or patient privacy. Consumer groups counter such information is kept out of reach at the expense of patients...