- Ex-Insys sales manager arrested in U.S. fentanyl-kickback case (reuters.com)
A former Insys Therapeutics Inc district sales manager was arrested...on charges he participated in a scheme to pay kickbacks to doctors to prescribe a drug containing the opioid fentanyl...Jeffrey Pearlman...was charged in a criminal complaint filed in federal court in New Haven, Connecticut, becoming the latest individual to face prosecution in connection with probes involving Insys' drug Subsys…The charges come as Insys faces a number of state and federal investigations involving Subsys as U.S. authorities seek to combat a national epidemic of opioid abuse...Prosecutors said Pearlman and sales representatives he managed induced doctors, advanced practice registered nurses and physicians' assistants to prescribe Insys' fentanyl spray by paying them to participate in sham "speaker programs."...As a result of the scheme, federal healthcare programs incurred millions of dollars in losses, prosecutors said...
- Drug Distributors Still Benefiting from Drug Price Inflation but Face Major Profit Challenges, Says New Drug Channels Institute Study (drugchannelsinstitute.com)
Drug Channels Institute...released its...in-depth analysis of the highly dynamic prescription drug distribution industry...2016–17 Economic Report on Pharmaceutical Wholesalers and Specialty Distributors reveals how list price increases of brand-name drugs are boosting the industry’s revenues, even as changes in the U.S. healthcare system threaten wholesalers’ profits...We project that U.S. drug distribution revenues at the Big Three public wholesalers—AmerisourceBergen, Cardinal Health, and McKesson—will exceed $400 billion in 2016...But any change in drug price inflation rates or in manufacturers’ rebate strategy will be very negative for wholesalers’ future revenues and profits...Consolidation among pharmacies and hospitals is making it difficult for wholesalers to capture value and profits from the booming specialty drug market...And contrary to many people’s expectations, specialty biosimilar drugs seem poised to deliver only minimal incremental profits for wholesalers...
- Can You Guess Which Big Pharma Has Raised Its Drug Prices 5 Times Since June 2014? (fool.com)
This is probably far from breaking news -- or in any way shocking -- but prescription drug prices are soaring...According to an analysis conducted by healthcare data company Truveris...prescription drug inflation topped 10% in 2015, spearheaded by an average price increase in branded pharmaceuticals of 14.77%...But what if I told you that a well-known Big Pharma has been regularly hiking drug prices in plain view and no one seems to be taking notice? In fact, this drug giant has increased its list prices...five times since June 2014. This company's average list price increases are as follows:
- June 2014: an average increase of 7.4%
- January 2015: an average increase of 8.8%
- June 2015: an average increase of 8.5%
- January 2016: an average increase of 10.4%
- June 2016: an average increase of 8.8%
...this works out to a cumulative increase over a two-year span of approximately 52% in list prices...Can you name this pharma giant?
- Merck’s Keytruda to make its China debut as a resort-island draw for medical tourists (fiercepharma.com)
Merck & Co.’s cancer treatment Keytruda could become a national tourist attraction in China, with a special approval for imports to the resort island of Hainan...Immuno-oncology stalwart Keytruda will be available at the Boao Lecheng International Medical Tourism Pilot Zone, designed to attract travelers for new treatments...Specifics on pricing and approved indications weren’t covered in the announcement, but it did say officials are considering other drugs for the program...The medical-tourism nod comes as BMS continues to navigate the tough approval process in China with its growing blockbuster med Opdivo--and Keytruda itself awaits regulatory approval for the rest of the country...the Hainan program will open up new treatments--including Keytruda--to affluent Chinese residents who can afford the travel and medical costs, while other patients will have to wait for regulators to approve them.
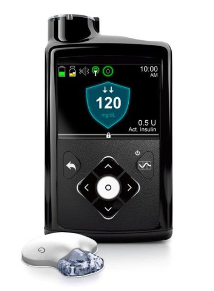
- FDA approves 1st ‘artificial pancreas’ for type 1 diabetes (upi.com)
The Food and Drug Administration...approved the first automated insulin delivery system -- a so-called "artificial pancreas" -- for people with type 1 diabetes...This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin…The device -- Medtronic's MiniMed 670G -- is what's known as a hybrid closed-loop system. That means it monitors blood sugar and then delivers necessary background insulin doses. The device will also shut off when blood sugar levels drop too low...this device isn't yet a fully automated artificial pancreas. People with type 1 diabetes will still need to figure out how many carbohydrates are in their food, and enter that information into the system, the agency noted..
- Indian union plots mass protest against ‘unfair’ treatment of pharma sales reps (fiercepharma.com)
A strike by Sun Pharma employees this week is just one symptom of a campaign by a prominent trade union to seek more equitable working conditions for pharma sales representatives...The Federation of Medical and Sales Representatives' Associations of India claims many reps working in India do so with contracts that contravene their working rights--as well as Indian employment law. It is trying to organize a mass demonstration on the streets of the capital, New Delhi, on November 21, and is threatening an all-out national strike if its demands are not met...around 500 Sun Pharma sales reps engaged in a one-day strike to protest what they call unfair treatment of workers who joined the company from Ranbaxy Laboratories, including unpaid wages and expenses, and demotions to a lower working grade…FMRAI alleges that unfair labor practices are rife in the Indian pharma market. Sales reps fear losing their jobs if they become ill--or simply at the whim of their managers...The union wants India's government to set statutory working rules for sales employees and to strictly enforce the existing Sales Promotion Employees Act. It's also calling for stiffer penalties--including the threat of jail--for employers found to have victimized staff...
- Senator urges FTC to step up investigation into maker of addiction treatment (statnews.com)
A US senator is urging the US Federal Trade Commission to accelerate its antitrust investigation into a drug maker that sells Suboxone (buprenorphine/naloxone), a medicine used to treat addiction to opioids and narcotics...The appeal by Senator Edward Markey comes less than a week after 35 states filed an antitrust lawsuit against Indivior (Reckitt Benckiser), accusing the company of engaging in a scheme to block generic competition. In doing so, the company "drove up prices and deprived states and consumers of lower costs," Markey wrote in his letter on Tuesday to FTC…Reckitt raised the price of its pill while lowering the price of the newer film version. And the patent on the new version does not expire until 2023. This is a pharmaceutical industry tactic known as forced switching, since patients and physicians have little choice but to consider a newer, higher-priced version...Markey also pointed out that the manufacturer has "significantly impeded the FTC investigation by attempting to deny the FTC access to thousands of pages of documents that are integral to the investigation." However, he also noted that the federal court overseeing the matter later ordered the company to produce the documents...So will the FTC step on the gas?
- Mylan CEO Bresch used a sky-high tax rate to lowball EpiPen profits by 66% (fiercepharma.com)
Mylan CEO Heather Bresch quoted one number that got a lot of attention last week on Capitol Hill: EpiPen profits weren’t all that large, just $100 for each package of two pens, on a list price of more than $600...Thing is, that number was misleading...In a securities filing...Mylan posted a chart detailing the EpiPen numbers. The profits figure Bresch gave during the congressional committee hearing included a 37.5% tax rate--and that understated the product’s profits by $187 million...Before taxes, EpiPen profits came in at $498 million--or $166 per package. Almost two-thirds higher than Bresch had said...And not only did Bresch fail to specify that she’d deducted taxes from that profits figure, but that tax rate was 5 times higher than what Mylan paid last year, thanks to a tax inversion deal that moved its domicile from the U.S. to the Netherlands...For 2016, Mylan has forecast larger profits and a bigger operating margin for EpiPen. According to the chart Mylan filed, It’s looking for $671 million in operating profits, up from last year’s $498 million. And it expects operating margins to be 75% for the product, compared with 72% in 2015...
- What’s next for Valeant’s Sprout biz? A huge impairment charge, analyst predicts (fiercepharma.com)
Poor sales trajectory, check. Scale-back in marketing effort, check. What’s next for Valeant’s Addyi and its maker, Sprout? An impairment charge...and a big one, at that...David Maris expects to see a whopper charge on the carrying value of Sprout--as in, as much as 90% or more...And he expects to see it "with next quarter’s results, if not sooner...Valeant has already warned in a regulatory filing of a potential impairment...sales of Addyi (flibanserin) --the controversial female libido drug that Valeant shelled out $1 billion to gain access to--are dismal. In July, they had reached just $696,235, Maris wrote, citing IMS data--a far cry from the $100 million to $150 million the Canadian drugmaker forecast for this year...when the "overdue" impairment does finally come, investors will feel the burn...the write-off cut into 2017 EPS and beyond by 14 cents per share, though that’s not necessarily even the worst part…
- Diabetes drugs are badly needed, but rarely make it to market (statnews.com)
Diabetes may be a widespread disease for which millions of people need treatment, but a new analysis finds that developing new medicines has been a risky proposition for drug developers...How so? Here are a few key findings:
- Only 1 in 13 investigational diabetes drugs that entered clinical testing from 1995 to 2007 ultimately received regulatory approval, compared with 1 in 8 for all investigational drugs…
- The likelihood that a diabetes drug entering clinical testing would make it to late-stage testing was about 13 percent...
- the most important challenge for drug makers...continues to be the regulatory approval process, which has grown more demanding in the wake of a controversy in 2008 over the heart risks of a widely used diabetes drug.
- so-called first-in-class approvals for diabetes — which refers to new types of medicines — represented almost 30 percent of all FDA approvals.
- of all new diabetes drugs that were approved by the FDA from 1995 to 2015, 15 percent received a so-called priority review designation...
- The 61 diabetes and other endocrine drugs approved from 1995 to 2015 accounted for 10 percent of all new medicines approved by the FDA during that time...