- Medicaid’s warning on government healthcare (washingtonexaminer.com)
One reason why Medicaid isn't so good is that many doctors won't actually accept Medicaid patients...If you rely upon the health insurance system for the poor, you'll likely not be able to get medical treatment in many places. Seeing as medical coverage is designed to provide access to treatment when needed, the system doesn't seem to work all that well. This is not a good thing. After all, a bit of bad luck, and one of us could be relying on Medicaid...READ MORE
- After breakout year, Moderna on track to generate $15B+ in 2022 thanks to more demand, higher prices: analysts (fiercepharma.com)
Moderna is hard at work ramping up production of its mRNA COVID-19 vaccine, which is projected to reap over $19 billion for the company by year’s end. But given that the pandemic is easing in parts of the world, what’s less certain is how 2022 will play out…After a breakout year, Moderna sees strong reason to believe 2021 won't be a one-off boom year. That's thanks to the predicted need for booster shots and additional supply deals coupled with stronger pricing power…READ MORE
- Eli Lilly hit with DOJ subpoena over New Jersey factory making COVID-19 drug (fiercepharma.com)
...Eli Lilly’s COVID-19 drug factory in New Jersey garnered unwanted headlines for quality control snafus and alleged document tampering. Now, the Department of Justice has stepped in to investigate...Lilly revealed the DOJ has issued a subpoena demanding documents relating to the site in Branchburg, New Jersey. The site produces doses of Lilly's COVID-19 antibody treatment, which has recently faced setbacks for its susceptibility to certain variants...In early May, Reuters reported that employees at the site accused an executive of altering FDA-required documents in an effort to downplay serious quality control problems...READ MORE
- Needles in haystacks: a new generation of AI-enhanced drug discovery companies (pharmaceutical-technology.com)
The search for novel therapies has long been a trial-and-error process that costs drug companies a vast amount of time and money. Now, with artificial intelligence (AI) set to transform the pharmaceutical industry more than any other emerging technology, a growing number of pharma and biotech groups are harnessing the cutting-edge tech to minimise the hit-and-miss nature of R&D and discover new therapies with previously impossible speed and accuracy...READ MORE
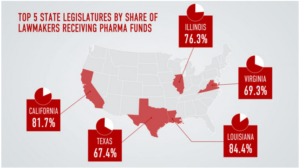
- Pharma funded more than 2,400 state lawmaker campaigns in 2020, new STAT analysis finds (statnews.com)
State lawmakers in Oregon have tried to lower high drug prices from nearly every angle: They’ve sought to cap how much people can pay for insulin, install a panel that could determine how much state agencies should pay for medicines, and even import drugs in bulk from Canada. Nearly every proposal has failed...One reason, at least according to the effort’s supporters: Two-thirds of the state legislature accepted at least one campaign check from the drug industry during the 2020 election cycle...READ MORE
- U.S. Supreme Court rebuffs J&J appeal over $2 billion baby powder judgment (reuters.com)
The U.S. Supreme Court...declined to hear Johnson & Johnson’s bid to overturn a $2.12 billion damages award to women who blamed their ovarian cancer on asbestos in the company’s baby powder and other talc products ...The justices turned away a J&J appeal and left in place a Missouri state court ruling in litigation brought by 22 women whose claims were heard together in one trial...READ MORE
- UNLV students can earn MD and MBA in 5 years (reviewjournal.com)
UNLV will launch a program this fall that allows students to earn both a medical doctor and master of business administration degree in five years...The university will join about half of the nation’s MD-granting medical schools — including University of Nevada, Reno — that already offer a similar dual degree program...For leadership roles in medicine — particularly, in hospital leadership — a master of business administration (MBA) or equivalent degree is often recommended or required...READ MORE
- White House says data is one key to improving pharma supply chain resilience (healthcareitnews.com)
Other recommendations include boosting local production, promoting research and development, and creating robust quality management maturity...The White House and several federal agencies...released a series of policy recommendations for addressing the vulnerabilities in U.S. pharmaceutical supply chains...The review came in response to an executive order signed by President Joe Biden...that directed the government to identify risks, address vulnerabilities and develop a strategy to promote supply chain resilience throughout sectors...READ MORE
- ICER, vocal critic of drug company pricing, turns scrutiny to insurers (biopharmadive.com)
The Institute for Clinical and Economic Review, a nonprofit and frequent critic of drugmakers for excessively high drug prices, now plans to assess how health insurance policies harm patient access to care...This follows research by the group that argued cost-sharing should not be structured to shift healthcare costs to patients when they have no medically appropriate lower-cost option...ICER's review will not, however, look at whether cost-sharing should be reduced for drugs on which payers receive large rebates or whether payers should be asked to demonstrate how their policies on drug access save overall healthcare costs...READ MORE
- 5 Frequently Asked Questions about Drug Diversion Management (drugtopics.com)
In reality, nearly half (44%) of hospital pharmacists experienced a diversion event within the past 12 months, which is up from 37% in 2019. The COVID-19 pandemic has exacerbated this “hidden epidemic,” a multi-faceted challenge which poses serious risks to patients, clinicians and healthcare systems...Here are 5 questions frequently asked by health systems seeking to accelerate and improve their efforts to detect and mitigate drug diversion incidents...READ MORE
- What are the biggest challenges in identifying drug diversion?
- How have responsibilities for addressing and resolving drug diversion issues evolved?
- What offers the biggest opportunity to improve diversion identification?
- When selecting automated systems and software, which capabilities are most critical?
- How do you justify the cost of technology in a difficult economic environment?