- CDER Publishes Drug Safety Report – FDA’s Center for Drug Evaluation and Research has published its second annual report on key safety programs and activities. (biopharminternational.com)
...FDA’s Center for Drug Evaluation and Research released its second annual Drug Safety Priorities report, which details drug safety initiatives carried out by CDER and FDA. The report highlights drug safety program milestones and gives an update on goals achieved in 2017. Efforts by FDA to ensure drug safety science, surveillance, and oversight are discussed...Detailed in the report are the agency’s efforts on pharmacovigilance, medication errors, and risk management...The report also goes into detail about the agency’s views on how real-world evidence can advance drug safety. An update on the agency’s efforts to combat the opioid crises is also provided...
- Pharmacy Week in Review: April 27, 2018 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharmacy Week in Review: April 20, 2018 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Amazon finds it’s not so easy to sell drugs—and nixes one plan entirely: CNBC (fiercepharma.com)
Amazon is running into challenges in its drug distribution push...Amazon's potential leap into pharmaceuticals has weighed on drug distributors and pharmacies for months, but those companies are getting relief from news that the online retail giant has put at least one of its plans on ice...a unit that sells bulk products to companies—hasn't been able to convince hospitals to get on board with its plan. One reason: The company doesn't have a proper cold-chain logistics network, which is crucial to distributing many drugs...Amazon has now backed away from the effort, the publication reported...The company is pressing ahead with other healthcare projects, though it's unknown whether they involve pharmaceutical sales...
- Illicit drug use should not be a crime, says Royal College of Physicians (bmj.com)
The Royal College of Physicians of London has joined calls for an end to criminal sanctions against people who take drugs such as heroin, cocaine, and cannabis for non-medical reasons...The college now endorses the stance of the Faculty of Public Health and the Royal Society for Public Health. In a 2016 report the RSPH concluded that the “war on drugs” fails to deter drug misuse but instead deters people with drug use disorders from seeking treatment and inhibits harm reduction efforts...Jane Dacre, president of the Royal College of Physicians, told The BMJ, “The criminal justice system is not the place to address the often complex needs of people addicted to drugs. (subscription required)
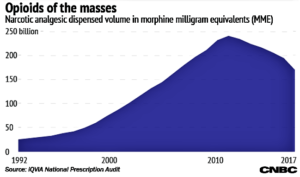
- How doctors in the US prescribe opioids — four charts (cnbc.com)Institute Reports Medicine Use and Spending in the US Review of 2017 Outlook to 2022 Medicine Use and Spending in the U.S. A Review of 2017 and Outlook to 2022 (iqvia.com)
The number of opioid pills prescribed peaked in 2011 and has since declined by 29 percent, according to a new report from the Iqvia Institute...The number of prescriptions accelerated its decline in 2017, with an even faster drop for high doses...More patients appear to be starting on medication-assisted therapies to treat opioid addiction...decline accelerated...helped by changes in regulation of opioid prescribing and in reimbursement policies from insurers...many of the programs that have been put into place in the past year or two seem to be having an impact...increased use of nonopioid pain treatments, like ibuprofen and other nonsteroidal anti-inflammatory drugs...as well as public awareness about overuse and misuse of opioids...
- FDA commissioner to drug middlemen: You’re part of the problem (cnbc.com)
Drugmakers may not be the only ones keeping less-expensive drugs off the market...The Food and Drug Administration has approved nine biosimilars, generic versions of biologic medicines, but only three are available...Manufacturers are using several schemes to "hamstring biosimilar competition," FDA Commissioner Scott Gottlieb said... he worries pharmacy benefit managers have been "complacent participants" in the schemes...PBMs and insurers may stick with branded biologics because they receive discounts from manufacturers on these treatments. That can leave consumers paying for costly treatments when less-expensive ones are available while PBMs make more money on these discounts, known as rebates...Many of these practices persist because high list prices enable lucrative returns across the drug supply chain as the spread between list and net price is carved up and shared among participants...he (Gottlieb) applauded insurers who have recently pledged to pass manufacturer's drug rebates directly on to some of their members...This is a bold action that will help create a fairer, more transparent market...I hope that other insurers, employers, and manufacturers follow their lead. I also hope that your industry will continue to innovate to make it more transparent to pass along these rebates...
- In a blow for pharma, Supreme Court upholds the hated IPR patent challenge (fiercepharma.com)
Branded drugmakers have said "no fair" to the U.S. Patent and Trademark Office's inter partes review system almost since its inception seven years ago. One reason? "It's unconstitutional," the companies contend...The Supreme Court...disagreed...In a 7-2 vote, the U.S. High Court held that the system is constitutional. It doesn't violate Article III of the Constitution, which gives the judicial branch alone the power to decide legal arguments. Nor does it violate the Seventh Amendment, the right to a trial by jury...It's a limited ruling specific to the issues in this particular case, Oil States Energy Services v. Greene’s Energy Group...Justices John Roberts and Neil Gorsuch dissented, saying only an independent judge—not a political appointee at the PTO—should be able to revoke patents, which they equated with personal property such as a home or a farm...PhRMA...said the "narrowly tailored decision" found only that IPRs are constitutional, not "efficient or fair." The arguments and a...ruling in another case—SAS Institute v. Iancu—mean it's "clear there are problems with the IPR process that need to be addressed,"...
- This Week in Managed Care: April 20, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Nevada opioid panel updated on efforts to reduce painkiller toll (reviewjournal.com)
The governor’s task force on the opioid crisis met for the second time...to receive a progress report on its efforts to rein in abuse and death resulting from prescription painkillers...Representatives of health care organizations and Nevada officials told members of the Governor’s Opioid State Action Accountability Task Force that progress was being made on four priorities identified by the panel at its first meeting...: prescriber education, treatment options, data collection and criminal justice interventions...Specifics included obtaining federal funding for three new treatment centers; development of informational presentations for schools and law enforcement; distribution of the opioid reversal drug naloxone to law enforcement; and creation of the Opioid Dashboard, a publicly available collection of state data related to the epidemic...At the task force’s third meeting in July, presenters promised to present updates on other task force goals, including identifying ways to compile real-time overdose data...