- How Doctors Are Getting Rich on Urine Tests for Opioid Patients (bloomberg.com)
Want to turn that pain clinic into a real moneymaker? Open your own urine-testing lab and start billing Medicare...The cups of urine travel by express mail to the Comprehensive Pain Specialists lab in an industrial park in Brentwood, Tennessee...Most days bring more than 700 of the little sealed cups from clinics across 10 states, wrapped in red-tagged waste bags...The high-tech testing lab’s raw material has become liquid gold for the doctors who own Comprehensive Pain Specialists. This testing process, driven by the nation’s epidemic of painkiller addiction, generates profits across the doctor-owned network of 54 clinics, the largest pain-treatment practice in the Southeast. Medicare paid the company at least $11 million for urine and related tests in 2014...the costly tests are medically justified to monitor patients on pain pills against risks of addiction or even of selling pills on the black market. “I have to know the medicine is safe and you’re taking it,”...As alarm spread about opioid deaths and overdoses in the past decade, doctors who prescribed the pills were looking for ways to prevent abuse and avert liability. Entrepreneurs saw a lucrative business model: persuade doctors that testing would keep them out of trouble with licensing boards or law enforcement and protect their patients from harm. Some companies offered doctors technical help opening up their own labs...“drug testing is not about medicine but about making money, and I am going to show you how to make a lot of money.”...
- CEO of Botox maker Allergan on Mohawk tribe patent deal: ‘It wasn’t desperation, it was tenacity’ (cnbc.com)Senators blast Allergan's tribal deal, social contract as 'hypocrisy' (fiercepharma.com)Allergan executives named in investor suit over alleged generics price fixing (fiercepharma.com)
"Mad Money" host Jim Cramer and Allergan CEO Brent Saunders discussed the biopharmaceutical company's prospects...Allergan's unusual patent deal with a Native American tribe was misunderstood by Wall Street...Allergan's controversial deal with the Saint Regis Mohawk tribe giving the group the patent rights to one of its key drugs was widely misunderstood..."It wasn't desperation, it was tenacity."...In exchange for the patents for Allergan's eye drug, Restasis, and $13.75 million (plus potentially $15 million in annual royalties), the tribe granted Allergan an exclusive license to sell the drug...The deal involving the billion-dollar drug drew widespread criticism, raising concerns that Allergan could now raise the price of Restasis while keeping it under patent protection...Saunders acknowledged big pharma's duty to patients and consumers..."The flip side of that is I have a social contract that says when we are successful, we'll price it responsibly and we'll make them accessible to those people who can't afford them,"..."And I think people got confused that said because I believe in strong intellectual property, I don't believe in making medicines affordable and accessible, and one has nothing to do with the other."
- This Week in Managed Care: November 3, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Potential CVS-Aetna merger: Top 6 industry implications (managedhealthcareexecutive.modernmedicine.com)
- The way could be paved for other new healthcare models...“Payers should pay close attention to the integration of pharmacy, retail, care management, and patient care that could be possible under a combined company,”...While being early to a merger like this could bode well for CVS-Aetna, it could also give other payers the roadmap they need to explore similar mergers, or scope similar partnerships without the full-scale merging of two companies...
- Intermediaries, which were intended to be the impartial arbiters, now have the ability to control markets on the demand (i.e., formulary) and the supply (i.e., provider networks, their own providers)...“While this has been a concern with the PBM industry for years, it is significantly exacerbated when the overall medical carrier is also the PBM,”...“Economically, why this is more significant is that unlike medical provider systems which have geographic diversity, making it difficult if not impossible to consolidate them, pharmacy chains have little diversity and are already organized into national chains...making it possible to buy and require the usage of their own chain, thereby controlling both what product is purchased, the price, and the supplier.”
- These complex mergers are new categories—neither payer nor provider—for which existing laws are insufficient to regulate...“It is almost impossible for a buyer to understand what they are buying, and in contracting, who is selling what,”...“Every deal now is a complex web of contracts, intermediaries, and providers. It is becoming harder and harder to understand the economics of what services are being purchased and how that compares to other alternatives, especially on the ‘vertically integrated bundles’ that are becoming the norm...
- Big data analytics technology will be front and center...“Big data will become even more important for private and government plans to more effectively coordinate and manage internally owned means of production,”..
- There will be increased negotiating power on large market drugs and fewer mouths to feed in the value chain...“Specialty higher priced drugs will likely not be too affected by the merger, but certainly larger market drugs are going to have potentially big players pushing hard on price and value to patients to drive share...”
- Patients might win—or lose...“The merger likely would lower prices and improve access, which is beneficial for both the pharmaceutical industry and patients,”...
- FDA commissioner warns drug companies of ‘disruptive’ regulations to fight opioid epidemic (cnbc.com)
The Food and Drug Administration is likely to take new actions on opioids that may be "disruptive" and "uncomfortable" to drugmakers, the agency's commissioner (Scott Gottlieb) said...In addition to seeking to treat opioid-addicted patients with alternative medications that don't produce a high, the FDA says it will look at ways to reduce exposure to the drug. That includes new ways of packaging and distribution..."For example, it's possible that a defined, short-term supply of medication could be packaged in a manner that limits the number of pills dispensed,"..."We're at a point in this crisis that we're going to have to think of ideas and taking actions that are going to be more disruptive and are going to be uncomfortable to some parties," Gottlieb told "Squawk Box." "But we have to take more vigorous action to get ahead of this."...Gottlieb said the agency is having discussions with drug companies about the new packaging solutions..."Something like this could move potentially quickly," he said. "We're invested in taking a hard look at this and seeing what the opportunities are."...
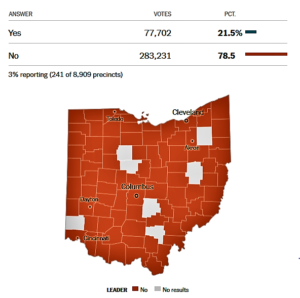
- Ohio Ballot Issue 2: Cap State Agency Drug Costs (nytimes.com)Handing pharma a win, Ohio voters overwhelmingly reject drug pricing measure (fiercepharma.com)
An initiative on Tuesday’s ballot in Ohio is aimed at reducing the cost of prescription drugs in the state. The measure would cap the price of prescription drugs purchased by the state government, including Medicaid...The measure drew strong resistance from drug makers, which spent more than $49 million to try to kill it, and the industry is not accustomed to losing political fights. Last year, it successfully killed a measure in California that was similar to the one in Ohio, but only after spending more than $100 million to do so.
- Pharmacy Week in Review: November 2, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- U.S. drug pricing watchdog gets funding to expand efforts (reuters.com)
The...Institute for Clinical and Economic Review (ICER) announced a three-year, $13.9 million grant from the Laura and John Arnold Foundation, which follows its initial two-year $5.2 million award from the foundation in 2015...ICER has previously issued reports outlining what it believes to be an appropriate cost for new medicines to treat high cholesterol, lung cancer, hepatitis C and other conditions, typically suggesting a value to patients that is a fraction of prices set by drugmakers...The new funding will enable ICER to evaluate all newly approved medicines, rather than select high profile drugs...Pharmacy benefit managers, insurers and government agencies have all used ICER reports in negotiating pricing and preferred formulary placements with manufacturers, ICER President Steven Pearson said in an interview, mentioning Express Scripts, CVS Health, the U.S. Department of Veterans Affairs and others...Rather than working from list prices as it did initially, ICER now attempts to “come up with a more precise estimate incorporating average net prices, taking rebates into account, to determine what it considers fair value-based pricing,” Pearson said...“We want to come up with an approach at ... determining when price increases are or aren’t justified,”...
- Director of UNLV HIV program kicked off campus (reviewjournal.com)UPDATE: UNLV reopens HIV clinic on day of court hearing (unlvfreepress.com)
The maternal-HIV program at UNLV that was suspended by the university six weeks ago without notice is now without a director...The university...placed Dr. Echezona Ezeanolue, the program’s director, and Dina Patel, a pediatric nurse practitioner, on administrative leave. They were escorted off the campus...Shawn Gerstenberger, dean of the school of community health sciences, asked the university to prosecute Ezeanolue and Patel for various “unsubstantiated claims of wrongdoing.”...the grant-funded program, which provides outpatient HIV primary care services to low-income, vulnerable and medically underserved women, infants, children and youth, was suspended by the university six weeks ago...President Len Jessup said previously that there were irregularities with the way the grant is being administered. Gerstenberger said an administrative audit is underway.
- CMS Shifts Coding and Payment Policy for Biosimilars Under Medicare Part B (raps.org)
The Centers for Medicare and Medicaid Services...announced that it would finalize a policy to separately code and pay for biosimilar products under Medicare Part B, signaling a win for industry...CMS said it is making the change as it was "persuaded that that there is a program need for assigning Part B biosimilar biological products into separate HCPCS [Healthcare Common Procedure Coding System] codes, specifically that this policy change will address concerns about a stronger marketplace, access to these drugs in the United States marketplace, provider and patient choice and competition."...industry groups and companies urged CMS to revise its biosimilar reimbursement policy to provide for separate HCPCS codes for each biosimilar and to reimburse each biosimilar based on its own average sale price..."Effective January 1, 2018, newly approved biosimilar biological products with a common reference product will no longer be grouped into the same HCPCS code. We will issue detailed guidance on coding, including instructions for new codes for biosimilars that are currently grouped into a common payment code and the use of modifiers....