- Report: Hanmi’s failure to report patient death in olmutinib study broke South Korea’s medical laws (endpts.com)
A Korean patient taking Hanmi’s cancer drug olmutinib died from a rare case of Stevens-Johnson syndrome a full 14 months before it was reported to the country’s health authorities, triggering a scramble that ultimately caused its partner Boehringer Ingelheim to abruptly withdraw from their $730 million partnership on the drug…Korean officials determined that Hanmi broke two medical laws related to monitoring and reporting clinical trials…the case dates back to the death in July 2015, when a patient taking olmutinib and two other drugs died from the lethal skin condition...the case dates back to the death in July 2015, when a patient taking olmutinib and two other drugs died from the lethal skin condition. The physicians involved in the case...reported the case to a monitoring agency...it was not reported as an unexpected serious adverse event...Hanmi and the monitoring agency finally reported the death to health officials in September, 2016...only after Hanmi said it first became aware of the incident...The case spurred accusations...that the influential Hanmi...had intentionally delayed reporting the death so it could get the drug approved in Korea.
- Pharmacy Week in Review: April 13, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Maryland lawmakers approve bill to fight drug price-gouging (reuters.com)
Maryland lawmakers have passed a first-in-the-nation measure that lets the state attorney general sue generic drug makers that sharply raise prices in a move aimed at fighting what legislators call "price-gouging."...The bill overwhelmingly was approved by the Democratic-controlled legislature on Monday and hailed by Maryland Attorney General Brian Frosh as a way to check sharply higher prices for crucial generic drugs…Governor Larry Hogan, a Republican, has not said whether he would sign the legislation...The bill allows Maryland's state authority on Medicaid...to let the attorney general's office know when it sees patients being charged an "unconscionable increase" for essential generic drugs...The attorney general could then seek an explanation from the manufacturer and sue to protect consumers, with a fine of up to $10,000 for each violation. A judge could order the company to reverse its price increase.
- UNR report shows health care shortage in Elko County (reviewjournal.com)
A report from the University of Nevada...shows Elko County is struggling to meet all the health care needs of its residents...Dr. John Packham presented the findings to the county’s Health Board...Packham says Elko County has "about a third of the providers for mental health as compared to the state average." He says other rural areas have seen these provider shortages but they’re more severe in Elko County due to the county’s population growth...the county is unique in that it has a younger population and doesn’t have some of the same health issues as other rural countries...According to the report, about 25 percent of the county’s population was on Medicaid and Medicare last year.
- University of California files appeal over CRISPR patents (reuters.com)
CRISPR that favored the Broad Institute…Jennifer Doudna of the University of California...and Emmanuelle Charpentier of the University of Vienna were first to apply for patent in 2012 after discovering how the primitive bacterial system...could be used to edit genomes in simple pieces of DNA…A team at the Broad Institute led by...Feng Zhang applied for a separate patent six months later, but paid for a fast-track review process, which landed them the first CRISPR patent in 2014. The Broad's patents were for showing that the CRISPR system could be used to edit more advanced, eukaryotic cells, including animal and human cells...In its...decision, an appeals board...determined that the Broad's CRISPR patents "did not interfere" with those awarded to the UC because they were sufficiently different, allowing them to stand...In the appeal...UC is seeking a reversal of the decision, which ended before actually determining who invented the use of CRISPR in eukaryotic cells. Major commercial applications of CRISPR are expected to be in eukaryotic cells...geneticist George Church said he expects the disputes will end in cross-licensing...I'm not that interested in the details of who pays who what. We're all going to do very well, including the patients. That was evident from the very beginning…
- Illuminating drug discovery by mapping all known drugs (pharmaceutical-technology.com)
A comprehensive map of all existing drugs and how they work, created from information from several huge datasets, could be used to kick-start the next wave of drug discovery. Not only could scientists use the map to suggest promising new targets for diseases as diverse as cancer, mental illness and inflammatory conditions, it could also provide invaluable insight into where existing drugs can be used to treat different diseases...Jointly led by scientists at the Institute of Cancer Research...the research brought together vast amounts of information from datasets including the canSAR database...the ChEMBL database...the...DrugCentral database to create a map of all 1,578 licensed drugs and their mechanisms of action...Dr Bissan Al-Lazikani, explains how this first-of-its-kind map could help tackle one of the biggest challenges facing the drug discovery field: how do you innovate without taking risks that make the whole system unviable?
- Why did you decide that a comprehensive map of all existing drugs and their mechanisms of action was needed for the drug discovery community?
- How did you put the map together? It must have been quite a task!
- What were the most interesting findings of the latest study?
- What are the biggest challenges facing the drug discovery field at present and how will your comprehensive map help overcome these?
- Doctor tells U.S. court drug not suitable for Arkansas executions (reuters.com)
A surgeon told a federal court in Arkansas...that a sedative the state plans to use in its lethal injection mix is not suitable for surgery and should be prohibited when Arkansas holds an unprecedented series of executions later this month...Arkansas plans to kill eight prisoners in dual executions...Death penalty opponents have said the rushed schedule is reckless and increases the chance of errors...The convicted murderers scheduled to die have asked U.S. District Judge Kristine Baker in Little Rock to halt their executions, saying the state's rush to the death chamber was unconstitutional...Lawyers for Arkansas, have told the court that the drug in question, midazolam, has been used in executions in other states and its lethal injection protocols pass constitutional muster...Arkansas Governor Asa Hutchinson...set the schedule, saying the state's midazolam supply expires at the end of April and it was in the interest of justice to hold as many executions as possible while Arkansas has the difficult-to-obtain drug.
- This Week in Managed Care: April 7, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network.
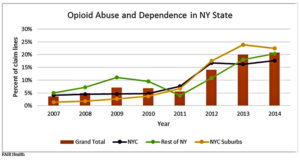
- The Growing Opioid Crisis: Spotlight on New York Private Claims Data (realclearhealth.com)
From 2007 to 2014, private insurance claim lines with opioid abuse and dependence diagnoses increased 487 percent in New York State. The greatest increase occurred in the New York City suburbs (Nassau, Rockland, Suffolk and Westchester), where the rise was 1,459 percent—compared to 324 percent for New York City and 310 percent for the rest of the state...These dramatic trends were identified when we investigated recent opioid-related data from New York State in our FAIR Health database of over 23 billion privately billed healthcare claims, the largest such repository in the country...In this article, we report the findings in terms of "claim lines," which are the individual services or procedures listed on an insurance claim. "Percent of claim lines" is the percent of all claim lines associated with a specific group of diagnosis codes...particularly the codes associated with opioid abuse or dependence—in a defined time period…
- Upstate New York
- Opioid-Related Diagnoses by Category
- Overdoses by Age and Gender
- Nevada State Board of Pharmacy Newsletter, April 2017 (bop.nv.gov)
- Newsletter Goes Electronic
- More on Opiate Addiction
- DEA Changes Registration Renewal Process
- ISMP Medication Safety Self Assessment for Community/Ambulatory Pharmacy
- CDC Publishes Resource to Foster Use of JCPP Pharmacists’ Patient Care Process
- FDA Issues Final Guidance on Repackaging Drugs by Pharmacies and Registered
- Outsourcing Facilities
- CriticalPoint Launches QP503A CertificationProgram for Sterile Compounding in 2017
- PTCB Suspends Implementation of Planned 2020 Accredited Education Requirement for Pharmacy Technicians
- ASOP Global Spreads Awareness About Illegal Online Drug Sellers and Counterfeit Medications
- New Interactive Map Tracks Pharmacist Vaccination Laws
- PMP Data Submission Accuracy
- Attention Pharmacy Managers!