- NHS report promises ‘over 1,300’ pharmacists in GP surgeries by 2019 (pharmaceutical-journal.com)
‘Next steps on the NHS five year forward view’ outlines progress made so far and highlights priorities for the next few years, including plans for 900 clinical pharmacists in GP surgeries by 2018 and over 1,300 by 2019...according to plans announced on 31 March 2017 by Simon Stevens, chief executive of NHS England...it expects GP surgeries to work in "hubs" or networks enabling them to share community nursing, mental health and clinical pharmacy teams and also work more closely with community pharmacists to make "fuller use of the contribution they make"...Sandra Gidley, chair of the Royal Pharmaceutical Society’s English Pharmacy Board, welcomes the document but says that the clinical skills of pharmacists are still an untapped resource...The National Pharmacy Association, the trade association for independent community pharmacies in the UK, shared this view in a statement. "Local pharmacies can do far more in urgent care and the management of long-term conditions, and take pressure off other parts of the system including GPs and hospitals...We will need to make sure that pharmacists have a significant role in clinical care, regardless of where they practice as the forward view is implemented."...
- Former StubHub CTO to launch mental health care app based in South Lake Tahoe (nnbw.com)
A Bay Area startup guru (Shawn Kernes, chief technology officer, StubHub) is gearing up to launch an on-demand mental health care app to reduce the cost and increase the availability of therapy across the country — and he's doing it from South Lake Tahoe...Enter: Larkr, an app designed to connect patients with certified mental health care professionals through video chat...There are 50 million people in the U.S. that are in need of some level of mental health care, and only about 20 million of them are actually receiving it...And in small towns with limited resources, it's even more difficult to find a practitioner when you need one...not to mention the cost when you do...By removing some of the costs associated with therapy, like office space and answering services, Larkr charges $85 for a 50-minute session and offers a simplified process through the app for submitting for reimbursement from insurance companies...Clients can book recurring appointments, get reminders for appointments, and have video therapy sessions from anywhere there is internet access...South Lake Tahoe is also a perfect example of the type of town that needs a service like Larkr...Kernes has plans to secure a physical office in South Lake Tahoe and hire a larger team as the startup progresses. The beta launch of the Larkr iPhone app is scheduled for July, with the full launch of the iPhone and Android apps slated for September...
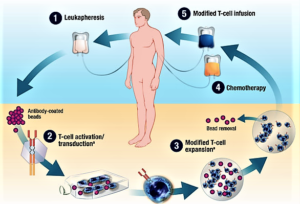
- The First CAR-T Drugs Have Left the Gate (fool.com)
Investors should keep an eye on this promising way to treat cancer...For all the talk about biotechs being nimble, it's a big pharma that looks like it'll be the first company to launch a chimeric antigen receptor T-cell (CAR-T) product...Novartis announced last week that the Food and Drug Administration accepted its application to market tisagenlecleucel-T...in patients with B-cell acute lymphoblastic leukemia who are relapsed and refractory to other therapies...A few days later, Kite Pharma completed its application for axicabtagene ciloleucel...Kite's application could be accepted early, putting it less than two months behind Novartis…Since CAR-T therapies are personalized treatments that have to be made individually for each patient, they're likely to be expensive to produce and therefore require a premium price. The first company to get a CAR-T therapy approved will set the price, which later companies may have to match unless they can justify a higher price with higher efficacy...With prices that will probably exceed those of current cancer treatments, investors should expect some pushback from insurers. One way Novarits and Kite can get around the cost issue is by offering money-back guarantees...Kite's and Novartis' CAR-T therapies are just the tip of the iceberg for this new way to treat cancer...
- This Week in Managed Care: April 7, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Health Canada to increase transparency (cmaj.ca)
Health Canada plans to increase transparency into its regulatory process for drugs and medical devices by publicly releasing clinical information submitted by companies who receive market authorization...Opening access to clinical information used to support the authorization has widespread health system benefits and can help Canadians make informed decisions about their health…Under the proposed transparency policy, the information will be kept confidential during the regulatory process but disclosed if the drug or medical device is approved for sale. According to Health Canada, this information is "far more comprehensive" than data available in sources such as medical journals and clinical trial registries...Companies will be able to request, however, that certain parts of the clinical information they submit be exempt from public release...Health Canada states that this transparency initiative will increase public confidence in the drug and medical device review process; help patients and health care providers make more informed decisions about drugs and medical devices; avoid duplication in clinical trials; and make clinical trial data available for secondary analysis to advance medical science...
- IBM receives patent that could accelerate discovery of more effective and safer drugs (drugstorenews.com)
IBM...announced its scientists have been granted a patent ("Method and system for exploring the associations between drug side-effects and therapeutic indications") on machine learning models to predict therapeutic indications and side effects from various drug information sources, which could accelerate discovery of more effective and safer drugs...IBM Research has implemented a cognitive association engine to identify significant linkages between predicted therapeutic indications and side effects, and a visual analytics system to support the interactive exploration of these associations...this approach could help researchers in pharmaceutical companies to generate hypotheses for drug discovery...strongly correlated disease-side-effect pairs identified by the patented invention could be beneficial for drug discovery in many ways. One could use the side-effect information to repurpose existing treatments (e.g. drugs causing postural hypotension could be potential candidates for treating hypertension). If a new drug is being designed for a disease that is strongly correlated with severe side effects, then special attention could be paid to controlling the formulation and dosing of the drug in the clinical trials to prevent serious safety issues...Lack of efficacy and adverse side effects are two of the primary reasons a drug fails clinical trials, each accounting for around 30% of failures...Computational models and machine learning methods that can derive useful insights from large amounts of data on drugs and diseases from various sources hold great promise for...improving the drug discovery process.
- European Regulatory Roundup: EMA Drafts Guidelines on Controlling False Positives in Clinical Trials (raps.org)
The European Medicines Agency has released draft guidelines about multiplicity in clinical trials. The text deals with how to mitigate the risk of false positives arising when clinical trials look at multiple treatment groups and endpoints...EMA’s guideline is applicable to most clinical trials. Once a study design expands beyond having two treatment groups, one predefined null hypothesis and a single primary variable — or adds an interim analysis — its sponsor needs to control for false positives. EMA cites the example of a trial that analyzes five subgroups independently. If each subgroup has a significance level of 2.5%, the likelihood of the study finding a statistically significant false positive hits 12%. As biotechs are under pressure to succeed, there is a risk they will seize on anomalies as evidence a drug works...To ensure trial designs diminish the risk of this happening, EMA has released a 15-page guideline. The draft runs through the situations in which it is necessary to adjust for multiplicity, when it is possible to draw reliable conclusions from subgroup analyses, when success against secondary endpoints can form the basis for additional claims and how to handle composite endpoints.
- Pharmaceutical packaging: where should drug companies focus their efforts? (pharmaceutical-technology.com)
The future of pharmaceutical packaging operations is far from straightforward. The Association for Packaging and Processing Technologies (PMMI) reveals where drug companies should be focusing their efforts. ...last year, the Association for Packaging and Processing Technologies released a comprehensive research report on the trends and opportunities in pharmaceutical and medical device packaging operations. The road ahead for drug companies certainly won’t be an easy one...Not only are companies scrambling to meet looming serialisation deadlines...they’re also being asked to manufacture more products for less money without sacrificing quality, and come up with innovative solutions for the pharmaceuticals of the future, which will be increasingly individualised, long-lasting and easy to self-administer. On top of that, mergers and acquisitions remain rife and generic drugs continue to gain market share...With both the pharmaceutical and medical device industries facing many of the same challenges and market realities, the two are also starting to converge, as they address new government regulations, often by purchasing new equipment, learn to produce more for less and work together on the combination products of the future.
- Serialisation
- Outsourcing
- New products, new packaging
- Nevada coalition wants drugmakers to reimburse diabetics (reviewjournal.com)
Nevadans dismayed by soaring drug prices told lawmakers...the state should take unprecedented action to curb costs...A coalition of hotel and casino owners, union leaders and Democratic lawmakers are attempting to mandate pharmaceutical companies refund people for overpriced American insulin...The bill targets diabetes medications...would require drugmakers to reimburse Nevada patients and insurers for what they pay above the highest price in other developed countries for the same prescriptions. Drugmakers would also have to reimburse people if American insulin prices increase more than inflation would suggest they should...Senate Bill 265 would require manufacturers to disclose the costs of all research, materials, manufacturing and administrative expenses that go into producing drugs for diabetics. Additionally, it would force them to notify government health agencies and insurance companies 90 days before they increase prices above the preceding year’s inflation rate...Nevada’s proposal would also have the state license all pharmaceutical sales representatives...Implementing that program in Nevada would cost about $350,000 and upkeep would cost $125,000 annually after that, according to the state Department of Health and Human Services...
- Ex-New York assemblyman, doctors charged in illegal opioid prescription scheme: officials (reuters.com)
A former New York assemblyman and a dozen pain clinic workers were arrested on Friday, accused of operating some of the largest "pill mills" in the northeastern United States and illegally prescribing more than 6 million opioid pills...Alec Brook-Krasny, who served in the New York State Assembly from 2006 to 2015 representing South Brooklyn, was charged with conspiracy and scheming to defraud by unlawfully selling prescriptions…Also arrested in the sweep of three pain clinics were at least one nurse practitioner, three physician's assistants, and two doctors including Dr. Lazar Feygin, who owned two of the clinics...They were accused of over-prescribing oxycodone...to clinic patients who showed signs of selling pills or abusing other narcotics...in exchange for oxycodone, the defendants also pressured patients to undergo unnecessary medical tests and procedures, then billed Medicare and Medicaid for millions of dollars...