- FDA scolds Indian API maker for not ensuring river water it uses is safe (fiercepharma.com)
The FDA has suggested an Indian API maker hire a consultant that understands FDA regulations after finding the company didn’t have a grasp of some basic requirements, including how to make sure the water that comes from a nearby river is sanitary before using it to make its products...In a warning letter to Badrivishal Chemicals & Pharmaceuticals, the FDA said its plant in Maharashtra has been using the river water since 2014 without scientific evidence that the system is capable of producing water that meets quality standards...plant employees knew the water for months was out of spec for total aerobic microbial counts when it was used in production, but they didn’t investigate or even seem to understand the process that the plant’s water system relies on to kill microorganisms...water was only one of the problems FDA investigators found...Records were found in trash bags behind a building that showed test results that were different from what had been officially recorded...
- Industry groups petition the FDA over off-label rule (mmm-online.com)
Industry groups are taking aim at a rule created by the Food and Drug Administration that could restrict drugmakers' truthful and non-misleading promotion of its drugs — commonly referred to as off-label promotion — by expanding the evidence the agency can use to assert that a drug has been misbranded...For example, under the new rule — pertaining to the FDA's definition of intended use — if a manufacturer discovers that one of its products is being used off-label and, as a result, adjusts its supply and scales its manufacturing to meet higher demand for those off-label uses, the FDA could use the company's internal documents describing that intention to scale up as evidence of a new intended use...The agency expanded the definition of intended use to now include “totality of evidence.”...The new intended use rule exposes manufacturers to a significant risk of liability for conduct that is entirely lawful and beneficial to the public health...
- Pharmacy Week in Review: March 17, 2017 (pharmacytimes.com)
Nicole Crisano, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharmacy among best-paying job categories in America (pharmacist.com)
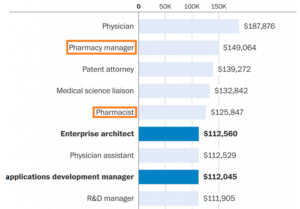
Pharmacy managers and pharmacists both landed in the top five of Glassdoor's annual list of the best-paying jobs in the United States. Glassdoor...noted health care jobs showed up in four of the top five spots. For the third year in a row, "physician" took the top slot, with a median base salary of $187,876...Pharmacy managers ($149,064) and pharmacists ($125,847) were listed at number two and number five, respectively. "Medical science liaisons" ($132,842)—specialists who work for pharmaceutical or biotech companies to establish relationships with medical experts—came in at number four.
- Nevada to expand mental health services with new community-based clinics (reviewjournal.com)
The U.S. Department of Health and Human Services announced...that Nevada will expand mental health care services with the implementation of new community-based behavioral health care clinics...The whole idea is to try to provide better integrative care to individuals that have both substance abuse and mental disorders...We’ve never done well in this state, and probably most other states, in coordinating so that patients are getting care by the same team…the two-year Certified Community Behavioral Health Clinic...will offer 24-hour mobile crisis services, outpatient mental health and substance abuse treatment as well as recovery support.
- This Week in Managed Care: March 17, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- How a Major Drugmaker Plans to Cure Disease… Without Drugs (fool.com)
Hacking into the body's nervous system may allow GlaxoSmithKline to commercialize an entirely new field of medicine...GlaxoSmithKline plc has the resources to pursue long-term bets in medicine that could eventually change the course of disease treatment and yield large payoffs...to literally eavesdrop on the body's electrical system and enter into the body's own internal conversations in order to heal disease...every organ has a nerve connection that regulates its function. Organs are controlled by patterns of electrical impulses transmitted through nerve fibers, but when organs dysfunction in chronic disease, the electrical patterns are different. That fact opens up the possibility that inserting patterns in nerves to certain organs can correct conditions that lead to disease...scientists are just beginning to realize the possibilities of using nerve signals to restore organs to normal function and actually treat disease...Bioelectronic medicines have the potential of doing to the pharmaceutical industry what biopharmaceuticals did to small molecules back in the 1980s…
- 45-foot truck takes health care on road in Southern Nevada (reviewjournal.com)
Medicine on the Move...The unusual clinic on wheels is a partnership between Health Plan of Nevada and Southwest Medical Associates. It has been operating since April, as many as six days a week. Drivers Ernie Drown and Chris Philips take the 45-foot truck to locations across the state, but primarily in Southern Nevada...The clinic can handle immunizations, X-rays, ultrasounds, and physical and laboratory exams. It has been to the Las Vegas Rescue Mission, FamilyConnect, St. Patrick’s Church food pantry, the Volunteers of America shelter for the homeless and more...The idea behind the mobile clinic was that we really need to reach our patients out in the community and make it easy for them to access health care,"’ said Toni Corbin, senior vice president of operations at Southwest Medical Associates. "We’re taking health care on the road to where it’s needed."
- Trump’s first budget seeks to slash $6B from NIH, raise FDA user fees (fiercebiotech.com)
President Donald Trump’s first budget will take $5.8 billion away from the National Institutes of Health, around 20% of its total, with FDA user fees also set to rise as biopharmas should "pay for their share."...The budget cut to the NIH...had around $30 billion in funds last year...The NIH...got a funding boost just last year when the 21st Century Cures Act was passed, a law that allowed the Institute an extra $4.8 billion in funding over the next decade...There was no direct mention of FDA cuts, but use fees are set to potentially double..Recalibrates Food and Drug Administration medical product user fees to over $2 billion in 2018, approximately $1 billion over the 2017 annualized CR level, and replaces the need for new budget authority to cover pre-market review costs. To complement the increase in medical product user fees, the Budget includes a package of administrative actions designed to achieve regulatory efficiency and speed the development of safe and effective medical products...
- New UK drug cost rules leave companies fuming (reuters.com)
British drugmakers...accused Theresa May's Conservative government of breaking a manifesto commitment to improve access to new medicines, following approval of new cost rules...The angry response from both Big Pharma and biotech firms comes despite a concession by government in the latest version of the scheme, which increases the cost threshold for certain drugs for rare diseases from the previously planned level...The row comes at a sensitive time for the government, which is about to trigger proceedings to leave the European Union but wants to encourage investment by strategic industries, including the high-tech pharmaceuticals sector...Drug companies are already concerned that Brexit could make Britain a less attractive market, especially if the country ends up outside the current EU-wide system for drug licensing...The new system means that new drugs costing the National Health Service more than 20 million pounds ($24 million) a year will no longer be automatically funded, even if they are cost-effective. Instead, companies will have to enter negotiations to justify their use and work out funding...