- This Week in Managed Care: January 27, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Pharmacy Week in Review: January 27, 2017 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Experts fret over a new hiring freeze at the FDA. Will the fallout be toxic to drug reviews? (endpts.com)
With hundreds of open positions and a slate of new initiatives to execute on at the FDA, President Donald Trump picked a particularly inconvenient time to declare a hiring freeze for the federal government...No one really knows how this is going to play out, but the speculation is rampant that the FDA will be forced to cut loose from its review timelines, which could have a big impact on a wide range of biopharma companies...the agency is expected to get a record 1600 ANDAs this year. Trump’s freeze could slow reviews on generics to a glacial pace, which in turn would interfere with the introduction of new discounted drugs that could go a long way to removing pressure on new drug prices...The only guarantee Trump can offer now is a rising level of uncertainty over the administration’s plans for the FDA. And that could take months to clarify during a particularly critical year for the biopharma industry...
- Top 5 Things to Know About Future Drug Spending (drugtopics.modernmedicine.com)
The United States and other countries will continue to spend more on specialty medications, and less will be spent on brand-name drugs...spending on drugs in the U.S. will grow at a much slower rate, according to the Quintiles IMS Institute report, "Outlook for Global Medicines Through 2021: Balancing Cost and Value."...
- THE GROWTH RATE for U.S. spending on medicines will decline by half, from 12% in 2015 to between 6% and 7% in 2017. Plus, prescription drug spending is forecast to grow between 6% and 9% through 2021…
- U.S. BRAND DRUG PRICES will increase at a slower rate, due both to competition from generics and Congressional backlash over soaring brand prices...Brand prices will increase at 8% to 11% — more slowly than the 12% to 15% in the past three years….
- SPECIALTY MEDICINES will lift the share of global heathcare spending from 30% in 2016 to 35% in 2021, driven by the adoption of new breakthrough medicines...
- PATIENT OUT-OF-POCKET COSTS are forecast to decline, despite rising brand prescription costs, as patients shift to newly available generics and receive co-pay assistance for brands…
- SEVERAL NEW THERAPIES are moving through the registration process around the world and are expected to come to market soon. In the anti-infectives and antivirals category, new treatments for HIV, bacterial disease, anthrax, hepatitis C, and malaria will be launched...
- Financial ties between researchers and the pharmaceutical industry might distort clinical trial results (pharmaceutical-journal.com)
Analyses of data from 195 clinical trials show that financial ties between researchers and the pharmaceutical industry were associated with a 3.57 greater odds of a positive trial outcome...Clinical trials involving principal investigators with financial ties to the pharmaceutical industry are more likely to report positive results...The researchers...say their findings — which held even after accounting for the influence of study sponsorship — suggest that financial ties could be leading to bias in the clinical evidence base...Given the importance of industry and academic collaboration in advancing the development of new treatments, more thought needs to be given to the roles that investigators, policy makers, and journal editors can play in ensuring the credibility of the evidence base...
- PhRMA-backed report analyzes ‘complex process’ of drug pricing (biopharmadive.com)
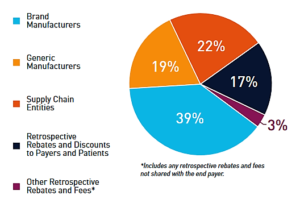
The PhRMA-backed report made the case that drugmakers are not solely to blame for ballooning treatment prices, which have come under intense scrutiny from state and federal legislatures, industry executives and research organizations...As competition in the pharmaceutical marketplace has increased in recent years, brand manufacturers have been making larger payments for market access to their medicines...Government-mandated discounts and fees have also increased over the last five years. Many of these discounts are not plainly visible, leading to misperceptions about the relative share of gross and net drug expenditures realized by brand manufacturers...each player has taken home large chunks of the drug sales revenue as well...non-manufacturers, which include wholesalers, PBMs, health plans and pharmacies, snag 42% of gross drug spending — which doesn't take into account any type of discount — from the initial point-of-sale payment for a drug by a payer or patient...Branded drugmakers take home 39% of those gross expenditures, with generics manufacturers gobbling up 19%...
- Critics eye repeal of ObamaCare prescription drug tax (thehill.com)
Employers and drugmakers are eager to say good riddance to an excise tax on brand-name prescription medicines that could get stripped under the latest GOP plan to repeal ObamaCare...While the law’s other major taxes, like the medical device or so-called Cadillac taxes, generated major campaigns seeking their repeal, the prescription drug fee has garnered little publicity. But it’s no negligible element among the law's funding sources. It's expected to bring in $27 billion over a 10-year period, according to the Joint Committee on Taxation's 2010 estimates...But the branded pharmaceutical fee never attracted bipartisan backing for repeal and it went into effect in 2010...consumers are "insensitive to changes in the drug prices."...if someone needs a specialty drug that is likely to be a brand-name product, his or her insurance is going to pay for it or that person will have to pay out of pocket for that amount...if the price happens to go up, you’re still going to pay for the drug...The fight over repealing other ObamaCare taxes means it's still unclear whether the prescription fee will be gone in the first round of the health law’s repeal measures...there could be other opportunities to get rid of it. ObamaCare repeal is looking to be...a series of legislative actions and regulatory actions over the course of the next year or so...
- ISMP Report Questions Safety of Hepatitis C Drugs (pharmacytimes.com)
A recently-published Institute for Safe Medication Practices report details updated concerns about newer drugs used to treat hepatitis C virus. The report, which is published in ISMP’s QuarterWatch, highlights concerns about the safety of HCV drugs in light of findings that point to associated liver failure and injury...The FDA designated 46 different drugs in development as breakthroughs last year, and although some of these medications have resulted in cure in an estimated 9 out of 10 patients treated, ISMP has identified hundreds of patients that reported antiviral failure...Liver failure occurred more frequently in men (55%) and in patients with a median age of 61 years old. Thirty one percent of cases resulted in death...Although the report questions the safety of these drugs, experts argue that the findings are inconclusive and prescribers should not be influenced by the results...These new data raise more questions than they resolve about the adverse effects of direct-acting antiviral drugs...A better understanding of what is occurring in hundreds of additional liver failure cases should be a priority for further investigation...
- Why biosimilars could be market disruptors (biopharmadive.com)
Just as generic drugs revolutionized the small molecule space, biosimilars are poised to have a major impact on the development and commercialization of biologics. These highly similar, but not necessarily interchangeable drugs, have been part of the European drug market for years, but are just starting to emerge in the U.S. While still early days, the developing biosimilars market is one to watch.
- Provider status legislation co-sponsored by 108 House Representatives (drugstorenews.com)
A little more than one week following its reintroduction in the Senate, provider status legislation is again being entertained in the House...the Pharmacy and Medically Underserved Areas Enhancement Act (H.R.592)...will make it easier for Medicare patients in underserved communities to receive care...The...Act would allow Medicare beneficiaries to receive basic care such as immunizations, diabetes management, blood pressure screenings and routine checks from pharmacists. The bill reached impressive levels of bipartisan support… There is currently no avenue for Medicare to directly reimburse pharmacists for providing this care...The work already is underway to build on the momentum that was started in the last Congress, to accelerate the campaign to enhance the quality, accessibility and affordability of patient care through pharmacist-provided services...Pharmacists are highly-accessible, clinically-trained medication experts who can improve health outcomes and reduce overall costs…We hope the common-sense, bicameral, bipartisan legislation, which also generated a lot of support in the previous Congress, can pass both chambers and make it to President Trump’s desk for his signature...