- FDA warning letter tells Clorox to clean up Aplicare plant (fiercepharma.com)
The FDA says the unit of Clorox that manufactures povidone-iodine drug products needs to clean a big mess at a plant in Meriden, Connecticut, which failed to follow steps to insure the sterility of its wound products—products that the FDA also said are “unapproved.”...The agency...posted a warning letter for the Aplicare plant…(it) said...the plant failed to implement adequate microbial controls...some of its products are unapproved because they do not comply with the FDA’s OTC Final Monograph for Topical Antifungal Drug Products.
- C-suiters: Keep an eye on these technologies in 2017 (fiercehealthcare.com)
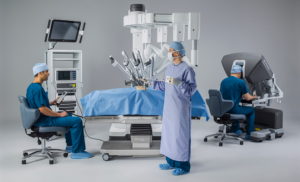
From Pepper, the emotional interactive robot, to the new da Vinci Xi Integrated Table Motion to pipeline vaccines, ECRI Institute highlights 10 healthcare technologies C-suiters (Chief-level executives) should keep an eye on in 2017...The organization's annual watch list outlines technologies that hospital leaders should bring into their hospitals—and which ones to keep out. But separating the facts from the hype isn't easy, they note...Here's the list of 10 technologies and research areas that ECRI says are poised to affect care delivery over the next 12 to 18 months:
- Liquid biopsies: ...these genetic tests that use blood or urine, rather than biopsied tissue, to identify genetic variants and mutations.
- Opioid addiction or relapse prediction: A couple of lab tests have emerged to find genetic variants believed to be associated with addiction, but critics are skeptical.
- Abdominal surgery improvements: Some major research universities are working on initiatives that add a web-based risk-assessment algorithm and interactive patient coaching in order to improve outcomes and cut costs of abdominal surgery.
- Long-term planning: Healthcare leaders should scan the horizon for important and disruptive new technology developments and care processes to revisit and refresh long-term planning efforts.
- LED infection control: New and highly effective technologies emit light in the “deep UV” range and can be used as a disinfectant on myriad surfaces.
- Robotic artificial intelligence: Two Belgian hospitals use the Pepper robot to support reception staff, but its programming comes at a hefty price.
- Robotic surgery tables: This table communicates with the da Vinci system’s robotic arms to reduce time required for re-positioning.
- New endoscopy technique: Using a near-infrared imaging technique, indocyanine green imaging enables physicians to visualize malignant tissue that is undetectable under conventional white light.
- Crohn’s disease treatments: Immunotherapy and stem cell treatments look promising, though approval is likely one or two years away.
- Vaccines for Type 1 diabetes: Keep an eye out for both preventative and therapeutic versions.
- This Week in Managed Care: January 6, 2017 (ajmc.com)
Laura Joszt, with The American Journal of Managed Care. Welcome to This Week in Managed Care, from the Managed Markets News Network.
- 7 takeover targets to look out for in 2017 (biopharmadive.com)
A promising drug candidate, an innovative technology, a great business model or just a bunch of venture funding. Any one of those factors can prime a biopharma for success, yet is also likely to attract attention from industry peers and competitors...While 2016 wasn't a groundbreaking year for M&A, industry followers believe next year might see an uptick amid a new tax plan under president-elect Trump, continued loss of patent protections for top-tier branded drugs and strong growth projections for promising fields such as gene editing and immuno-oncology...With that in mind, here's a crop of takeover targets likely to make their way into the spotlight in 2017…
- Spark
- Tesaro
- Axovant
- Biogen
- BioMarin
- Vanda
- Actelion
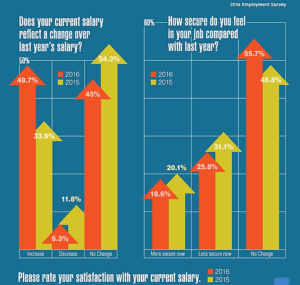
- Career Consensus: 2016 Employment Survey Europe-based bio/pharma employees are unified on satisfaction with employment conditions—and dissatisfaction with salaries (images2.advanstar.com)
While the pending exit of the United Kingdom from the European Union has generated uncertainty for industries and employees, including the bio/pharma markets across Europe, bio/pharma employees also expressed concerns about compensation, job security, and career development in the 2016 Pharmaceutical Technology/Pharmaceutical Technology Europe annual employment survey. Overall, the 2016 survey responses were similar to opinions about the employment market as the 2015 survey, but respondents also expressed stronger desires to seek better pay and career opportunities. Nearly three-quarters agreed or strongly agreed that their job was secure despite changes at their companies. More than half of the respondents reported an organizational change in their companies; 26.7% via merger or acquisition and 24.8% by downsizing/restructuring.
- Joint Commission reaffirms ban on texting orders (fiercehealthcare.com)
Citing burdens on clinicians that could lead to treatment delays, the Joint Commission said its ban on doctors using text messages to order treatments will stay in place for the time being...The Joint Commission...with the Centers for Medicare & Medicaid Services...are working out guidelines for how doctors can use messaging apps and texting to make orders...a number of concerns that led to the decision:
- Using texts or other messaging apps to order treatments could increase the burden on nurses or other clinical staff who would be responsible for inputting such data into electronic health records
- Talking in-person allows for easier clarifications if there are questions about an order, and allows for better confirmation of directives
- ...any clinical decision support alerts triggered during the EHR process, the clinician inputting the information into the system will have to take time to contact the ordering physician to resolve the issue, potentially causing treatment delays
- Greek prosecutors raid Novartis offices, disclose wide-ranging probe into bribery allegations (fiercepharma.com)
Greek officials announced Tuesday that they are investigating Novartis for bribery in the wake of local media reports raising questions about the company. It is the fourth set of bribery allegations…to go public in the past year…Greek authorities have interviewed scores of sources and raided Novartis offices in Greece, according to multiple local news outlets. Justice Minister Stavros Kontonis ordered the inquiry after "denunciations concerning bribes paid to functionaries by Novartis" appeared in the press… Greece’s investigation follows a similar probe launched in South Korea last February and corruption allegations leveled by a Turkish whistleblower…South Korean prosecutors indicted a half-dozen Novartis executives for issuing improper rebates to local doctors…last year, Novartis agreed to pay $25 million to settle an SEC investigation into bribery allegations in China…Novartis offered Chinese doctors lavish entertainment…and other inducements to boost prescriptions of its drugs in the country.
- The FDA to study whether people can recognize misleading drug ads (mmm-online.com)
The FDA has proposed two studies that would seek to determine how well consumers and healthcare providers can identify deceptive prescription drug claims in branded marketing materials...The first study would investigate the ability of consumers and HCPs to detect false or misleading promotions when showed two mock pharmaceutical websites, one site oriented toward providers and a separate one for consumers...The second study would focus on examining whether the participants can identify the difference between implicit and explicit deceptive claims...is the claim overtly wrong? Or is it misleading because it understates certain information?...and whether those claims affect their attitudes and intentions toward the promoted drug...The notice comes as the agency grapples with an industry pushing for the sharing of truthful, non-misleading information with more stakeholders outside of a drug's approved label
- Pharmacy Week in Review: January 6, 2017 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- FDA releases guidance on electronic drug applications (biopharmadive.com)
The Food and Drug Administration is ironing out new rules for how drug companies can electronically submit medications for approval, and on Dec. 28 unveiled a draft of what they have planned so far…The draft largely focused on manufacturing establishment information (MEI) — things like a production facility's address, what it's manufacturing, how to reach the person in charge of scheduling inspections, as well as a unique facility identifier, which the agency uses to keep track manufacturing plants…The FDA is requiring companies to electronically submit MEI for each facility, but all in a single list. Companies must provide those lists for new drug applications (NDAs), abbreviated new drug applications (ANDAs), biologics license applications (BLAs), and any other supplemental materials… Once the final version of the guidance gains approval, drugmakers will have 24 months before the new rules become mandatory…The agency noted several reasons for the push toward electronic MEI submissions. The primary one was the convenience of having all the information on a single file versus non-electronic applications, where it can be scattered.