- This Week in Managed Care: October 14, 2016 (ajmc.com)
Justin Gallagher, associate publisher of The American Journal of Managed Care. Welcome to This Week in Managed Care, From the Managed Markets News Network...the top stories in managed care included HHS releasing the final rule for the Medicare Access and CHIP Reauthorization Act, a commentary on the downside of drug coupons, and the World Health Organization called on countries to enact a soda tax.
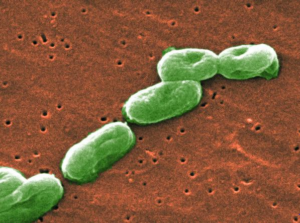
- Feds blame multistate B. cepacia outbreak on PharmaTech plant’s water system (fiercepharma.com)
After a months-long investigation, federal officials have nailed down the source of a Burkholderia cepacia outbreak that made its way into several states and infected dozens of patients...the Centers for Disease Control and Prevention and the FDA said they had detected the B. cepacia bacteria in the water system at Florida-based CMO PharmaTech. The company had produced 10 contaminated lots of constipation drug docusate sodium before 6 distributors shipped the meds around the country. In total, 60 people in 8 states became infected...The outbreak started in late June in ventilated cystic fibrosis patients. During the investigation, CDC officials said infections could be life-threatening in patients with compromised immune systems or lung conditions.
- What experimental drug? Most companies don’t post compassionate use policies (statnews.com)
As patients clamor for greater access to experimental medicines, a survey released Tuesday finds that just 19 percent of 100 drug makers publicly post policies about their programs for obtaining these drugs, which are known as compassionate use. Moreover, only one of those companies posted information about specific procedures for making requests...The findings underscore arguments by a growing number of patient advocacy groups and lawmakers that the process for gaining access to experimental medicines is difficult, a complaint that has generated criticism of the Food and Drug Administration and sparked social media shaming campaigns of some companies...But other say the frustration directed at the FDA is misplaced. That’s because drug makers are actually the final gatekeepers and may deny requests in order to meet strict criteria needed to win FDA approval for a medicine...The FDA should clearly state in policy how adverse events under compassionate use will affect the ongoing clinical trial process for companies. While the FDA claims that this is a rare occurrence, the reality is that the FDA’s failure to issue clear policy leaves companies in a precarious position of jeopardizing their investments. If companies had clarity from the FDA about how offering drugs under the compassionate use program would impact ongoing trials, they’d be more likely to make their policies public and, possibly, to participate.
- Manufacturer of cosmetic and pharmaceutical products expands into Dayton (nnbw.com)
Cosmetic Enterprises, Ltd., a growing manufacturer of cosmetic and pharmaceutical products, is planning to expand its operations into Dayton...CEL, headquartered in Pacoima, Calif., purchased a vacant 60,000-square-foot building along with an adjacent 18 acres for potential development..."During our due diligence we reviewed many different regions and states and found Nevada to be manufacturing friendly that is centrally located to our existing customer base…Cosmetic Enterprises is a great addition to Lyon County...Not only are they creating the kind of higher paying jobs that help boost the region, they will be great corporate citizens...CEL produces cosmetic and skin care products, such as shampoo, moisturizers, skin enhancing lotion and skin protecting products, using botanicals and other raw materials...
- Pfizer’s Lyrica patent appeal fails in U.K., endangering bid to protect $5B med (fiercepharma.com)
Pfizer’s divide-and-conquer approach to Lyrica in the U.K. just hit a wall. The Court of Appeal upheld a ruling that struck down key patent claims on Lyrica and cleared Actavis’ generic of infringing it...The...case centered on a “carve-out” approval for Actavis’ Lyrica generic, a type of regulatory nod that branded drugmakers see as a threat...drugmakers want to prolong their monopoly access to patients, and they use follow-up patents to extend their protection past the time when IP coverage expires on the original compounds...some recent “skinny” regulatory nods--from the FDA as well as international regulatory agencies--that clear generics only for particular indications have complicated those efforts, because they give generics makers an entreé onto the market while so-called method-of-use patents remain in effect...The patent at issue in this case covered Lyrica’s use as a pain treatment; the patent on pregabalin itself, the active ingredient...had already expired. These days, Lyrica is used more often for pain than for its original indication as a seizure drug...The company hopes to now take its fight to the U.K. Supreme Court...Pfizer maintains its strong belief in the validity and importance...of the patent…
- Humana’s slashed Medicare rating may signal higher bar for all insurers (cnbc.com)
Humana says it plans to fight the Obama administration over a large reduction in the ratings of its 4-star Medicare Advantage plans for coverage starting in 2018...Star ratings are a key quality measure under Medicare, and lower ratings could result in reduced reimbursement rates for Humana...Not having 4-stars on a significant portion of your plans will hurt you financially because you will not be able to achieve the bonus reimbursement that higher rated plans receive…The company said issues resulting in reduced stars included delays in resolving enrollee complaints over claims...Clearly Humana is going to fight this...it's too early to determine what the real impact of the star rating reduction could be on Humana's earnings in 2018...They're raising the bar, in the sense that it is becoming increasingly demanding...to get the highest ratings on Medicare plans...In other words, insurers are not getting worse, but rather facing tougher comparisons...The good news is that the quality of everyone is improving, but [CMS] still wants to mark on a curve… so, by definition, not everyone can be a five-star...
- Pharmacist Training for Controlled Prescriptions (pharmacytimes.com)
Carlos Torrado, PharmD, JD, PRS explains the training pharmacists receive with respect to prescriptions for controlled substances.
- Huge Valeant price hike on lead poisoning drug sparks anger (statnews.com)
A drug used to treat lead poisoning is causing a toxic reaction among hospitals and poison control centers after Valeant Pharmaceuticals jacked up the price more than 2,700 percent in a single year...At issue is a decades-old, intravenous treatment for severe and life-threatening cases of lead poisoning, which occur infrequently, but generally require supplies to be on hand...Calcium EDTA, Valeant acquired the drug in 2013…By December 2014...increases took the price to $26,927 (from $950)...Ever since, poison control specialists have been angry, especially since there are few viable options...Their reaction is a case study in the exasperation doctors and hospitals feel at the multitude of price hikes of all sizes that they see month after month...create frustration and squeeze budgets behind the scenes...There’s no justification for the astronomical price increases...which limit availability of the drug to children with life-threatening lead poisoning...
- Rising drug prices are making hospitals feel ill (statnews.com)
While much of the attention over prescription drug prices is focused on consumers, a survey released on Tuesday finds that hospitals are also spending much more than in the past. Between 2013 and 2015, the average annual drug spending for patients who stay in community hospitals increased by of 23.4 percent...And on a per admission basis, hospital spending on drugs jumped nearly 39 percent, to $990...the increase in prices outpaced reimbursement rates from payers, retail spending on medicines, and the pharmaceutical price inflation...the survey found that more than 90 percent of the hospitals surveyed reported that recent price hikes for inpatient drugs had a moderate or severe effect on managing costs...The furor has put the pharmaceutical industry on the defensive as many drug makers are scrambling to justify their price hikes. Some drug makers are pointing fingers at pharmacy benefits managers, and the convoluted pricing system in which rebates that are paid to win coveted placement on lists of drugs for which insurance coverage is provided...The drugs that are increasing in price generally are not new and are off patent. There is no competition, so hospitals are forced to pay in order to make the best patient care decisions...These price increases are unsustainable for hospitals who are taking care of sicker and sicker patients…
- How Pharmacy School Curriculums Have Changed (pharmacytimes.com)
Lynette Bradley-Baker, PhD, RPh, vice president of public affairs and engagement of the American Association of Colleges of Pharmacy, describes how pharmacy school curriculums have changed.