- Buyers remorse or fraud? Teva and Mexican brothers slug it out over sour deal (statnews.com)
Drug makers are constantly foraging for deals to bulk up their pipelines, but not all go according to plan...Consider the nasty spat between Teva Pharmaceuticals and two Mexican brothers, Fernando and Leopoldo Espinosa...they have filed dueling lawsuits after their $2.3 billion deal went sour…At issue was a move made last year by Teva...to expand in Mexico by purchasing Rimsa, one of the country’s largest independent pharmaceutical manufacturers, from the Espinosa brothers. At the time, Teva hailed its acquisition...as a "significant platform for growth" in the second-largest market in Latin America...Teva claims the brothers engaged in chicanery, ploys, and misrepresentations about their business in order to walk away with huge profits, according to its lawsuit...Rimsa was "was engaged in a years-long scheme to sell defective and unlawful products and to conceal those violations from Mexican regulators...Teva lawsuit goes on to claim that Rimsa submitted fraudulent information to regulators about ingredients suppliers and lied about laboratory tests, including the stability tests that must ensure the stated shelf life of each product was accurate and that the product would remain stable, and, therefore, safe and effective…
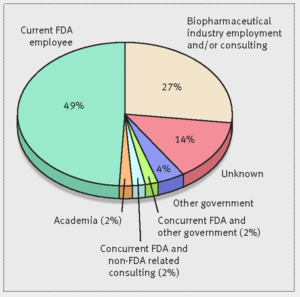
- Many ex-FDA drug reviewers take ‘revolving door’ to pharma industry (medcitynews.com)Future jobs of FDA’s haematology-oncology reviewers (bmj.com)
More than a quarter of the Food and Drug Administration employees who approved cancer and hematology drugs from 2001 through 2010 left the agency and now work or consult for pharmaceutical companies…Dr. Vinay Prasad, a hematologist-oncologist and assistant professor at Oregon Health and Science University, sought to understand the so-called "revolving door" between the FDA and the pharmaceutical industry, which he said is often discussed but hadn’t been quantified...Going to work for industry after leaving the FDA is not inherently bad, but it does raise some questions...If you know in the back of your mind that your career goal may be to someday work on the other side of the table, I wonder whether that changes the way you regulate...Are you more likely to give [companies] the benefit of the doubt? Are you less likely to beat them up hard over [using bad comparisons in drug studies]?...
- Mylan CEO Bresch used a sky-high tax rate to lowball EpiPen profits by 66% (fiercepharma.com)
Mylan CEO Heather Bresch quoted one number that got a lot of attention last week on Capitol Hill: EpiPen profits weren’t all that large, just $100 for each package of two pens, on a list price of more than $600...Thing is, that number was misleading...In a securities filing...Mylan posted a chart detailing the EpiPen numbers. The profits figure Bresch gave during the congressional committee hearing included a 37.5% tax rate--and that understated the product’s profits by $187 million...Before taxes, EpiPen profits came in at $498 million--or $166 per package. Almost two-thirds higher than Bresch had said...And not only did Bresch fail to specify that she’d deducted taxes from that profits figure, but that tax rate was 5 times higher than what Mylan paid last year, thanks to a tax inversion deal that moved its domicile from the U.S. to the Netherlands...For 2016, Mylan has forecast larger profits and a bigger operating margin for EpiPen. According to the chart Mylan filed, It’s looking for $671 million in operating profits, up from last year’s $498 million. And it expects operating margins to be 75% for the product, compared with 72% in 2015...
- What’s next for Valeant’s Sprout biz? A huge impairment charge, analyst predicts (fiercepharma.com)
Poor sales trajectory, check. Scale-back in marketing effort, check. What’s next for Valeant’s Addyi and its maker, Sprout? An impairment charge...and a big one, at that...David Maris expects to see a whopper charge on the carrying value of Sprout--as in, as much as 90% or more...And he expects to see it "with next quarter’s results, if not sooner...Valeant has already warned in a regulatory filing of a potential impairment...sales of Addyi (flibanserin) --the controversial female libido drug that Valeant shelled out $1 billion to gain access to--are dismal. In July, they had reached just $696,235, Maris wrote, citing IMS data--a far cry from the $100 million to $150 million the Canadian drugmaker forecast for this year...when the "overdue" impairment does finally come, investors will feel the burn...the write-off cut into 2017 EPS and beyond by 14 cents per share, though that’s not necessarily even the worst part…
- European regulators appeal rulings that prevent release of drug data (statnews.com)
Once again, drug makers and European regulators are clashing over disclosing information...the European Medicines Agency is appealing two different rulings by the General Court of the European Union that prevent it from releasing data to third parties…
- ... first order blocked release of a case study report for Translarna...which is sold by PTC Therapeutics...Until PTC objected, the EMA planned to provide access to the report in response to a request, albeit with redactions that agency officials maintain are in accordance with their own regulations…
- ...second order blocked the release of three toxicity studies for Bravecto...sold by Merck’s animal health unit, which also objected to disclosure.
Our approach to transparency has been welcomed by many of our stakeholders, and these court cases are a good opportunity to test our rules on making available to the general public the documents on which EMA’s scientific opinions on medicines are based...The episode was closely watched because it arose as the pharmaceutical industry faced growing disclosure pressure following scandals over safety or effectiveness data that were not publicly shared. Drug makers have argued that disclosing certain data may compromise trade secrets or patient privacy. Consumer groups counter such information is kept out of reach at the expense of patients...
- Ex-Insys sales manager arrested in U.S. fentanyl-kickback case (reuters.com)
A former Insys Therapeutics Inc district sales manager was arrested...on charges he participated in a scheme to pay kickbacks to doctors to prescribe a drug containing the opioid fentanyl...Jeffrey Pearlman...was charged in a criminal complaint filed in federal court in New Haven, Connecticut, becoming the latest individual to face prosecution in connection with probes involving Insys' drug Subsys…The charges come as Insys faces a number of state and federal investigations involving Subsys as U.S. authorities seek to combat a national epidemic of opioid abuse...Prosecutors said Pearlman and sales representatives he managed induced doctors, advanced practice registered nurses and physicians' assistants to prescribe Insys' fentanyl spray by paying them to participate in sham "speaker programs."...As a result of the scheme, federal healthcare programs incurred millions of dollars in losses, prosecutors said...
- Drug Distributors Still Benefiting from Drug Price Inflation but Face Major Profit Challenges, Says New Drug Channels Institute Study (drugchannelsinstitute.com)
Drug Channels Institute...released its...in-depth analysis of the highly dynamic prescription drug distribution industry...2016–17 Economic Report on Pharmaceutical Wholesalers and Specialty Distributors reveals how list price increases of brand-name drugs are boosting the industry’s revenues, even as changes in the U.S. healthcare system threaten wholesalers’ profits...We project that U.S. drug distribution revenues at the Big Three public wholesalers—AmerisourceBergen, Cardinal Health, and McKesson—will exceed $400 billion in 2016...But any change in drug price inflation rates or in manufacturers’ rebate strategy will be very negative for wholesalers’ future revenues and profits...Consolidation among pharmacies and hospitals is making it difficult for wholesalers to capture value and profits from the booming specialty drug market...And contrary to many people’s expectations, specialty biosimilar drugs seem poised to deliver only minimal incremental profits for wholesalers...
- This Week in Managed Care: October 1, 2016 (ajmc.com)
Justin Gallagher, associate publisher of The American Journal of Managed Care. Welcome to This Week in Managed Care, From the Managed Markets News Network.
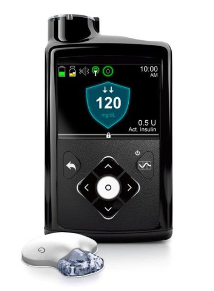
- FDA approves 1st ‘artificial pancreas’ for type 1 diabetes (upi.com)
The Food and Drug Administration...approved the first automated insulin delivery system -- a so-called "artificial pancreas" -- for people with type 1 diabetes...This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin…The device -- Medtronic's MiniMed 670G -- is what's known as a hybrid closed-loop system. That means it monitors blood sugar and then delivers necessary background insulin doses. The device will also shut off when blood sugar levels drop too low...this device isn't yet a fully automated artificial pancreas. People with type 1 diabetes will still need to figure out how many carbohydrates are in their food, and enter that information into the system, the agency noted..
- Indian union plots mass protest against ‘unfair’ treatment of pharma sales reps (fiercepharma.com)
A strike by Sun Pharma employees this week is just one symptom of a campaign by a prominent trade union to seek more equitable working conditions for pharma sales representatives...The Federation of Medical and Sales Representatives' Associations of India claims many reps working in India do so with contracts that contravene their working rights--as well as Indian employment law. It is trying to organize a mass demonstration on the streets of the capital, New Delhi, on November 21, and is threatening an all-out national strike if its demands are not met...around 500 Sun Pharma sales reps engaged in a one-day strike to protest what they call unfair treatment of workers who joined the company from Ranbaxy Laboratories, including unpaid wages and expenses, and demotions to a lower working grade…FMRAI alleges that unfair labor practices are rife in the Indian pharma market. Sales reps fear losing their jobs if they become ill--or simply at the whim of their managers...The union wants India's government to set statutory working rules for sales employees and to strictly enforce the existing Sales Promotion Employees Act. It's also calling for stiffer penalties--including the threat of jail--for employers found to have victimized staff...