- Implications of final AMP rule for community Rx (chaindrugreview.com)
On February 1, 2016, the Centers for Medicare & Medicaid Services released its Final Rule interpreting the provisions of the 2010 health care reform law that address the Medicaid Drug Rebate Program. While much of the Final Rule speaks to drug manufacturer rights and responsibilities in the MDRP, significant portions are directly applicable to chain drug stores...the Final Rule addresses (a) reimbursement for multisource drugs dispensed to Medicaid patients, (b) changes in the Average Manufacturer Price (AMP) that will impact pharmacy reimbursement for multisource products, (c) reimbursement for single-source drugs dispensed to Medicaid patients and (d) changes to the dispensing fees pharmacies will receive for dispensing drugs to Medicaid patients...An overarching theme expressed in the Final Rule is CMS’ desire to move states away from their historic practice of overpaying for ingredient cost while simultaneously underpaying for dispensing fees...If the Final Rule plays out as anticipated, in the coming year ingredient cost reimbursement will fall for single- and multiple-source products, but dispensing fees will rise...This reflects a desired shift away from a "spread-based" model of pharmacy Medicaid participation toward a "service-based" model. It’s currently unclear if the increased fee income to pharmacies will balance the lost reimbursement revenue.
- Multisource product ingredient cost reimbursement
- Changes in AMP affecting multisource drug reimbursement
- Single-source product ingredient cost reimbursement
- Medicaid pharmacy dispensing fees
- What banned drugs do cheating athletes take? (washingtonpost.com)
Hundreds of substances are named on the World Anti-Doping Agency’s list of banned drugs, and thousands more are prohibited through phrases such as "and related substances." That way, the rules cover drugs that may not have been detected or even invented yet...Most, but not all, fall into these broad categories:
- Muscle-building steroids
- Pick-me-up stimulants
- Hormones and drugs that modulate hormones
- Drugs that hide other drugs
- Calming beta blockers
- Asthma drugs
- Recreational drugs
- Anti-inflammatory steroids
- Other ways to break the rules
- Veeva Streamlines Drug Giants’ Ability To Get Medicine Approvals (investors.com)
When health care behemoth Johnson & Johnson decided last year to use Veeva Systems' software for gathering data from clinical trials needed to get a new drug approved, Veeva's rivals took notice..."They (J&J) said we're going to standardize this globally, and within 12 months we had 6 customers (among the 20 biggest drugmakers) do the exact same thing,"...one... goal in mind -- replace biopharma companies' legacy systems with cloud-based software designed to make their painstaking regulatory requirements a lot less painful...The cost of developing a new drug has soared to $2.6 billion, up from about $1 billion in 2000 in inflation-adjusted dollars, according to the Pharmaceutical Research and Manufacturers of America. Because the stakes are so high, more large drug developers have decided in rapid succession to junk their prior technology investments...The key to Veeva's success is that it has brought together enterprise software expertise with industry-specific expertise...
- Pfizer agrees to settle shareholder class action on Celebrex and Bextra (fiercepharma.com)
Pfizer’s Bextra (valdecoxib) has not been on the market in more than a decade, but the litigation tied to it and pain drug Celebrex (celecoxib) continues to play out. The New York drugmaker has now put to rest a long-running class action by Pfizer shareholders who said a controversy over the drugs’ safety whacked the stock price and cost them a lot of money...Pfizer last week reached an agreement-in-principle to resolve the securities class action case for all defendants...the resolution is pending court approval and the terms are confidential until a formal agreement has been approved...The pain drugs made Pfizer a lot of money, at one time being among the company’s best-selling meds. But the controversy over their risks has also cost it tremendously. In 2009, Pfizer agreed to hand over $2.3 billion to settle a Department of Justice probe into its marketing of Bextra...And last year, the drugmaker settled another related investor suit for $400 million.
- EU regulators say China’s Jinan Jinda still missing manufacturing mark (fiercepharma.com)
When EU regulators last year came down on China’s Jinan Jinda Pharmaceutical for slipshod manufacturing work, the company brought in a U.S. consultant to get its Zhangqiu, Shandong, plant in order. But a recent follow-up found that Jinan Jinda continues to ship its antibiotic to some European customers even though its improvement efforts fall short of what they need to be...the European Directorate for the Quality of Medicines said it has been recommended that Jinan Jinda's nitrofurantoin antibiotic be banned from the EU and that the plant's certificate of compliance be suspended, or even "voided."...The investigators reported that there were critical deficiencies in the way the plant handled raw data safety, control and out-of-specification reviews. There were major deficiencies in training, change control, quality assessment, process and cleaning validations.
- Pharmacy benefit managers are restricting access to lots of drugs next year (statnews.com)The ‘gouge factor’: Big companies want transparency in drug price negotiations (statnews.com)
In the latest bid to control prescription drug spending, the nation’s largest pharmacy benefits managers are again excluding dozens of medicines from their lists of products that are covered by health insurance…Express Scripts notified its customers that next year, 85 medicines will be excluded from its national formulary, and, as a result, the PBM hopes to recognize about $1.8 billion in savings, up from $1.3 billion this year. The number of excluded medicines, by the way, is down slightly from the 88 prescription drugs that were excluded from its 2016 formulary...CVS Health released its own 2017 formulary that will exclude another 35 medicines, including 10 that were called "hyperinflationary." As a result, CVS is now excluding a total of 131 medicines and boasts such moves have saved more than $9 billion over the last five years…Recently, a group of two dozen of the largest names in Corporate America — including Macy’s, Coca-Cola and American Express — formed an alliance (Health Transformation Alliance) to find ways to lower health care costs. One notion they are exploring is to change the way PBMs are paid due to concerns that the rebates the PBMs collect from drug makers on behalf of clients are not always passed along. The PBMs have disputed this argument.
- GSK and Google launch bioelectronics venture (pmlive.com)Q&A: Glaxo exec says bioelectronics is ‘not science fiction’ (statnews.com)
GlaxoSmithKline has joined forces with Google's Verily Life Sciences to establish a new company dedicated to the development of bioelectronic medicines...Galvani Bioelectronics - named for the 18th century Italian bioelectricity pioneer - will combine GSK's drug discovery and development expertise in disease biology with Verily's expertise in highly miniaturised technologies, including data analytics and software development for clinical application...Together, we can rapidly accelerate the pace of progress in this exciting field, to develop medicines that truly speak the electrical language of the body...Bioelectronic medicine is concerned with the electrical signals firing between the body's nervous system and organs, working to regulate the faulty nerve impulses that occur in many illnesses.
- Rogue online sellers feeding drug abuse (chaindrugreview.com)Internet Drug Outlet Identification Program Progress Report for State and Federal Regulators: July 2016 (s3.amazonaws.com)
Drug abuse, including misuse of prescription drugs, remains at alarmingly high levels...Feeding this epidemic, the National Association of Boards of Pharmacy says, are rogue Internet drug outlets illegally dispensing prescription controlled substances...illicit online drug sellers "hiding behind sleek websites" are making it easier for people struggling with drug abuse to purchase controlled-substance medications. However, since these rogue websites often sell counterfeit and tainted substances, buyers can raise the risk of overdose and death...researchers used popular search engines (Google, Bing and Yahoo), shopping websites (eBay, Craigslist, Yahoo! Shopping, Overstock.com, Etsy, Oodle, eCRATER, Bonanza, Sell.com, Blujay and Alibaba) and social media sites (YouTube and Twitter) to gauge the availability of the opioids Demerol, Dilaudid, Duragesic, Exalgo, Kadian, Lorcet, Lortab, Percocet, Roxicet, Vicodin and Zohydro. Opioids were easiest to find and readily available by using search engines, the researchers discovered. Still, no matter which online avenue was used, researchers found that the final destination was a dedicated website selling opioids illegally.
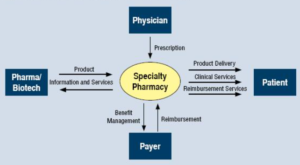
- What Services Should Patients Expect from a Specialty Pharmacy? (specialtypharmacytimes.com)
There are 2 important stakeholders when it comes to specialty medications: the patients and the providers. Both groups have a vested interest in the clinical outcome..."what would I expect if I were a specialty patient or a specialized provider?"...Once the reality of the condition begins to set in, then the needed medication must be started to improve clinical outcomes. How would the ideal specialty pharmacy interact with both the patient and their provider?...The ideal specialty pharmacy would have clear upfront communication with both parties. Proactive communication and referral management for the provider would allow the specialist to know precisely what services and capabilities the specialty pharmacy provides...The provider then could inform the newly diagnosed patient that...their expertise, and that of the specialty pharmacy, will ensure the best possible clinical outcomes. At this point, the referral has been sent to the specialty pharmacy, and the patient's interaction with the specialty pharmacy begins.
- Dept of Health to launch sepsis awareness campaign across UK (pmlive.com)
An awareness campaign to improve public recognition of sepsis will be launched across the UK this year, carried out by the Department of Health and Public Health England...Working in collaboration with the UK Sepsis Trust, the initiative aims to better public understanding of the blood poisoning condition that requires quick diagnosis and treatment to effectively combat...Approximately 150,000 adults and children in the UK are affected by sepsis annually, with around 44,000 of those dying each year. If not identified and treated quickly, survivors of sepsis can be left with life-changing disabilities... better sepsis care could save 13,500 lives every year and save the NHS £314m annually...The public awareness drive - called Just ASK: could it be sepsis? - hopes to equip the general public with knowledge of the condition so that they might recognise it and prompt healthcare professionals to fast-track diagnosis of the time-sensitive illness.