- More than 1 million OxyContin pills ended up in the hands of criminals and addicts. What the drugmaker knew (latimes.com)
In the waning days of summer in 2008, a convicted felon and his business partner leased office space on a seedy block near MacArthur Park. They set up a waiting room, hired an elderly physician and gave the place a name that sounded like an ordinary clinic: Lake Medical...The doctor began prescribing the opioid painkiller OxyContin – in extraordinary quantities. In a single week in September, she issued orders for 1,500 pills, more than entire pharmacies sold in a month. In October, it was 11,000 pills. By December, she had prescribed more than 73,000, with a street value of nearly $6 million...Purdue Pharma, the maker of OxyContin, tracked the surge in prescriptions. A sales manager went to check out the clinic and the company launched an investigation. It eventually concluded that Lake Medical was working with a corrupt pharmacy in Huntington Park to obtain large quantities of OxyContin.
- Sounding the alarm
- What Purdue knew
- Pills prescribed by Santiago
- Following the pills
- The skid row connection
- Pharmacist complaints
- Reports of concern
- ‘I was sitting on a gold mine.’
- ‘It really takes the ‘G’ a long time to catch up with these jokers’
- China healthcare costs forcing patients into crippling debt (reuters.com)
As China's medical bills rise steeply, outpacing government insurance provision, patients and their families are increasingly turning to loans to pay for healthcare, adding to the country's growing burden of consumer debt...While public health insurance reaches nearly all of China's 1.4 billion people, its coverage is basic, leaving patients liable for about half of total healthcare spending, with the proportion rising further for serious or chronic diseases such as cancer and diabetes...Medical loans are just part of China's debt mountain - consumer borrowing has tripled since 2010 to nearly 21 trillion yuan, and in eight years household debt relative to the economy has doubled to nearly 40 percent...
- Alibaba Health Expands Into New Consumer Business After Setbacks (bloomberg.com)
Alibaba Health Information Technology Ltd…the subsidiary of China’s biggest e-commerce operator is attempting to revive its fortunes by tapping new areas of growth -- like consumer safety...China...scrapped plans to implement a compulsory drug coding system intended to identify counterfeit medicines that would have used a platform developed by Ali Health. The Alibaba Group Holding Ltd. subsidiary says it is now using the same technology to build a commercial tracking platform that will have wider applications beyond medicines...Ali Health’s new system could be used to track the origins of products such as baby formula, farm produce and even wine for Chinese consumers concerned about food safety…Ali Health is currently talking to partners to expand use of the technology, and continues to run the existing drug tracking system for the government while awaiting a final decision. The China Food & Drug Administration suspended the electronic coding system earlier this year for use in the drug industry as it made draft amendments to existing rules, allowing the use of other methods to track medicines back to their origin...
- Drug maker is warned over missing study about child opioids use (statnews.com)
...one drug maker has failed to provide the Food and Drug Administration with a required study about the effect its powerful painkiller might have on teenagers...At issue is Xartemis XR (oxycodone/acetaminophen), which Mallinckrodt Pharmaceuticals received regulatory approval to sell two years ago. At the time, the company was also required to conduct a so-called post marketing study to determine its safety in youngsters between 12 and 17 years old…as of June 24, the company blew past a March 31 deadline for submitting its pediatric assessment. The drug maker also failed to respond to an April 25 "noncompliance" letter for which the FDA provided a new 45-day deadline to either submit the data or request an extension...The assessment was required under the Pediatric Research Equity Act...What happens if the company fails to comply and submit the data to the FDA? According to federal law, the agency could determine that Xartemis XR is misbranded. That would not lead to a product withdrawal, but the FDA could pursue an injunction or seizure proceedings...
- Drug maker loses battle over Canada’s right to impose a price cap (statnews.com)
A closely watched skirmish over the cost of prescription drugs has ended in defeat for a company that sought to challenge the right of a Canadian agency to impose a price cap on a pricey medicine...Canada’s Federal Court dismissed a constitutional challenge that Alexion Pharmaceuticals filed against the Patented Medicine Prices Review Board, which...sought to keep a lid on the cost of the company’s Soliris (eculizumab) medication...The...agency had asked Alexion to lower its price and repay sales generated by the drug from 2012 through the first half of 2014..."It is far from surprising," said Richard Gold, a professor at McGill University..."Alexion may still appeal in hopes that the Supreme Court of Canada will alter the law. I think the [possibility that the court will find] the provisions unconstitutional are remote. Parliament can create and limit patent rights as it wishes, but pharmaceutical companies have been known for being obstinate in the face of logic."
- Theranos dealt severe blow by CMS (drugstorenews.com)
Less than one month following Walgreens decision to terminate its relationship with Theranos, the blood-testing lab on Thursday announced that the Centers for Medicare & Medicaid Services has revoked the company's CLIA certificate, among other sanctions. The revocation of Theranos' CLIA certificate precludes the owners and operators of Theranos from owning, operating or directing a lab until at least July 2018...In addition to the revocation of Theranos' CLIA certificate, the full list of CMS sanctions include:
- Limitation of the laboratory’s CLIA certificate for the specialty of hematology;
- A civil money penalty;
- A directed portion of a plan of correction;
- Suspension of the laboratory’s approval to receive Medicare and Medicaid payments for any services performed for the specialty of hematology; and
- Cancellation of the laboratory’s approval to receive Medicare and Medicaid payments for all laboratory services.
- Pharmacy Week in Review: July 8, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Pharmacy Journal Examines National Trends in Prescription Drug Spending (ashp.org)National trends in prescription drug expenditures and projections for 2016 - Abstract (ajhp.org)
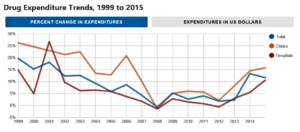
A sharp rise in prescription medication prices — driven by the introduction of new, expensive specialty medications and huge increases in the costs of older drug products with few competitors — drove an 11.7 percent increase in spending on medications in 2015, according to a new report published in AJHP (American Journal of Health-System Pharmacy)...The report projects an 11 to 13 percent increase in total drug expenditures in hospitals and health systems in 2016, which includes a 15 to 17 percent increase in clinic spending and a 10 to 12 percent increase in hospital spending...pharmacists need to be diligent about understanding what factors influence drug spending within their own organizations. "By understanding the key cost drivers, pharmacy leaders can implement programs to target them," he noted, adding that such strategies could include working with prescribers to use equally effective but less expensive medications or developing programs such as antibiotic stewardship to reduce unnecessary use of drugs...
- Doctors turn militant over Venezuela’s health crisis (reuters.com)
A dozen doctors hold a hunger strike in the corridors of an Andean city hospital. In another provincial city, hundreds of protesting medics suspend appointments...With eight out of 10 medicines now scarce, according to the main pharmacy group, protesting doctors are demanding that President Nicolas Maduro's socialist government declare a national health crisis and allow foreign humanitarian aid...In June, Pino (Christian Pino, a surgeon at the Merida hospital) read a list of doctors' demands in Venezuela's National Assembly before the opposition-led legislature declared a state of medical emergency and approved channels for foreign humanitarian aid...But the...Supreme Court shot down the assembly's proposal. Government officials deny Venezuela is facing a humanitarian crisis and say there is no need for humanitarian assistance...Idabelias Arias, the head of the emergency ward at a pediatric hospital in Barquisimeto..."Doctors are doing war medicine here."
- Pfizer agrees to opioid marketing deal to escape a lawsuit by Chicago (statnews.com)
Pfizer reached an agreement with the city of Chicago to follow a code of conduct for marketing opioids that officials hope will become standard practice for other companies...The drug maker agreed last week to disclose in all promotional materials that opioids carry a risk of addiction and not to promote them for any unapproved uses...Pfizer must make clear that there is insufficient research about the effectiveness of opioids if used beyond 12 weeks…Some industry watchers, meanwhile, said that the four-year agreement, which does not pertain to any other city or state, largely requires Pfizer to adhere to existing laws and regulations governing drug marketing. For this reason, they said the agreement breaks little new ground in the battle against inappropriate or overprescribing of opioids..."This landmark agreement is a big step in the right direction to help protect and educate the public about the true risks and benefits of highly potent and highly addictive painkillers," said Chicago Mayor Rahm Emanuel in a statement...It’s in Pfizer’s interest to highlight the addictive properties of opioids because they have a competing product under development (tanezumab)...When a company comes out slamming a particular class of drugs, it’s generally because they have a competitor in the wings. Think of this as prelaunch marketing, which can start long before the launch...