- Q&A: Why the World Health Organization plans a fair pricing model for drugs (statnews.com)
...World Health Organization wants to develop a fair pricing model for pharmaceuticals...the agency plans to convene governments, patient groups, and drug makers to fashion the notion into something realistic. The trick is to find the right balance between access to affordable medicines and enticing companies to develop new and improved medicines, while also ensuring lower-cost generics remain available. We spoke with Suzanne Hill, the secretary to the WHO Expert Committee on the Selection and Use of Essential Medicines, about how the agency hopes to get this done…
Pharmalot: This seems rather lofty or at least complicated. How is this supposed to get done?
Hill: We want to get evidence together that looks at the price drivers — how manufacturers set prices and what we know and don’t know about that. And we need to understand the questions surrounding R&D costs. We also want to understand the strategies that countries, payers, and health systems are using to manage prices and which ones may be effective and which aren’t. We also want to understand the return on investment that manufacturers need to ensure that supplies of medicines are continued...
- Court ruling on biosimilar launches could increase health care costs (statnews.com)
A federal appeals court...that biosimilar makers must always notify their brand-name rivals six months before launching expensive biologic medicines. The decision may have a significant impact on near-term health care costs, because it will effectively delay competition for these pricey drugs...The stipulation is designed to give a brand-name company time to determine what, if any, patent challenges can be pursued before a biosimilar is launched. In a closely watched case last year, the federal appeals court already ruled that biosimilar companies must wait until they actually receive Food and Drug Administration approval before giving 180-day notice to a brand-name rival...today’s decision will answer an important policy question and serve as a broader benchmark for the entire pharmaceutical industry going forward...A great deal is at stake for patients and payers because biosimilars are estimated to save as much as $44 billion in US health care costs over the next decade. Many insurers and analysts forecast that biosimilars will cost 10 percent to 30 percent less than brand-name biologics, although the number is a moving target, given that companies raise biologics prices to anticipate competition...
- This Week in Managed Care: July 2, 2016 (ajmc.com)
Justin Gallagher, associate publisher of The American Journal of Managed Care. Welcome to This Week in Managed Care, from the Managed Markets News Network.
- FDA warnings slam Chinese drugmakers, including ViiV partner (fiercepharma.com)
...two Chinese companies with ties to Western drugmakers have been called on the mat to account for problems in their manufacturing. The FDA has issued warning letters to plants operated by Shanghai Desano Chemical Pharmaceutical and Chongqing Lummy Pharmaceutical, slamming them both for manipulating testing and turning in falsified batch test results on APIs...
- Shanghai Desano Chemical Pharmaceutical - FDA...criticized the facility for conducting "unofficial" tests of drug batches that it kept out of its official record...also disturbed by finding many electronic logs of production deviations in a folder titled "GMP Anomalies" that had never been investigated…
- Chongqing Lummy Pharmaceutical...FDA...warning letter slams the drugmaker for widespread and serious data manipulation of batch analyses...In one egregious case, the FDA...an analyst set the gas chromatography personal computer clock back to make it appear as if testing had been done...7 months earlier. The analyst then performed 5 injections to produce falsified results for long-term stability for a finished API lot, deleted four and reported only the results of the final injection as passing in the quality-control...
- Gilead’s new price hikes on HIV drugs anger AIDS activists (statnews.com)Gilead hikes price of HIV therapies, other drugs by up to 10 percent (statnews.com)
As part of a strategy to switch patients to newer HIV treatments, Gilead Sciences...raised prices on a pair of older HIV medications that face patent expiration...triggering still more criticism by AIDS activists of its overall pricing strategies...the company raised the wholesale acquisition cost...for the two older medicines — Complera (emtricitabine, rilpivirine,tenofovir) and Stribild (cobicistat, elvitegravir, emtricitabine,tenofovir) — by 7 percent, to $2,508 and $3,469 a month, respectively. This follows price hikes of 7 percent and 5 percent last January...Meanwhile, Gilead left intact prices for two much newer versions of these drugs — Odefsey (emtricitabine, rilpivirine, tenofovir) and Genvoya (elvitegravir, cobicistat, emtricitabine, tenofovir alafenamide) – which remain priced at $2,346 and $2,578 per month...By boosting prices for the older HIV treatments twice in just six months, Gilead is clearly hoping that doctors will prescribe its newer drugs. And physicians have an added impetus for doing so: the newest Gilead medications are more potent and studies have indicated they are likely to cause fewer side effects...price hikes has angered AIDS activists, who say the higher prices reflect indifference to the ability of payers to absorb higher drug costs...AIDS Healthcare Foundation believes the price hike is a good reason to push to invalidate Gilead patents...The small savings from the cheaper new drugs pales in comparison to how cheap they would be if they went generic...
- CVS to Pay $3.5M Over Allegations of Forged Prescriptions (dddmag.com)
CVS Pharmacy has agreed to pay $3.5 million to settle allegations that dozens of its Massachusetts pharmacies violated federal law by filling forged prescriptions for addictive painkillers and other controlled substances...Attorney Carmen Ortiz announced the settlement...CVS says it entered into the agreement to avoid the expense and uncertainty of further legal proceedings...settlement resolves two investigations by the Drug Enforcement Administration after reports of forged oxycodone prescriptions. One involved hundreds of forged prescriptions at 40 CVS stores in Massachusetts and New Hampshire. The other involved 120 forged prescriptions at 10 CVS stores in and around Boston.
- Audit slams Nevada dental board’s system for handling records and complaints (reviewjournal.com)
A legislative audit of the state dental board revealed systemic issues with the board’s operations, including poor record keeping and questions about how the public’s complaints are investigated...The report, presented at the June 16 meeting of the Sunset Subcommittee of the Legislative Commission, highlighted the Nevada State Board of Dental Examiners’ lack of an independent committee to review outcomes of investigations into misconduct allegations...The review process complaint was just one of several issues investigated by the auditors, who also found the board lacked "an effective process for accurately determining the amount of investigative costs" to dental professionals under scrutiny.
- J&J must pay $70 million to male teen who took Risperdal and developed large breasts (statnews.com)
Johnson & Johnson...was ordered...to pay $70 million to a male Tennessee teenager who claimed its Risperdal antipsychotic pill caused him to grow enlarged breasts. The finding by a Pennsylvania state court jury was not only the latest, but it is the biggest defeat to date in what has become another sprawling litigation over the drug....jury found that J&J failed to properly warn Risperdal could cause gynecomastia...also determined that the company "intentionally falsified, destroyed, or concealed records" that Risperdal could cause boys to develop breasts...J&J has a bad track record when it comes to marketing Risperdal...In 2013, the company paid more than $2.2 billion to resolve criminal and civil charges of illegally promoting the drug for unapproved uses...J&J reputation. The health care giant has portrayed itself as a trustworthy corporate brand, but has endured several episodes that have sullied its well-honed image...
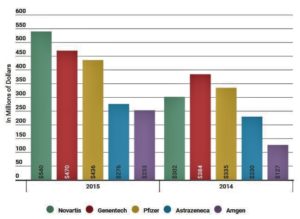
- These companies paid the most to docs last year (medcitynews.com)
The Physician Payment Sunshine Act was meant to shed light on the financial relationship between drugmakers, biotech, and medical device companies whether those payments be for general or research purposes...The Centers for Medicare and Medicaid Services has recorded these payments now for two complete years in a row with partial data available for 2013...The infographic below, created by MedCity News, based on data from CMS’s Open Payments website...shows the companies that spent the most money in 2015 in comparison with the amount they spent the year before and overall payments.
- Anthem, Express Scripts Face Legal Challenge Over Prescription Drug Prices (realclearhealth.com)Anthem sues Express Scripts for $15 billion over drug pricing (modernhealthcare.com)
Anthem and its pharmacy manager Express Scripts overcharged patients with job-based insurance for prescription drugs, alleges a lawsuit that seeks class action status for what could be tens of thousands of Americans...the latest wrinkle in a battle that has already pitted the major national insurer and its pharmacy benefit manager against each other in dueling legal actions...The case alleges that insured workers paid too much because Express Scripts charged "above competitive pricing levels" and Anthem, in effect, allowed those higher prices...Those actions...violate the firms’ responsibilities under a 1974 federal benefits law called the Employee Retirement Income Security Act...This action seeks to recover losses suffered by the plaintiffs…who overpaid and continue to overpay for the portions of the costs of prescription drugs…they are responsible for paying as plan participants," says the lawsuit, filed as Burnett v. Express Scripts and Anthem.