- Lawmakers look to prevent drug makers from thwarting generic competition (statnews.com)
For the third time...congressional lawmakers have introduced a bill designed to end a practice that generic drug makers say is used by brand-name rivals to thwart competition...Known as the Creating and Restoring Equal Access to Equivalent Samples Act, the bipartisan legislation comes amid ongoing complaints that brand-name drug makers sometimes refuse to provide samples to generic companies. They need samples to prove their copycat versions are equivalent to the brand-name drugs in order to pass regulatory muster...The legislation would allow the secretary of Health and Human Services to vet the request for samples and create a "cause of action," presumably making it easier for a generic company to go to court in hopes of forcing a brand-name rival to make samples available...Whether the legislation will gain any traction is unclear...
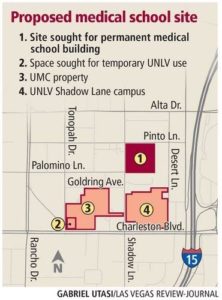
- Clark County commissioners OK site for UNLV’s new medical school (reviewjournal.com)
UNLV’s nascent medical school has a place to call home after clearing one final bureaucratic hurdle to claim a spot inside the Las Vegas Medical District...Clark County Commission...unanimously approved a plan giving the university a 9-acre site at 625 Shadow Lane, where it plans to build its new medical school. The panel also voted to let the school lease space from nearby University Medical Center while it raises money for its own building...Commissioner Chris Giunchigliani said the project has been a "long time coming" and called the school’s development "a historical moment."...School officials expect to break ground for the new building in about three to four years..."This is going to have such an impact on generations to come," Commissioner Lawrence Weekly said. "Students (are) going to be able to not have to dream about being a doctor or nurse and go to school out of state — they’ll be able to stay at home."
- Proposed Roseman medical school fails to get preliminary accreditation (reviewjournal.com)
A budding Southern Nevada medical school was dealt a blow Thursday as it learned a committee declined to grant the school’s request for preliminary accreditation, likely delaying the college’s opening...The medical school is part of the private, nonprofit Roseman University of Health Sciences...Roseman spokesman Jason Roth said it’s unclear how much of a delay the news could cause in the development of the medical college. The school is awaiting a letter from the accreditation committee outlining the reasons for the rejection...Institutions denied preliminary accreditation can choose to appeal, according to the LCME website (Liaison Committee on Medical Education). Rejected schools must wait a year before reapplying for accreditation...Roth called the news a temporary setback...
- Pharmacy Week in Review: June 17, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- NIH rejects petition to override patent on pricey prostate cancer drug (statnews.com)
National Institutes of Health...rejected a request by several consumer groups to override the patent on a prostate cancer drug because the medicine is more expensive in the United States than elsewhere...(consumer) groups petitioned the NIH to take this step, which is known as a march-in right, to help US patients because federally funded research was used to create Xtandi (enzalutamide). The drug is sold by Astellas Pharma and has an average wholesale price in the United States of more than $129,000, about two to four times more than what other high-income countries are paying...Under federal law, a march-in right allows an agency that funds private research to require a drug maker to license its patent to another party in order to "alleviate health and safety needs which are not being reasonably satisfied" or when the benefits of a drug are not available on "reasonable terms."...the NIH denied the petition because there was no information to suggest that Xtandi is or will be in short supply...The consumer group plans to submit an appeal...and said it will base its appeal on the NIH’s "flawed legal rationale" about the use of march-in rights and "its lack of analysis concerning its refusal to use a royalty-free license."
- Drug company-sponsored meals tied to more prescriptions (reuters.com)
Doctors who received even one free meal from a pharmaceutical salesperson were more likely than others to prescribe the drug being promoted, even when a generic equivalent was available, according to a new study...Each year in the U.S., $73 billion is spent on brand name drugs for which there is an equivalent generic available, and patients pay for $24 billion of that amount themselves...The brand name drugs and the generics are "so similar that there’s no benefit," from using the brand name versions...doctors who received even one sponsored meal from one of the pharmaceutical companies were more likely to prescribe the target drug over a generic alternative, compared to doctors who did not receive sponsored meals. As the number of meals and meal value increased, relative prescribing rates also increased...It’s not clear from this study whether receiving meals caused doctors to change their prescribing patterns, but "humans are very responsive to gifts...
- Factory snag hits GSK supply, causing Danish anaesthetic shortage (reuters.com)
Manufacturing problems at a factory in Italy have disrupted production of some GlaxoSmithKline medicines, leading to shortages of a commonly used opioid anaesthetic in Denmark...The..drugmaker said...it had temporarily suspended manufacturing at its Parma site, which makes sterile products, to investigate environmental monitoring...The factory has now recommenced manufacturing. However, a spokeswoman said a small number of countries had experienced shortages of certain products since April...Denmark, doctors said they were running out of the anaesthetic Ultiva (remifentanil)...
- Roche announces availability of LightMix modular Zika Virus Assay (reuters.com)
Swiss drugmaker Roche said...its LightMix Modular Zika Virus Assay (CE) is available in markets accepting the CE mark for patients with signs and symptoms of the Zika infection where the virus is known to be present...provides healthcare professionals an immediate option to detect the virus," Uwe Oberlaender, the head of Roche Molecular Diagnostics...
- Dr. Zachery Halford Named 2016 Next-Generation Pharmacist® Finalist (blog.roseman.edu)
Parata Systems and Pharmacy Times 2016 Next-Generation Pharmacist® Awards Gala, which honor pharmacists, technicians, student pharmacists and industry advocates who are defining the future of the industry, will be on August 6, at the Museum of Fine Arts in Boston. Dr. Zachery Halford, Assistant Professor of Pharmacy...for Roseman University’s College of Pharmacy, was named one of the finalists in the Specialty Pharmacist category...
- Which Is It: Prescription Drug or Tolkien Elf? (entertainment.howstuffworks.com)
Can you spot the prescription drug names among Elf names from J.R.R. Tolkien's legendarium? Test your Elven race IQ.