- FDA Approves New Version of Oxycodone (painnewsnetwork.org)
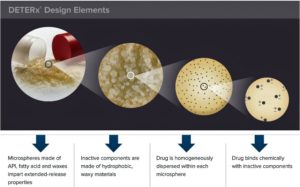
Food and Drug Administration has approved a new extended released version of the opioid painkiller oxycodone that has abuse deterrent properties unlike anything else on the market...Xtampza – can be ingested in capsule form, but users can also sprinkle the capsule contents on soft foods or into a cup, and then directly into the mouth...The medication, which can also be ingested through a feeding tube, is the sixth opioid pain medication with an abuse deterrent formula to be approved by the FDA...Xtampza is made by...Collegium Pharmaceutical with proprietary technology (DETERx technology platform) that combines oxycodone with fatty acid and waxes to form small spherical beads that are placed inside the capsule...The beads are designed to resist breaking, crushing, chewing, dissolving and melting, methods long used by drugs abusers to snort or inject opioids.
- FDA reconsiders training requirements for painkillers (hosted.ap.org)
Food and Drug Administration is reconsidering whether doctors who prescribe painkillers like OxyContin should be required to take safety training courses...The review comes as regulators disclosed that the number of doctors who completed voluntary training programs is less than half that targeted by the agency...Under the current risk-management programs, drugmakers fund voluntary training for physicians on how to safely prescribe their medications...many experts - including a previous panel of FDA advisers - said those measures don't go far enough and that physician training should be mandatory...The FDA's initial ideas to improve safety included mandatory certification for doctors and a national registry to track patients taking the drugs. But industry pushed back. Drugmakers and the pain specialty groups they fund argued that certification would be too burdensome for doctors, leaving many patients undertreated. And patient groups said that registries would unfairly stigmatize those who rely on painkillers to deal with long-term pain....
- Pharmacy Week in Review: April 29, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Walgreens Agrees to $500K Settlement for Overcharging Customers (pharmacytimes.com)
Walgreens has agreed to pay $500,000 in penalties, fees, and costs related to misleading advertising practices that led to New York customers being overcharged for products..."Businesses are required to ensure that their advertisements are truthful and not misleading..."When consumers purchase products at retail stores in New York, they should be able to rely on the prices displayed in advertisements and on shelf tags and not have to worry about being overcharged when they get to the register."...Walgreens has agreed to reform its advertising and business practices in New York...It must remove expired shelf tags within 36 hours, restrict the use of "Smart Buy" or "Great Buy," and refrain from using "Last Chance" or "Clearance" tags when the item is available at a reduced price for an extended period of time...Walgreens will also conduct internal and external price check audits in stores. If a store fails 2 consecutive external audits, it will have to pay a $2500 penalty.
- Valeant annual report reveals new issues, company overhauls board (reuters.com)
Valeant Pharmaceuticals International Inc...issued a 2015 financial report that met an important deadline for creditors, revealing new details on a range of financial and legal issues, and it also announced changes to its board...Shares in Valeant fell 4.0 percent to $33.81...and are well down from their all time high of $263.70 in August...The publication of the report satisfies demands from Valeant's creditors, but also shows the difficult road ahead...We think investors should sell on this catalyst since the business is quite weak and faces a number of headwinds...The annual report does not answer some more basic questions about what strategy Valeant will adopt...They continue to dig out of a very troubled situation that will take time...The company...revealed it is being investigated by the U.S. Department of Justice in North Carolina...relating to the production, marketing, distribution, sale and pricing of three of its life-saving drugs...Massachusetts and New York are also investigating similar issues... Valeant said that seven current board members would not be standing for re-election at its June 14 shareholder meeting...
- In China’s tougher drug market, minnows open back door for ‘Big Pharma’ (reuters.com)
Armed with Beijing funds and friends in the right places, Chinese drug minnows are thriving, luring money from 'Big Pharma' majors struggling to restore the strong growth they once enjoyed in the world's second-largest medicine market...Chinese healthcare mergers and acquisitions nearly tripled last year to more than $50 billion, helped by giants like GlaxoSmithKline PLC and Eli Lilly and Co tapping small biotech and research innovators. The targets offer vital regulatory know-how as Beijing builds a domestic drug industry...For Big Pharma, acquisitions, licensing deals and joint ventures offer a back door into a market where Beijing expects healthcare spending to rise to $1.3 trillion by 2020. The majors need the opening: their China growth has stalled to low single-digit pace from over 20 percent just four years ago as branded generics have lost their shine...
- Pfizer to pay $784.6 million to resolve Wyeth false claims lawsuit (reuters.com)
Pfizer Inc has agreed to pay $784.6 million to resolve allegations that Wyeth, which it acquired in 2009, underpaid drug rebates to Medicaid, the federal health insurance program...The settlement resolves claims by the U.S. and states that Wyeth knowingly reported false and fraudulent prices on two of its anti-acid drugs, Protonix Oral (pantoprazole) and Protonix IV...Wyeth offered hospitals deep discounts on bundles that included both drugs when made available to staff and patients...Wyeth wanted to induce hospitals to buy and use Protonix Oral, a drug they would otherwise have little incentive to prescribe because of other drugs that were already on the market and competitively priced...Wyeth hid the bundled discounts from Medicaid...
- Hawaii looks to allow psychologists to prescribe drugs (hosted.ap.org)
State lawmakers are poised to make Hawaii one of a handful of states that allow psychologists to prescribe medication in hopes of increasing access to mental health services...The...bill would allow psychologists to prescribe medication if they undergo special training. It sets requirements including 400 hours of training, supervision of 100 patients and passing an exam created by the American Psychological Association...So far, psychologists in the Department of Defense can prescribe medication, along with those in New Mexico, Louisiana and Illinois. Just this week, lawmakers in Iowa approved a similar bill. Supporters of the Hawaii bill say those states can serve as a model for increasing access to mental health care...Outreach workers say Hawaii is in the midst of a mental health crisis. More Hawaii residents die by suicide than in car accidents, according to the Hawaii Department of Health...But opponents including the American Psychiatric Association and the Hawaii Medical Association say the bill would put Hawaii residents with mental illness at serious risk. They say some psychologists might not have the proper medical training needed to safely prescribe drugs that can cause deadly reactions, especially when mixed with other medications...
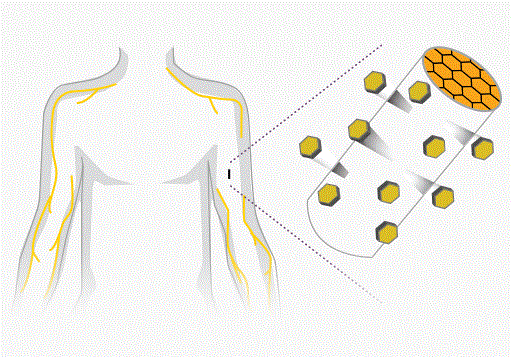
- New implant set to join fight against U.S. painkiller epidemic (reuters.com)
Two companies are on the cusp of taking a new treatment for opioid addiction to the U.S. market at a time when lawmakers are seeking ways to arrest an epidemic of heroin and painkiller abuse...Titan Pharmaceuticals Inc and...Braeburn Pharmaceuticals have together developed a matchstick-sized implant that analysts expect will be approved next month, despite mixed reviews...Implanted into the arm, the treatment is designed to be less vulnerable to abuse or illicit resale than the oral drugs that are currently used to treat opioid addiction...Two drugs are predominantly used to treat opioid addiction: methadone, which is dispensed only in government-endorsed clinics, and the less-addictive buprenorphine, which exists as a pill or strip of film...The implant, known as Probuphine, offers an alternative by administering buprenorphine for up to six months after users have first been stabilized on the oral form of the drug...Food and Drug Administration have raised reservations about possible complications from the insertion and removal of the 26mm long implant...
- The Complex Math Behind Spiraling Prescription Drug Prices (nytimes.com)
The soaring cost of prescription drugs has generated outrage among politicians and patients. Some cancer drugs carry price tags of more than $100,000 a year, and health plans are increasingly asking people to shoulder a greater share of the cost...Americans regularly cite drug prices as a top health care concern...Higher drug prices threaten to raise insurance premiums and patients’ out-of-pocket expenses and can cost taxpayers more because of Medicaid and other government programs. But drug companies say the prices reflect the enormous investment of time and resources that go into bringing a drug to market and argue that many times, their drugs can prevent more expensive medical interventions like surgery and hospitalization...So how much do drugs cost?...A drug’s path from the manufacturer to the patient is circuitous, and many middlemen are paid along the way...The pharmaceutical company sends the drug to a distributor, which takes a fee and then sells the drug to a pharmacy, which pockets its own fee before dispensing the medication to a patient. If a patient is insured, a pharmacy-benefit manager is paid for processing the transaction between the pharmacy and the insurer or employer. The pharmacy-benefit manager also handles the rebates that flow from the drug maker to the insurer or the employer...The good news first: The vast majority of drugs dispensed in the United States...are generics, which are low-cost alternatives to brand-name drugs. If your doctor writes you a prescription, there is a very good chance it will cost you $10 or less...patients who are the sickest and require the most expensive drugs are the most vulnerable to soaring drug prices..."It’s sort of embedded in the health care system that the price is never the price, unless you’re a cash-paying customer,"...