- FDA blasts California compounding pharmacy for facility ‘contaminated with filth’ (fiercepharma.com)
Auro Pharmacy has had a number of run-ins with FDA investigators...The FDA has had a long and troubled history with compounding pharmacies...sometimes those facilities bring it on themselves...Auro Pharmacies operated a veritable house of horrors at its...outsourcing facility, with ants in the sterile production areas and visibly dirty work surfaces, FDA investigators found during an August 2018 inspection...Those poor conditions could have produced supposedly sterile drugs that were "contaminated with filth,"...The FDA knocked Auro with a 10-observation Form 483 in August, 2018 that led to a voluntary recall of all the pharmacy's affected drugs and a stoppage to all sterile production...Even worse for Auro, the FDA followed up its August 2018 look-in with another round of inspections in September of last year that turned up most of the same sanitary issues, including "filth" on the end of hood-cleaning wands, and cracked and scratched production hoods...That inspection lead to a separate 11-observation Form 483 sent in October...READ MORE
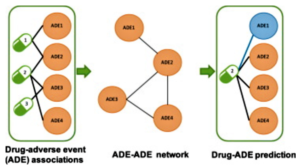
- New Machine Learning Tool Can Predict Adverse Drug Effects (drugtopics.com)Development of an adverse drug event network to predict drug toxicity (sciencedirect.com)
A new computer algorithm might be the next step toward accurate prediction of adverse drug reactions..Researchers from Harvard Medical School and the Novartis Institutes for BioMedical Research announced the creation of an open-source machine learning tool capable of predicting drug adverse effects (Aes)...The study, published in The Lancet journal EBioMedicine, examined 2 databases: 1 that reported adverse drug reactions and another with 184 proteins that specific drugs are known to interact with. Investigators constructed a computer algorithm to develop associations between the drug reactions and the 184 individual proteins...The algorithm discovered 221 associations, some known and some new. These associations indicated which proteins contribute to certain AEs and which may not...The new algorithm could help predict these AEs before the drug goes to human clinical trials, as well as before and after it enters the market...READ MORE
- FDA Publishes Guidance on CGMP Requirements During COVID-19 (pharmtech.com)
FDA published guidance on June 19, 2020 detailing the agency’s recommendations for current good manufacturing practices (CGMP) requirements for addressing COVID-19 infection in employees engaging in drug manufacturing. The guidance was issued to help mitigate and prevent effects on drug safety and quality by employees confirmed to be either infected with COVID-19 or potentially exposed to someone with COVID-19...READ MORE
- Flu shot makers gear up—and get creative—for a critical vaccination season (fiercepharma.com)
With the novel coronavirus continuing its global spread and a second wave threatening the United States later this year, experts worry an influx of influenza patients and COVID-19 patients will hit U.S. hospitals at the same time...pharma's working to ramp up not only for the increased demand but also for the logistical challenges of vaccinating millions of people during a pandemic...Manufacturers distributed about 170 million flu vaccine doses to the U.S. last year. This year, they're aiming to increase that by about 20 million...Vaccine makers are already producing their doses, with plans to start shipping later this summer...READ MORE
- Colorcon launches drug authentication platform (outsourcing-pharma.com)
The company’s SoteriaRx platform offers on-dose technology and detection services for authenticating a broad range of medications...(which)...protect patients from counterfeit products...on-dose authentication provides a powerful tool for tracking medicines from plant to patient and provides a level of supply chain authenticity and transparency not previously available. By incorporating microtags, the pill itself effectively becomes a barcode which can be digitally read and recorded, providing instant authentication...the incorporation of microtags essentially turns the pill itself into a barcode. This can be digitally read and recorded for authentication..READ MORE.
- U.S. demand outstripping supply of steroid treatment for COVID (reuters.com)Hospitals see shortages of a cheap steroid that one study says helps Covid-19 patients (statnews.com)FDA Drug Shortages (accessdata.fda.gov)
Soaring hospital demand for the steroid dexamethasone, which British researchers say significantly reduces mortality among severely ill COVID-19 patients, is outstripping supply of the drug, but hospitals have so far been able to treat patients out of their inventories, according to Vizient Inc, a drug buyer for about half of U.S. hospitals...Hospitals and other health-care customers advised by Vizient increased orders of the drug by more than 600% after the researchers announced their findings last week. Manufacturers were only able to fill around half of those orders...dexamethasone reduced death rates by nearly a third among COVID-19 patients requiring mechanical breathing assistance...The injectable version of dexamethasone has been in shortage in the U.S. since February of last year, according to the U.S. Food and Drug Administration...READ MORE
- Rising Costs Explain Why Canada Is Switching to Biosimilars (centerforbiosimilars.com)
Following in the footsteps of British Columbia, other Canadian provinces are working to implement their own biosimilars initiatives that would switch patients from some of the most costly reference biologics to biosimilar counterparts...A look at the numbers explains why Canadian provinces are introducing automatic biosimilar switching policies...In Ontario, one of 4 provinces that are moving forward with forced switching, a total of $800,000 was spent on publicly funded biologic medications in 2018, up nearly 3-fold from $259.4 million in 2010, and the projection is for the total to reach $1 billion in 2021...“The Biosimilars Initiative is a result of PharmaCare’s evidence-informed strategy to better optimize our public resources, get the best value for new treatments and services, and improve access to biologic medications for patients,” BC government officials said...READ MORE
- WHO lays out ambitious plan to deliver 2 billion coronavirus vaccine doses (biopharmadive.com)
The World Health Organization, together with partner organizations, aims to secure 2 billion doses of coronavirus vaccines by the end of 2021, unveiling...a creative plan to ensure high-risk groups around the world have access to any vaccine that's successfully developed... Through the WHO's plan, countries would be able to pool their resources to invest in the development a broad portfolio of experimental vaccines, obtaining in return a guaranteed share of the resulting supplies. The idea is to lessen the risk of betting on any one vaccine, while creating a mechanism by which doses are fairly allocated during the initial stages of a vaccine's availability...READ MORE
- Vaccine makers face biggest medical manufacturing challenge in history (reuters.com)
Developing a COVID-19 vaccine in record time will be tough. Producing enough to end the pandemic will be the biggest medical manufacturing feat in history...From deploying experts amid global travel restrictions to managing extreme storage conditions, and even inventing new kinds of vials and syringes for billions of doses, the path is strewn with formidable hurdles, according to Reuters interviews with more than a dozen vaccine developers and their backers...Any hitch in an untested supply chain - which could stretch from Pune in India to England’s Oxford and Baltimore in the United States - could torpedo or delay the complex process...READ MORE
- America’s global leadership in biopharmaceutical manufacturing (catalyst.phrma.org)
Unfortunately, there are a lot of assertions floating around that tell a misleading story about pharmaceutical manufacturing, diminishing America’s leadership and suggesting our reliance on other countries has put us at risk of potential shortages. In reality...the biopharmaceutical industry supports more than 4 million jobs across the United States, directly employing more than 811,000 Americans. Of those jobs, nearly 120,000 are high-wage manufacturing jobs, which is double the percentage of manufacturing jobs compared to the private sector overall...Discussions about enabling more manufacturing in the United States are important, but let’s not forget that the United States already sustains a substantial manufacturing presence that is part of a larger global network. We cannot replace all global manufacturing with solely U.S. manufacturing without upsetting the entire biopharmaceutical supply chain to the detriment of patients...READ MORE