- Pharmacy Week in Review: March 4, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
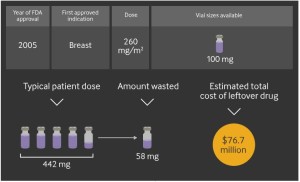
- Study: $3B will be wasted on unused portion of cancer drugs (cnbc.com)Overspending driven by oversized single dose vials of cancer drugs (bmj.com)
High prices for cancer medicines aren't the only reason they cost insurers and patients so much...Waste pads the bill…because infused cancer drugs are distributed in...vials that usually contain more medicine than most patients need. Most of the time that excess is thrown out, even though it's perfectly good — and worth hundreds or thousands of dollars...Researchers...estimate that wasted cancer medicine in the U.S. this year will add up to nearly $3 billion in excess costs...cancer medicines are one of the highest-priced, fastest-growing drug categories...study...in BMJ...details how drugmakers, hospitals and cancer doctors make money on unused cancer medicine. It focuses on the top 20 drugs for multiple cancer types packaged in single-dose vials and for which the dose depends on the patient's weight, finding that 1 percent to 33 percent of those 20 cancer drugs, on average, remains in vials after each dose is administered...The researchers say regulators could require manufacturers to supply multiple vial-size options, and the FDA and other federal health agencies need to reconcile their disparate guidelines on vial size and when leftover medicine from one vial can be given to another patient. Currently, sharing generally is only allowed within six hours of when the vial is opened to ensure the medicine's sterility.
- His Job Is to Sell a $1,000 Pill for $10 Without Losing Money (bloomberg.com)
Gregg Alton has what seems like a disorienting job at Gilead Sciences Inc. He’s paid to figure out how to sell the drug Sovaldi, which infamously retails in the U.S. at $1,000 a pill, for relatively next to nothing...The instructions for pricing the cure, which wipes out hepatitis C in just 12 weeks, basically go like this: "Get to as many patients as possible in low-income nations --and not lose money," Alton says. "It’s very simple."...Actually, it’s not so simple, but it is controversial. Decisions about what to charge around the world for life-saving remedies have spurred debate ever since Big Pharma began offering some discounts after a backlash in the ’90s, when groundbreaking HIV treatments reduced deaths in wealthy countries and not poor ones. Criticism has been harsh with Sovaldi, one of the most expensive and best-selling drugs in history...He offers a rare look at how a pharmaceutical giant walks the line between shareholder expectations on the one hand and global public health needs on the other. It starts with his team looking at the map through the lens of per-capita income. Countries that are well-off go into one bucket. The rest are sorted by wealth and rate of hepatitis C infection...What’s frustrating, he says, is that the good Gilead does in other countries is overshadowed by reverberations from the U.S. sticker shock. However his company tries to explain the value it brings to patients, it can’t compete, he says. "It’s never going to be as powerful as ‘$1,000 a pill? Is that fair?’"
- 13 Smart Ways to Cut High Drug Costs (time.com)
Your medicine may come with a new side effect: financial pain. Prescription-drug spending grew 12.2% in 2014—five times as fast as the year before—according to the Centers for Medicare & Medicaid Services. And the sickest Americans bear the biggest burden. Some 43% of those in fair or poor health say it’s somewhat or very difficult to afford their medications, and 37% say they’ve skipped out on filling a prescription because of cost...What has changed? Generic drugs, long an affordable alternative to name-brand medicines, have become part of the problem. The average price of the 50 most popular generic drugs increased 373% between 2010 and 2014...One culprit is consolidation: After a decade of mergers, three big companies now control 40% of the generics market...Weaker competition means drug companies can charge your insurer more. Meanwhile, pricey new miracle drugs—like hepatitis C treatment Sovaldi are also a key factor forcing up overall medication costs...Fortunately, there are plenty of ways for you to save. By making strategic changes in the medications you take (with your doctor’s okay, of course), the places you buy them, and the insurance plan you elect, you may be able to shave 40% or more off your total prescription-drug costs this year. Here are the steps you need to take.
- Substitute generics for name brands.
- Combine pills—or split them.
- Check the formulary for your insurer’s favorites.
- Jump through your insurer’s hoops.
- Ask your plan to make an exception.
- Use mail-order options.
- Use a preferred drugstore.
- Beware of online pharmacy scams.
- Check your insurer’s formulary lists.
- Check for tricky deductibles.
- Price out Medicare options.
- Clip coupons.
- Ask for help.
- ADA Issues Recs for Management of Diabetes in Primary Care (physiciansbriefing.com)Diagnosis and Management of Diabetes: Synopsis of the 2016 American Diabetes Association Standards of Medical Care in Diabetes (annals.org)
New recommendations have been developed for diabetes, focusing on areas of importance for primary care providers...a systematic review to revise or clarify recommendations for diabetes diagnosis and management for primary care providers, based on new evidence. The recommendations were rated and reviewed, and approved by the American Diabetes Association Board of Directors. Feedback from the larger clinical community was incorporated...The researchers summarized the ADA standards in eight areas of importance to primary care providers: diagnosis, glycemic targets, medical management, hypoglycemia, cardiovascular risk factor management, microvascular disease screening and management, and inpatient diabetes management. An individualized approach was recommended for each area, with self-monitoring emphasized as a key component of care...
- IBM’s Cancer Moonshot: Using Computer Science To Battle Tumors (forbes.com)
IBM is launching a cancer initiative...teamed up with Stanford University, San Francisco State University and two UC schools—San Francisco and Berkeley—to spearhead the application of a new discipline it calls "cellular engineering" to oncology research...The idea behind cellular engineering is to scrutinize cell "morphology," or the shape and structure of cells, to get to the root of what causes tumors. Understanding that process could lead to new ways of detecting and controlling the disease, says Simone Bianco, an Italian physicist...at the IBM Almaden Research Center...I asked Bianco to explain how IBM is lending its know-how to cancer research. Here is an edited excerpt from our conversation.
- What is cellular engineering and how can it be applied to cancer?
- Once you gather information about what’s happening on a cellular level in cancer, how can it be used?
- What will IBM’s academic partners bring to this effort?
- Are there other uses for cellular engineering beyond cancer?
- Pharma’s Big New Cancer Drug May Be an Old Red Dye
Rose Bengal (PV-10), a cheap industrial chemical that turns yarn and food bright red, has been used as a diagnostic staining agent for some time. Now, some scientists are looking at its potential to fight various forms of cancer...Provectus Biopharmaceuticals...is testing a reformulated version of the industrial dye on melanoma, the deadliest form of skin cancer...Rose Bengal’s potential against cancer was discovered by accident. The salt was first patented in 1882 as a wool dye and has been used for years as a diagnostic stain in tests for jaundice in newborns and to detect eye damage...In 1998, scientists....were looking for a safe photoreactive agent to use in an investigation of lasers against cancer. Rose Bengal fit the bill...As it turned out, the Rose Bengal solution appeared to work on its own to dissolve tumors when directed injectly (directly injected) into them...Provectus executives say the small development tab—along with relatively low manufacturing costs and easy handling requirements—could make PV-10 a less expensive new treatment...Provectus plans to put itself up for sale once its drug is approved...
- Another of AmerisourceBergen’s Pharmedium facilities cited by FDA (fiercepharmamanufacturing.com)Form FDA 483 Inspectional Observations (fda.gov)
Drug distributor AmerisourceBergen has pledged to set the bar high on quality at Pharmedium Healthcare Holdings, the compounding pharmacy company for which it laid out $2.6 billion. The challenges, however, keep mounting as FDA inspections continue to note issues at its manufacturing facilities...FDA posted another Form 483 for a Pharmedium compounding facility last week, this one at the company's headquarters in Lake Forest, IL. It followed an inspection in November that turned up half a dozen observations. In the past year, the FDA has issued Form 483 observations at Pharmedium facilities in Texas, Mississippi, New Jersey and Tennessee, as well as at its Illinois headquarters...inspectors noted...that the Chicago-area facility had released 16 lots of products that were out of spec for potency and purity. They also noted that results from stability testing were not used in determining expiration dates.
- AstraZeneca sells drug for $70 million as divestment drive continues (reuters.com)
AstraZeneca has sold rights to a non-core drug to a unit of Kyowa Hakko Kirin for an upfront payment of $70 million, a day after getting $500 million for two ageing heart medicines...The British-based drugmaker has been divesting peripheral products to help pay the bills as it invests in new medicines, while taking a profit hit from the loss of patent protection on a raft of former blockbusters...Such divestments, or "externalization" deals, contributed $1.1 billion to revenue last year and the company has said the figure is likely to be higher in 2016.
- Takeda aims to unlock $2B in Entyvio sales with multipronged DTC push (fiercepharmamarketing.com)
Takeda is going behind closed doors with its first DTC campaign for Entyvio (vedolizumab), a new biologic treatment for inflammatory bowel diseases. Bathroom doors, that is...The Japanese drugmaker uses a series of bathroom doors in its ad campaign, asking if it's "time for a different perspective." A...TV spot launched Feb. 7...has racked up $2.9 million on more than 2,000 airings...This condition is such an isolating condition and such a stressful condition. Patients are kept from doing the important things they want to be doing in their lives and as a result they miss out on a lot...In testing many different creative ideas with Crohn's and ulcerative colitis patients...the bathroom doors resonated well because patients noted it is a view they are all too familiar with...But some patients even took it to the next level in saying that the doors metaphorically represent that barrier between their disease and being able to do all of the things they want to do. Emotionally, we've touched upon the reality of their disease in a very obvious bathroom door representation, but also a representation of just barriers in general...Analysts have projected blockbuster-level sales for Entyvio by 2020...Marketing will be key to gaining share in a crowded IBD space where it's up against established...brands such as AbbVie's Humira, Amgen's Enbrel, and Johnson & Johnson's Remicade...