- Bayer’s Aleve and GSK’s Flonase top list of OTC TV ad spenders for 2015 (fiercepharmamarketing.com)
In a head-to-head prescription versus over-the-counter drug battle, prescription wins. At least when it comes to advertising...TV ad spending for the top 10 prescription drugs tallied $876.3 million, while over-the-counter spending among the top 10 brands totaled just $514 million for TV ads in 2015…Bayer's pain reliever Aleve topped the OTC list with almost $75 million in TV spending. Aleve also had the highest number of different TV spots as well with 23 creative executions running in 2015. GlaxoSmithKline's Flonase at No. 2 was no surprise after the drug went off patent and GSK went on an aggressive push to keep the brand thriving through the spring allergy season...The top 10 OTC list by estimated TV media spending by iSpot.tv for 2015 follows:
- Aleve (Bayer): $74.5 million
- Flonase (GlaxoSmithKline): $73.1 million
- Claritin (Merck): $58.4 million
- Advil (Pfizer): $58.3 million
- Nasacort (Sanofi): $50.3 million
- Alka-Seltzer (Bayer): $49 million
- Allegra (Sanofi): $43.8 million
- Tylenol (J&J): $37 million
- Nexium (Pfizer): $36.3 million
- Zyrtec (J&J): $33.1 million
- The biosimilar ploy that could save billions in health care costs (statnews.com)
As drug makers race to develop cheaper versions of complicated biologic medicines, some companies are pursuing a tactic that could prove a win for themselves, patients, and the health care system as a whole...They are running studies designed to convince doctors and insurers that patients can be easily switched from expensive biologics...to so-called biosimilars, which are almost identical variants. Their goal is to encourage these kinds of switches without waiting for the Food and Drug Administration to decide whether a particular biosimilar has the exact same clinical benefit as its expensive, brand-name counterpart...Standard FDA approval is good enough for physicians to write prescriptions, but only an "interchangeable" designation will enable pharmacists to substitute a biosimilar for a brand-name biologic without contacting doctors for permission first...more and more companies are eyeing switching studies as a shortcut to expanded market share...drug makers are pursuing this workaround that, as an upshot, could bring lower-cost medicines to more people...
- Cleveland Clinic recruits participants for Alzheimer drug trial (reviewjournal.com)221AD302 Phase 3 Study of Aducanumab (BIIB037) in Early Alzheimer's Disease (EMERGE) (clinicaltrials.gov)
The Cleveland Clinic Lou Ruvo Center for Brain Health is seeking participants with mild Alzheimer's disease to test a drug that has been shown to stop the progression of symptoms in some volunteers...about 250 facilities worldwide participating in a clinical trial called Emerge, which will examine the effects of experimental drug B11B037...The drug...has been shown to remove toxic protein buildup in the brain; researchers believe the buildup may be the cause of Alzheimer's...It has also been shown to stop the progression of symptoms...the Ruvo Center will enroll 10 participants, but more than 2,500 patients will be enrolled worldwide...Individuals participate in trial testing for free, but as with every new drug, there are potential health risks...Side affects of the drug have been shown to include headaches, dizziness, nausea, vomiting and constipation...For further information, call the Ruvo Center at 702-483-6000 or study coordinator Monica Guerra at 702-701-7893.
- Syphilis outbreak strikes Clark County (reviewjournal.com)
Reported syphilis cases in Clark County have skyrocketed 128 percent since 2012, with the number of early syphilis cases reported constituting an outbreak, according to the Southern Nevada Health District...The health district said in a news release Wednesday morning the increase has largely affected men and is asking health care providers who treat early syphilis patients to report cases to the district..."We are considering this an outbreak because disease rates in our community are continuing to climb and we see no plateau at this point," health district Chief Health Officer Dr. Joe Iser said…"Our goal is to make people aware of the problem, encourage health care providers — especially those who treat men — to report cases to us so we can begin notifying partners, get them into treatment, and stop the spread,"...
- Those PBM moves pharma loves to hate? They work, CVS says–so get used to them (fiercepharma.com)
Drugmakers may not be too keen on the formulary management techniques and exclusive deals pharmacy benefits managers have been using to combat rising drug prices. But new numbers from a PBM giant suggest they work--and that pharma better get used to them...CVS Health announced that its prescription drug trend--a metric for growth in prescription spending--had dropped to 5% in 2015, down from an all-time high of 11.8% in 2014. And with many of the usual drivers staying consistent over both periods--including brands and price inflation among generic and specialty drugs—the company is chalking up the drop as proof its tactics are doing their job...CVS' "proactive pharmacy management strategies were successful in mitigating the impact of rising drug costs in today's highly volatile prescription drug market,"..."[W]e work to help patients avoid unnecessarily expensive medications as part of our focus on making prescriptions affordable."..CVS and its main rival, Express Scripts, have both been actively pruning their preferred formularies over the past few years…Unsurprisingly, pharma hasn't taken too kindly to the moves, but some companies have been more accepting of the current climate than others...GlaxoSmithKline and Novartis, to name a couple, have been working to avoid payers' wrath...
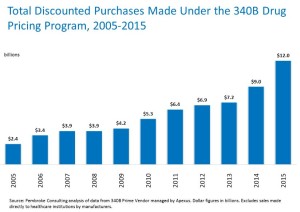
- 340B Purchases Hit $12 Billion in 2015—and Almost Half of the Hospital Market (drugchannels.net)
According to new data...discounted purchases made under the 340B Drug Pricing Program hit $12 billion in 2015. That’s a whopping 67% higher than the 2013 figure. I estimate that the undiscounted value of these purchases exceeds $17 billion...Most 340B purchases are made by hospitals. My...number-crunching below reveals that hospitals now receive 340B discounts on more than 44% of their drug purchases...two years ago, the 340B program is taking over the hospital market...How much of this money goes to uninsured and needy patients? No one knows, and the hospitals aren’t saying much...340B purchases have been growing much, much more quickly than have hospitals’ total drug purchases. From 2005 to 2015, total hospital drug purchases grew by 31%, compared with the 400%+ growth in the total 340B purchases...340B has infiltrated almost half of the hospital market...The 340B program is highly controversial, partly because the 340B legislation does not specify or restrict how covered entities should utilize funds generated by the program. Hence, it’s troubling to see that uncompensated care as a percentage of hospitals’ total expenses has remained at about 6% for many years, despite booming 340B purchases...Defenders of the 340B status quo argue that covered entities should be able to use discounts to reduce their cost of operations, without any transparency or accountability...The Office of Inspector General has documented how 340B-eligible hospital outpatient departments earn tremendous profits from the Medicare Part B program. Gross profit margins are about 60% compared with 3% to 4% for a non-340B outpatient program...
- Management Tools for Controlling Specialty Drug Costs (specialtypharmacytimes.com)
The increasing utilization and costs of specialty drugs will have a substantial impact on overall health care costs during the next decade...Pharmacy benefit manager tools that have successfully controlled costs in the traditional small-molecule drug categories will be critical to manage the increasing costs and requirements of the specialty drug category…
Financial Impact of Specialty Drugs: Current and Projected...by 2020, 9 of the 10 best-selling drugs (by revenue) will be specialty drugs...spending could reach $400 billion, or 9.1% of national health spending...Specialty benefit design and management tools employed by PBMs will also play a significant role in controlling specialty drug spending, as well as ensuring optimal patient care and support.
Specialty Benefit Design and Management...Specialty drugs frequently have unique shipping and storage needs, as a result specialty pharmacies are better equipped to procure, store, and dispense these treatments than traditional retail pharmacies. Furthermore, pharmacists and personnel at specialty pharmacies provide patient education and clinical support beyond the capabilities of a retail pharmacy...The tools in the PBM arsenal are critical to ensure appropriate care for patients needing specialty drugs while managing the often extraordinary costs.
Comprehensive management approaches that monitor and balance patient care outcomes and costs will help PBMs ensure that new, innovative medications are readily available and affordable to the patients who need them most.
- How should FDA revamp its off-label marketing rules? An expert panel backs big changes (fiercepharma.com)
After a series of court decisions put its regulatory approach in question, the FDA promised new guidance on off-label drug promotions. That was more than a year ago. Now, a group of health policy wonks and legal experts has come out with some suggestions...The recommendations from the Duke-Margolis Center for Health Policy include one big change that might simplify the free-speech debate over off-label marketing. The agency could revamp its approach to official labeling to include clinical data on off-label uses, graded for evidence quality, and even post-marketing data generated through the agency's own Sentinel surveillance system…Changing official FDA labeling...the Duke-Margolis Center suggests using pilot programs...One might use the Sentinel system. Another might allow provisional off-label use claims based on evidence that's not enough for a new indication, contingent on Sentinel keeping watch on the new use. Overall, the idea would be to develop a tiered labeling system, with the strongest supporting data on the highest tier, and lesser quality evidence farther down...more radical suggestion? Setting up a third-party data-review organization with the FDA's involvement...[That] would assess new off-label promotional material and give it a quality-control score based on the quality of evidence backing it up…The FDA clearly wants to roll out its own guidance, rather than letting the courts decide. In December, the agency settled an off-label marketing dust-up with Pacira over its Exparel marketing, and it's on the verge of a deal in Amarin's off-label lawsuit involving its cardiovascular med Vascepa.
- Some doctors slow to sign up for electronic death certificate filing (reviewjournal.com)
Nevada's Office of Vital Statistics issued a call this month for relevant physicians to sign up as quickly as possible for a system that allows doctors to fill in required information for death certificates electronically. That system became mandatory statewide Jan. 1...Some doctors did not sign up after the state began notifying them of the change in fall because they have had issues registering or have outright refused to participate, vital records program officer Jason Lewis said...Karen Massey, chief administrative officer at Northern Nevada Emergency Physicians, said physicians have struggled with some requirements of the system, including its incompatibility with Windows 10 and Apple computers. The changeover has been time-consuming...I'm dismayed when it sounds like physicians haven't been responsive about it...Physicians have raised several concerns and have been working with the state to get them resolved...
- Should Pharmacists be called Doctors? – PLR 4 (pharmacyliferadio.com)
Today we touch on a TOUCHY subject: should pharmacists call themselves doctors?...Lots of people have opinions on this, and there is a lack of facts to cut thru the fog of confusion...(podcast 20:36) Today we:
- review the major reasons why pharmacists SHOULD call themselves doctors
- review the reasons why pharmacists SHOULDN’T call themselves doctors
- what we think
- and what you should do to figure out for yourself