- Hollywood Presbyterian hack signals more ransomware attacks to come (healthcareitnews.com)Hollywood Presbyterian Medical Center Pays Hackers $17K Ransom (nbcnews.com)
As hackers hold Hollywood Presbyterian Medical Center’s data and demand $3.4 million Bitcoin to give it back, experts say the "hostage situation" likely signals more ransomware attacks to come...There is no style to this attack...it was likely messaging-based, whether a malicious link in an email or perpetrated via a social network and, basically, an employee fell for it...Such attacks are particularly alluring to cybercriminals...because they are reasonably easy to pull off and have a big impact...the cybercriminals are demanding the hospital pay a $3.4 million ransom if they want their data back...In the meantime, executives declared the hospital in a state of emergency and employees are reverting to paper and faxes to communicate..."This incident really sheds light how weak the core of many providers' internal infrastructure is...It's very common for hospitals to have a large number of outdated and vulnerable systems on the network...
- With drug costs rising, it’s time for pharma companies to open their books (statnews.com) Budget of the United States Government, Fiscal Year 2017 (whitehouse.gov)
For years, drug makers have argued that the rising prices of prescription medicines reflect, in part, the rising costs of discovery and development...Buried inside the White House budget proposal released last week is language (on page 62) that would require drug makers to publicly disclose various data, including research and development costs. The administration hopes to use the information as part of a plan to negotiate lower prices for the Medicare drug program, known as Part D...Obama joins a growing list of lawmakers seeking transparency in order to understand drug pricing...From California to Massachusetts, state legislators have proposed bills that would force drug companies to open their books so the public can see how much is spent to develop, manufacture, and market new medicines. Just last month, New York Governor Andrew Cuomo included a disclosure demand in his own state budget proposal...Whether these efforts will succeed is unclear. Drug makers have been lobbying aggressively to defeat the bills and, indeed, in many states the legislation has stalled.
- Indonesia clears decks for foreign API manufacturers
The administration of Indonesian President Joko Widodo has cleared the decks for foreign firms to own 100% of active pharmaceutical ingredient operations....Indonesia's three largest pharmaceutical companies are the biggest importers of raw materials and the change is expected to help them meet soaring demand as the country widens access to medical insurance...The change in foreign investment restrictions could help reduce the reliance on importing raw materials used to make drugs...domestic industry needs investments of more than $1 billion to cut dependence on imports of APIs and related raw materials...
- Solving the Puzzle of High Cost Specialty Drugs (specialtypharmacytimes.com)
Jon Hamrick, executive vice president, Biotech and Specialty Services at Therigy LLC, discusses the implications of rising drug costs on specialty pharmacy.
- Retail Pharmacy Clinics: Top Players and the Coming 2016 Pause (drugchannels.net)
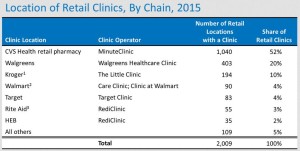
As low-cost generics come to dominate retail dispensing activity, clinics have emerged as a way for drugstores and other retail pharmacy outlets to diversify into non-dispensing revenues...There are now about 2,000 retail clinics. Below, I examine the chains with the biggest market share: CVS Health, HEB, Kroger, Rite Aid, Target, Walgreens, and Walmart...In 2015, the number of clinics grew slowly. In 2016, I project another year of slow growth as the big players retrench...
- CVS Health’s MinuteClinic remains the largest and fastest-growing retail clinic business, now operating more than half of all retail clinics.
- Walgreens is the second-largest retail clinic operator, with more than 400 Healthcare Clinics (formerly Take Care clinics). The number of clinics in Walgreens retail stores declined in 2015.
- Kroger has emerged as the third-largest retail clinic operator with its Little Clinic business, which operates inside nearly 200 Kroger, Dillon’s, Fry's, JayC, and King Soopers stores.
- Rite Aid changed its clinic strategy with the 2014 acquisition of RediClinic. As of January 2016, Rite Aid operated 41 RediClinics....a further 35 retail locations.. in HEB grocery stores...At 14 Rite Aid locations, the company leases space to other clinic operators...
- Walmart launched its own Care Clinics in 2014. There are now 17 Care Clinics in Walmart stores...Walmart also leases space to independently owned and operated Clinic at Walmart operators...operate inside 73 Walmart stores.
- Recipharm commits $45M to counteract fake drugs (fiercecro.com)
Contract drugmaker Recipharm is promising to spend $45 million (€40 million) over the next three years to improve drug traceability in an effort to comply with new regulations and combat the rise of fake medicines...The money will go toward new coding and serialization technologies...an investment that will allow the company to better tag drug products with identifiable markers that can help prevent counterfeiting. The move comes on the heels of the EU Falsified Medicines Directive, which will require new measures in drug manufacturing starting in 2019, according to the company...Recipharm already implements track-and-trace procedures in Asia and is now expanding its efforts in Europe to help clients on the continent prepare for the new standards...
- Zika update: Vaccine race swells, PaxVax CEO on how to stop ‘chasing epidemics’
As the Zika virus continues to spread, more biotechs are announcing their Zika vaccine programs. Meriden,.. Protein Sciences,..GeoVax Labs and.. PaxVax are the latest...Getting caught off-guard by epidemics like this has happened time and time again. And "chasing" outbreaks instead of anticipating them rarely results in a vaccine being developed in time. Witness the most recent Ebola epidemic: Merck's experimental vaccine, the furthest along in a crowded field, won't be submitted for regulatory approval until 2017, more than two years after the outbreak started...we had known about Ebola for decades...Companies got a head start in 2014 from partly developed candidates that had been shelved away. It is not so with Zika. "Almost everyone is pretty much starting from scratch...To avoid this and have programs in place before an outbreak hits...governments and nongovernmental organizations...should create economic incentives for companies to make vaccines for neglected diseases like Zika...the FDA's priority review voucher system, in which a company developing a vaccine for a neglected tropical disease receives a transferable voucher for expedited FDA review. Malaria and dengue have been on the list of neglected diseases for years, but Zika is not yet on the list
- Pharma Nerds Aren’t Pharma Bros (forbes.com)
Martin "Pharma Bro" Shkreli’s testimony – or lack thereof – at a House Oversight Committee hearing justifiably angered many people...Overnight, he became the embodiment of a pharmaceutical industry routinely accused of putting profit-seeking before patient health...But policymakers shouldn’t jump to conclusions...There are many important differences between the "Pharma Nerds" developing innovative life-saving drugs – including Gilead Sciences’ Sovaldi – and the "Pharma Bros" exploiting loopholes to profit from generics like Daraprim...What’s the difference between daraprim and Sovaldi?...quite a bit...Sometimes, short term monopolies are good for markets and society..Patents encourage innovation in sectors where the upfront costs of developing a product are very high. Monopoly patents are designed to ensure that innovators that they can recoup these costs and earn a healthy profit before generic competitors enter the market...Monopoly patents – with all the implied pricing power – are necessary for producing innovative drugs...But generic drugs are not "innovative," although they are critical in maintaining a healthy and competitive pharmaceutical market…Daraprim is a generic, and generics shouldn’t – with some very narrow exceptions – benefit from the same monopoly protections that innovator drugs do...patents will continue to work well for most products, and governments should continue to prioritize strong IP to promote medical innovation...Lowering prices by fiat will just tell investors to take their dollars elsewhere.
- Genes, bugs and radiation: WHO backs new weapons in Zika fight (reuters.com)Brazil reports more cases of microcephaly under investigation (reuters.com)FDA recommends ban on blood collections from Zika-affected areas (reuters.com)Zika vaccine shows promise in mice, lifting maker Inovio (reuters.com)
Countries battling the Zika virus should consider new ways to curb disease-carrying mosquitoes, including testing the release of genetically modified insects and bacteria that stop their eggs hatching...Given the magnitude of the Zika crisis, WHO encourages affected countries and their partners to boost the use of both old and new approaches to mosquito control as the most immediate line of defence...WHO also highlighted the potential of releasing sterile irradiated male mosquitoes, a technique that has been developed at the United Nations’ International Atomic Energy Agency...Fighting the infection at source by eliminating the Aedes aegypti mosquitoes responsible for transmission is moving up the public health agenda, especially as the same insects also spread dengue, chikungunya and yellow fever...An alternative approach uses Wolbachia bacteria, which do not infect humans but cause the eggs of females that mate with infected males to fail to hatch. Mosquitoes carrying Wolbachia have been shown to reduce mosquitoes' ability to transmit dengue...
- What’s China doing to be innovative in pharma, biotech? (medcitynews.com)
The question of whether China can innovate in science and technology is one that is often debated and remarked upon. Having read so many (Western) opinions about China’s ability to innovate, I was interested in what I could learn from talking with leaders at R&D-intensive organizations in China. How do they characterize their own innovation abilities?...Conventional wisdom here in the U.S. has long been that China doesn’t innovate. After all, a lot of products in China are very familiar-looking (and I’m not even talking about blatant intellectual property violations – just good old-fashioned "me too" competition)...No one innovates for the sake of it. We innovate when adequate solutions to our problems don’t already exist, and when someone has already solved a similar problem, we borrow...the test of whether China can innovate can’t be answered from what it has achieved while still following modern economies...but rather by what it does when it is in the leading group. As China joins the global community, the time is rapidly running out when it can easily source innovations from outside – and will have to prove that it can innovate from within...