- Angered by Walgreens deal, Express Scripts blocks access to Valeant’s Glumetza (fiercepharma.com)
PBM giant Express Scripts has long been a leader in the fight to tamp down drug prices, and now, it's using its formulary power to freeze out a diabetes drug from controversial Valeant...The company will exclude the Canadian drugmaker's Glumetza--an extended-release, brand-name version of generic med metformin--as soon as copycats become available on Feb. 1...Valeant hiked the price of Glumetza by more than 800% in 2015, Express Scripts says; as of July 31, the drug's list price stood at $10,020 for 90 tablets, up from $896 in January 2013..."By excluding Glumetza from these formularies, Express Scripts clients are ensuring that their patients be dispensed the more affordable generic formulation of metformin,"...noting that "branded Glumetza will not be allowed to process" under any circumstances.
- Washington’s Prescription For Cheaper, And Unregulated, Medicines (forbes.com)
In 2011, in an effort to bring a lower-cost drug to the market, the Food and Drug Administration was forced to accept the widespread compounding of a specialty drugs...The agency’s submission involved an old, and previously cheap, generic medicine that had secured some renewed exclusivity, and was being sold as a higher-priced specialty drug. But the FDA’s move deliberately undermined the agency’s own approval requirements...It sent an unambiguous message that FDA wouldn’t try to enforce its already weakened authority over compounding–so long as the price of the knock-off drug was right...The compounding of drugs grew precipitously over the next few years...the passage of the 2013 Drug Quality and Security Act...closed loopholes that allowed compounding outfits to operate beyond the bounds envisioned when the local practice of pharmacy was carved out from typical FDA oversight...some are seeking to co-opt some key provisions in that law as a way to once again expand compounding well beyond the practices’ historical concept. Once again, these efforts are being insincerely waged in the name of increasing drug competition and lowering costs…The co-opting of the practice of pharmacy compounding in this manner also puts the entire practice of pharmacy at risk. Compounding remains an important pharmacy activity...But each time outfits try to exploit the practice simply as a way to evade traditional regulation of drug manufacturers; they create risks and bad outcomes that ultimately lead to new restrictions. That was the legacy of the 2013 passage of the DQSA. Now some who instigated that law want to do it all over again.
- Redwood City opens food pharmacy for low-income diabetes patients (ktvu.com)
A very different kind of pharmacy opened in Redwood City on Wednesday. It's called a food pharmacy and is designed to encourage low-income people suffering with diabetes, to eat a healthy diet...It's the first of its kind ever to open in California...With a doctor's prescription, low-income diabetes patients can get food at this special pantry inside the Samaritan House Health Clinic, for free...The food is donated by the Second Harvest Food Bank, which says diabetes and other diseases run rampant among low-income people who often can't afford to eat healthier, or don't know how..."At Second Harvest, our clients have told us that one out of every three adults that we serve are suffering from diabetes. That's more than three times the national average. So it is a big problem among low-income communities," said Kathy Jackson, director of the food bank...The food pharmacy officially opened on Wednesday as a pilot program expecting to provide 100 diabetes patients with a ticket to healthier eating habits...Doctors say the cost of food is a lot less than the cost of treating the effects of a worsening disease.
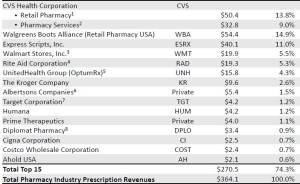
- The Top 15 Pharmacies of 2015 (drugchannels.net)Largest U.S. Pharmacies Ranked by Total Prescription Revenues, 2015 (pembrokeconsulting.com)
Next week, Drug Channels Institute will release our updated, revised, and expanded 2016 Economic Report on Retail, Mail, and Specialty Pharmacies...provides a sneak peek at the largest pharmacies, ranked by total U.S. prescription dispensing revenues for calendar year 2015…We estimate that total revenues of retail, mail, and specialty pharmacies reached $364.1 billion in 2015, up 12.1% from 2014. The top tier of dispensing pharmacies—CVS Health, Walgreens Boots Alliance, Express Scripts, Walmart, Rite Aid, and UnitedHealth Group’s OptumRx—accounted for about 64% of U.S. prescription dispensing revenues in 2015...many of the largest pharmacies are now central-fill, mail and specialty pharmacies operated by such PBMs and payers as Express Scripts, Caremark, and UnitedHealth. This reflects the growing role of specialty drugs in the pharmacy industry. We estimate that specialty drugs account for 35% or more of revenues at these pharmacies.
- China fines 5 companies for monopoly on drug sales (fiercepharmaasia.com)
China's National Development and Reform Commission has imposed fines totaling $608,000 on 5 mainland drugmakers and distributors for price-fixing of allopurinol, which is considered an essential medicine in China and is widely used to treat gout and hyperuricemia...The NDRC fined drugmakers Chongqing Qingyang, Jiangsu Shimaotianjie, and Shanghai Xinyi, which have been the only three domestic producers of the tablets since 2014, according to the reports. They also fined distributors Chongqing Datong and Shangqiu Huajie..."Five companies have been fined for colluding to manipulate pricing and dividing the market of a common tablet," said...deputy inspector of the Price Supervision and Anti-Monopoly Bureau..."Even though the fine is not the largest issued since the NDRC began bringing actions against pricing collusion, it is significant."
- We Can Beat Zika And Malaria–If The FDA Allows (forbes.com)The Emerging Zika PandemicEnhancing Preparedness (jama.jamanetwork.com)
Zika virus infection, the scary new disease for which there is no vaccine or treatment, is “spreading explosively” from Africa and Southeast Asia...The United States and 20 other countries...have reported cases of the virus since Brazil reported the first cases of local transmission last May. Delivered by varieties of mosquitoes...it has boosted interest in mosquito-borne diseases...What’s needed is...modern genetic engineering techniques to more effectively prevent mosquitoes from delivering the viruses and parasites that cause disease….The FDA has long delayed the approval of a November 2011 application for a field trial to test a new biological control agent for the mosquito species Aedes aegypti. Although that field trial is concerned specifically with dengue fever, A. aegypti...also transmits Zika...Oxitec has created a new way to control Aedes aegypti. Male mosquitoes are bred in the laboratory with a specific genetic mutation that, in the absence of a certain chemical, causes their offspring to die before reaching maturity...This safe and effective control technique has been approved in Brazil and open field trials of these mosquitoes have been conducted in Brazil, the Cayman Islands, Panama and Malaysia...Eight months have passed since FDA promised last May to publish for public comment a routine environmental assessment of the Oxitec field trial in Florida. Only after FDA reviews the comments will FDA consider whether to grant approval. This delay is unnecessary and unconscionable.
- Q&A: Data sharing bid ‘a remarkable turning point’ for drug research (statnews.com)
In an extraordinary move, the International Committee of Medical Journal Editors last week issued a proposal to require researchers to share their clinical trial data as a condition for publication. And the researchers would also have to submit plans for how their data can be shared. The journal editors, who represent such periodicals as The New England Journal of Medicine and the Annals of Internal Medicine, believe data sharing "will help to fulfill our moral obligation to study participants, and we believe it will benefit patients, investigators, sponsors, and society." The move comes after protracted tussling over access due to safety scandals that revealed trial data for some products was never fully published or disclosed. We spoke with Harlan Krumholz, who runs the Yale Open Data Access project and works with companies to publish trial data, about the implications.
- Why does this matter?
- So how big a step is it for these journal editors to issue such a statement?
- Why do you think it took this long to take this step?
- Of course, a researcher may not follow through and share data even after submitting a plan, which raises a question about enforcing the requirement.
- What do you make, though, of the editors at The New England Journal of Medicine? In a recent editorial, they expressed reservations and set off a ruckus by saying that some researchers worry about ‘research parasites.’
- How much push back do you anticipate from industry?
- Walgreens Boots Alliance outlines 3 growth initiatives at shareholders meeting (drugstorenews.com)
Walgreens Boots Alliance this year moved its annual shareholder's meeting...to...Manhattan, approving three proposals made by the board, reviewing the company's recent events and looking ahead to the future...all of which executives said would help deliver a banner year for Walgreens in 2016.
- Rite Aid acquisition - The addition of rite aid to our network offers the potential for significant cost savings and purchasing efficiencies in addition to opportunities in terms of important market penetration...
- Valeant Pharmaceuticals partnership - ...offer its branded dermatology and ophthalmology products at a reduced cost to the consumer...This partnership...is a demonstration of how partnerships can create value while reducing costs for the system as a whole...
- Boots’ beauty proposition - transforming Walgreens into must-visit destination centers for WBA's owned beauty brands...as part of the company's initiative "Beauty 2000"...
- Payers see price leverage with entry of Merck hepatitis C drug (reuters.com)
...pharmacy benefit managers expect the launch of Merck & Co Inc's new hepatitis C pill to improve their leverage in price negotiations with drugmakers...The Food and Drug Administration...approved Merck's Zepatier (elbasvir and grazoprevir) for treatment of patients infected with the most common form of the liver-destroying virus, genotype 1, as well as the less common genotype 4...The list price for the new drug is $54,600 for a 12-week regimen - compared with $94,500 for Gilead Sciences Inc's Harvoni. A multi-pill regimen, Viekira Pak, from AbbVie Inc has a list price near $83,000..."We look forward to working with Merck," Express Scripts said..."Having multiple, clinically effective options allows us to again leverage competition and make medicine more affordable for our clients while ensuring appropriate patient access."..."Given Merck's interest in participating in such a large market, we model and fully expect increased price competition and we also view Merck's list price as a rational way to stay out of the drug pricing spotlight,"...
- Pharmacy Week in Review: January 29, 2016 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.