- Here’s what you need to know about Zika virus (cnbc.com)Zika Virus (cdc.gov)U.S. researchers call for WHO to take rapid action on Zika (reuters.com)Four Zika cases found in New York among returning travelers (reuters.com)Zika virus: US scientists say vaccine '10 years away' (bbc.com)
A new mosquito-borne virus is fast spreading across the Americas and the Caribbean, with cases reported among travelers returning to the U.S...Zika virus outbreaks are proliferating across multiple countries and territories in the region, such as Puerto Rico, Mexico, Brazil, Panama and Venezuela. They have been associated with increased reports of birth defects, including babies with very small heads, and of paralysis in adults...There is no vaccine or specific drug to treat the virus.
- How is Zika transmitted?
- Birth defects, paralysis?
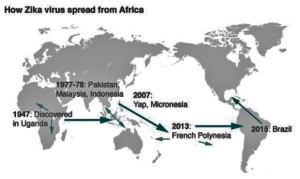
- How the outbreak started
- Could it reach the US?
- Prevention
- Ed Markey blocks vote on FDA chief over opiate approvals (bostonglobe.com)Three senators now holding up FDA nominee — for three different reasons (washingtonpost.com)
Senator Edward Markey is using Senate rules to block the nomination of a proposed new leader of the Food and Drug Administration in an attempt to force the agency to rescind its approval of a prescription opioid for children and change its regulatory practices...Using parliamentary procedures, the Massachusetts Democrat has put a "hold" on the nomination of Dr. Robert Califf, which prevents the Senate taking a confirmation vote...Markey, who has made the growing wave of opioid addiction one of his legislative priorities, is demanding that the FDA agree to reverse its 2015 decision allowing the pediatric use of the prescription painkiller OxyContin. He also wants the agency to commit to convening expert advisory panels to provide advice whenever considering the approval of an opioid drug, and to ensure that the risks of drug addiction and abuse are taken into account whenever the agency considers approving a prescription opioid...
- Deficiencies Found at Theranos Lab (wsj.com)
Federal inspectors will soon release details on problems at blood-testing facility...U.S. health inspectors have found serious deficiencies at Theranos Inc.’s laboratory in Northern California...The problems were found during an inspection by the Centers for Medicare and Medicaid Services, the chief federal regulator of clinical labs, at the blood-testing company’s facility in Newark, Calif. Failing to fix the problems could put the Theranos lab at risk of suspension from the Medicare program...inspection results are expected to be publicly released soon...
- CMS final rule addresses decades-long Medicaid reimbursement issue (drugstorenews.com)
The end of what has become a decades long battle around fair reimbursement for Medicaid programs and pharmacy appears to be in sight...the Centers for Medicare & Medicaid Services issued the Covered Outpatient Drugs final rule with comment that addresses key areas of Medicaid drug reimbursement and changes made to the Medicaid Drug Rebate Program by the Affordable Care Act. According to a fact sheet published by CMS, this final rule assists states and the federal government in managing drug costs, establishes the long term framework for implementation of the Medicaid drug rebate program and creates a more fair reimbursement system for Medicaid programs and pharmacies...the final rule is designed to ensure that pharmacy reimbursement is aligned with the acquisition cost of drugs and that the states pay an appropriate professional dispensing fee. The final rule:
- Creates an exception to the FUL calculation, which allows for the use of a higher multiplier than 175% to calculate the FUL based on acquisition costs for certain multiple source drugs;
- Establishes actual acquisition cost as the basis by which states should determine their ingredient cost reimbursement so payments are based on a more accurate estimate of the prices available in the marketplace, while still ensuring sufficient beneficiary access;
- Implements the use of the term professional dispensing fee to ensure that the dispensing fee paid to pharmacies reflect the cost of the pharmacist’s professional services and cost to dispense the drug product to a Medicaid beneficiary;
- Clarifies that states are required to evaluate the sufficiency of both the ingredient cost reimbursement and the professional dispensing fee reimbursement when proposing changes to either of these components; and
- Requires states to specify in the Medicaid state plan that reimbursement methodology to pharmacies that purchase drugs through the Federal Supply Schedule and the 340B Drug Pricing Program is consistent with overall AAC requirements.
- Biosimilar drugs get boost as UK cost agency backs their use (reuters.com)
Cheaper "biosimilar" copies of expensive biotech drugs received a boost in Britain...when the country's health cost-effectiveness agency NICE (National Institute for Health and Care Excellence) said patients needing such medicines "should be started with the least expensive drug"...So-called biosimilars are gaining ground in Europe, which has been faster to adopt their use than the United States, offering savings to healthcare systems and threatening sales of companies making original products...The latest ruling by the National Institute for Health and Care Excellence is a further victory for the biosimilar lobby and may help two cheaper copies win market share from five other original brands...The potential for biosimilars to win business from pricey original brands is not only a focus for healthcare providers but also a growing concern for investors, worried about the impact on large drug company earnings...A growing number of biosimilar versions of top-selling biotech medicines are set to reach the market in the next few years, although the scale of their impact is unclear...
- The biggest implications of ICJME’s new clinical trial data sharing proposal (biopharmadive.com)Sharing Clinical Trial Data — A Proposal from the International Committee of Medical Journal Editors (nejm.org)Chasing transparency in the pharmaceutical industry: How much is enough? (biopharmadive.com)
Editors, who wrote an editorial discussing the issue in last week’s New England Journal of Medicine, “many funders around the world—foundations, government agencies, and industry—now mandate data sharing. “...Things have evolved a great deal since November 2014, when the National Institutes of Health proposed forcing pharma companies to put all data from clinical trials—successful and otherwise—online. This so-called “transparency initiative” was supported by more than 80,000 individuals and 500 organizations with a vested interest in clinical trial data transparency. Even earlier, in 2007, a U.S. law came into effect making failure to register clinical trials illegal.
- Fast-forward to January 2016
- ICJME: “Data-sharing guidelines are needed”
- The power of individual de-identified patient data
- No data-sharing plan, no publication
- Concerns about research parasites
- Dialing back the paranoia
- Novo aims to be ahead of pharma pack with Iran investment (tehrantimes.com)
Novo Nordisk, the world’s top insulin supplier, expects to be ahead of the pack now nuclear sanctions are lifted in Iran, thanks to a continuous presence and a pre-emptive investment plan...(Iran) is an attractive target for Big Pharma seeking to sell new medicines to its large and growing population, but going in at ground level and securing good relations with authorities will be key, industry executives say...Medicines were an exception in the nuclear sanctions imposed on Iran, thanks to exemptions for essential drugs and other humanitarian goods. Yet shipping drugs into the country was far from trouble-free, due to tight curbs on financial transactions and restrictions on technology reaching the Islamic republic...“With the sanctions being lifted, we can operate more freely,” Jakob Riis, Novo’s head of marketing told Reuters at the World Economic Forum in Davos, Switzerland...The Danish company has maintained a staff of around 130 in Iran through the sanctions era and it now plans to more than double that, adding 160 additional staff, following a decision in September to invest 70 million euros ($76 million) on a factory...
- Big Pharma’s bet on Big Data creates opportunities and risks (reuters.com)
Novartis wants every puff of its emphysema drug Onbrez to go into the cloud...The Swiss drugmaker has teamed up with U.S. technology firm Qualcomm to develop an internet-connected inhaler that can send information about how often it is used to remote computer servers known as the cloud...This kind of new medical technology is designed to allow patients to keep track of their drug usage on their smartphones or tablets and for their doctors to instantly access the data over the web to monitor their condition...It also creates a host of "Big Data" opportunities for the companies involved - with huge amounts of information about a medical condition and the efficacy of a drug or device being wirelessly transmitted to a database from potentially thousands, even millions, of patients...Wireless interfaces are a great benefit to certain patient groups...But...connectivity also means vulnerability...
- Feds tell judge Martin Shkreli can go to Washington to testify before Congress (statnews.com)
Martin Shkreli appears to be a big step closer to testifying before a congressional committee...US Department of Justice wrote a letter Sunday to a federal judge overseeing a securities fraud case against Shkreli to say “the government does not object to (Shkreli) traveling to Washington, D.C., to comply with the House Committee’s subpoena” to testify at a hearing on prescription drug pricing. As part of his $5 million bail order, Shkreli is prevented from traveling outside parts of New York City and nearby Long Island...The letter paves the way for Shkreli to appear before the House Oversight and Government Reform Committee, which wants to explore the rising cost of medicines. A hearing had been scheduled for Tuesday, but it has been postponed to Feb. 4 due to the recent blizzard.
- Global Fund rushes HIV drugs to Uganda amid shortage (reuters.com)
The Global Fund, a partnership that sends HIV drugs to poor countries, says it plans to send an advance supply of antiretroviral therapy to Uganda, after the East African country ran out three months before the end of last year...Health activists say the shortage, which began last September, hit about 240,000 patients on publicly funded treatment programs, forcing them to modify their treatment or stop altogether. Private-sector clinics were not affected...The government said a weak currency and insufficient foreign exchange had hampered its ability to finance drug imports...Some activists said they suspected runaway election spending was behind the shortfall, but officials denied the charge...The government needs to mobilize resources to fill the gaps and find a long-term solution...