- 2015: A banner year for personalized medicine (catalyst.phrma.org)
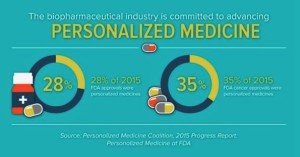
2015 was a record year for personalized medicine approvals, according to a new analysis from the Personalized Medicine Coalition. This news confirms the growing role of personalized medicine as an approach to treatment that can improve outcomes for patients and also create important efficiencies in the health care system. Personalized medicine is an emerging field of medicine that uses diagnostic tools to identify specific biological markers to help assess which medical treatments and procedures will be best for each patient. Personalized medicine also takes into account patients’ medical history, circumstances and values in developing targeted treatment and prevention plans...45 novel new drugs approved in 2015, the new analysis indicates that 28 percent of novel new drugs approved by the Food and Drug Administration...were personalized, or precision, medicines...Some of the personalized medicine highlights from 2015 include:
- Two new medicines for patients with different forms of non-small cell lung cancer;
- A new combination therapy for patients with cystic fibrosis;
- Two new medicines to help patients with a difficult-to-treat form of high cholesterol; and
- A new targeted therapy for melanoma.
- Licorice Coughing Liquid Recall, Presence of Morphine (infozine.com)Master Herbs, Inc. Issues Voluntary Nationwide Recall of All Lots of Licorice Coughing Liquid Due to the Presence of Morphine (fda.gov)
Master Herbs, Inc. (Ma Ying Long Pharmaceutical Group) is voluntarily recalling ALL LOTS of Licorice Coughing Liquid, cough syrup in 100 ml bottles to the consumer level. This product has been found to contain morphine, which is an opioid, and it is not declared on the label. Opioid is an ingredient of Compound Camphor. Compound Camphor is declared on the label of the product, but not its ingredients...The product is used for the temporary relief of cough due to cold, minor throat and bronchial irritations...identified by the Chinese Product Name: Licorice Coughing Liquid The product was distributed to Chinese grocery stores in various cities in California, New Jersey, Hawaii, Illinois, Ohio and Nevada.
- Levaquin users slap J&J with $800M RICO suit, claiming pharma giant hid serious side effects (fiercepharma.com)
Johnson & Johnson is facing yet another lawsuit over its antibiotic Levaquin from people who claim that the company hid serious side effects. The latest legal action comes a couple of months after an FDA panel flagged serious problems associated with the entire class of antibiotics and voted to change the meds' labels...Five plaintiffs allege in a new suit that J&J and its Janssen unit deliberately mislabeled and misbranded Levaquin (levofloxacin), playing down harmful side effects for its own financial gain. The plaintiffs sued J&J and others--including former FDA commissioner Margaret Hamburg--under the federal Racketeer Influenced and Corrupt Organizations act, a law typically used to prosecute organized crime. "These concerted efforts resulted in significant harm and/or death to consumers of Levaquin, including plaintiffs," according to the suit...The plaintiffs are seeking more than $120 million in compensatory damages and more than $750 million in punitive damages to drive home "the seriousness of their egregious conduct and to deter similar conduct in the future."...The latest suit marks another chapter in J&J's Levaquin saga. The company has encountered thousands of lawsuits over the past several years from plaintiffs claiming that it did not adequately warn patients about the drug's dangers.
- Sanofi layoffs expected as reorganization goes into effect (statnews.com)
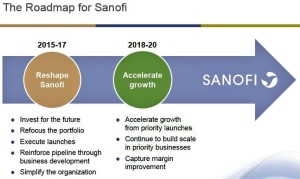
As part of a new "strategic roadmap" unveiled recently, Sanofi plans to announce layoffs that are being described as "sizable" and that will affect a broad swath of company operations in various locations. Some cuts are expected to be...in France, where the drug maker is headquartered, although extracting concessions from French labor unions is notoriously difficult. As a result, a disproportionate number of job losses may occur in the United States. A layoff notice, in any event, is expected to be filed shortly with state officials in New Jersey, where Sanofi maintains a large corporate campus...The drug maker...employs about 110,000 people worldwide, including roughly 17,000 people in the US...Sanofi encountered price discounting in the long-acting insulin market...This was thanks to its Lantus product, which generated about 18 percent of sales in the first half of last year, but faces pressure from payers seeking discounts...a follow-on product is not showing signs of generating needed replacement revenue and a biosimilar version of Lantus will become available at the end of this year. And a deal to sell the MannKind inhaled insulin product known as Afrezza was just ended due to terrible sales.
- Late Change in Insurance Plan Leaves 700 Without Health Care (kolotv.com)
Pam Perondi feels she's been blindsided by her health insurance provider, Prominence Health Plan..."It feels like bait and switch."...She says her open enrollment period was closing in mid-December. At the same time, Prominence was informing the Carson Medical Group, which includes the lion's share of the Carson medical community, it was cutting its compensation to them by a third....That was more than the medical group could absorb...Administrator Michael Lollich says the group held off for a week, hoping for reconsideration. Then on the 28th, they notified their patients they were no longer accepting Prominence insurance...The news came after the deadline for Pam Perondi and 700 others to make their decision...Carson Medical Group says it feels for its former patients...the group had no choice and the late notification left them with no time to work on a solution...Perondi isn't sure what she'll do now..."I'm just angry," she says, "and I feel sorry for all the others this has happened to."
- China ingredient maker’s plant found in ‘devastated state’ by authorities (fiercepharmamanufacturing.com)
China, which manufactures most of the ingredients for Western products, has a reputation for having some pretty questionable manufacturing operations. Czech inspectors got a firsthand look at one plant that they said posed "extreme risks" to consumers...According to two reports on the plant posted by the European Medicines Agency, authorities dropped by the Huanggang City facility of Hubei Hongyuan Pharmaceutical in October after the plant had been mentioned as an intermediate manufacturing site for the antibiotic metronidazole. The company acknowledged in its introduction that the site didn't follow EU GMP standards, the report says. During a walk-through, inspectors confirmed that...They said they found a plant in a devastated state, with "huge layers of dust and product" that indicated that neither the plant nor the equipment was being cleaned or that equipment was being maintained. It was noted the situation posed "an extreme risk of cross-contamination." Additionally, almost none of the products that they saw had been labeled and there was no batch manufacturing documentation...Western authorities have stepped up inspections of Chinese plants, both of those owned by Chinese companies and those owned by Western drugmakers, and found problems in both cases.
- U.K. competition watchdog targets pharma for ‘substantial fines’ (fiercepharma.com)
Look out, pharma: Britain's competition watchdog is coming for you....The U.K.'s Competition and Markets Authority is planning to levy "substantial fines" this year, marking a "big step up in the scale and impact" of its enforcement activity, Alex Chisholm, head of the agency, told the Financial Times. And one of those fines will likely be directed at a pharma company...The CMA has been probing drugmakers including GlaxoSmithKline and Pfizer for alleged wrongdoing. "In high-value markets with big players, they should face big fines,"...For Pfizer, though, a resolution could still be a ways off. In August, the CMA accused the drugmaker and partner Flynn Pharma of running afoul of U.K. and European laws by jacking up costs for their epilepsy drug...The pair charged "excessive and unfair prices"...the CMA said at the time, raising costs by as much as 2,600%. Regulators plan to hand down a decision in the case in about three months..."While businesses are generally free to set prices as they see fit, those that hold a dominant position have a special responsibility to ensure that their conduct does not impair genuine competition and that their prices are not excessive and unfair,"...
- Pharmapreneurs – the Pharmacist Entrepreneur – Pharmacy Podcast (pharmacypodcast.com)
Dr. Blair Thielemier interviews pharmacist & entrepreneur – Simone Sloan about what drives entrepreneurship, leadership, and transforming our pharmacy industry. (23:19 min)
- Medical publishers eye new ad viewability rules (mmm-online.com)
The Association of Medical Media plans to hold a meeting to establish an industrywide viewability standard...A group of medical publishers will attempt to develop a new industry standard for digital advertising viewability, a much debated topic that has vexed the broader advertising industry...Viewability—how long is long enough for a user to have viewed a digital ad successfully—has cropped up in recent years as one of the more challenging media issues, and one that needs to be addressed as the business of digital advertising matures. In healthcare, experts say, it's no different..."There's more pressure to account for dollars,"...The group's perspective is that professional medical content differs from other online advertisers..."It is distinguished from general web content in many ways, including in its scarcity and its value to the end reader...industry experts have been saying that drugmakers will likely put more emphasis on digital advertising as brand marketing becomes more targeted to certain patient populations and manufacturers lose direct access to physicians and other prescribers. That could expose more advertisers to the vagueness of viewability and highlight the need to find a solution that makes sense given the nuances of medical advertising, notably content aimed at healthcare providers.
- FDA slaps Pfizer’s Hospira unit for ‘misleading’ YouTube ad (fiercepharma.com)
In the first enforcement action from the FDA's marketing police this year, the Office of Prescription Drug Promotion put Hospira in the hot seat over a YouTube video for its sedative Precedex (dexmedetomidine)...The OPDP sent an untitled letter dated Jan. 14 to the Pfizer-owned company, charging the video "omits risks and material facts" about the drug. The agency also rebuked Hospira for publishing the promotional video without submitting it to the OPDP for review...The letter orders Hospira to "cease violating the FD&C Act, as described," and submit a written response before Jan. 29. The response should include a plan for "discontinuing use of such violative materials," the letter states.