- Bayer opens large China plant for traditional Chinese and Western OTC meds (fiercepharmamanufacturing.com)
Bayer boosted the reach of its international consumer health business when it picked up Chinese herbal and OTC special Dihon in 2014. Now it has boosted its production capacity with a new plant that will manufacture both Western OTC products and traditional Chinese medicines (TCM)...The German drugmaker last week opened the first phase of the 1.4 billion yuan ($213 million) plant in Majinpu in the Yunan province of China. At 111,534 square meters (1,200,542 square feet), it is Bayer's second largest over-the-counter products manufacturing site in the Asia-Pacific... first phase of construction has secured GMP approval for part of the TCM production line...additional work is ongoing that will allow the company to start manufacturing Bayer's key TCM product, Dan E Fu Kang, which is marketed as a gynecological medicine for women's health indications including dysmenorrhea...
- New Northern Nevada HOPES Facility to Open Soon in Downtown Reno (ktvn.com)
“Right now we're kinda cramped, we're in a small trailer, we can't fit all of our staff yet. We actually have people who are waiting to come on board,” says Dr. Chris McDonnell...The 38,000-square foot building at 5th and Ralston will allow Northern Nevada HOPES to go from serving 600 patients to 10,000. One highlight is the dedicated pediatric pod with 12 exam rooms, two treatment rooms and a unique "care team center."..“Four care teams that will be in here and that will include a provider, a behavior health specialist, an RN, a medical assistant as well as a case manager,” says Sharon Chamberlain, CEO Northern Nevada HOPES.
- BioMarin CEO on drug pricing, approval (video.cnbc.com)
BioMarin CEO Jean-Jacques Bienaime, discusses drug pricing, the sentiment of investors in biotech and his expectations for the company's Duchenne muscular dystrophy drug.
- Will GSK’s no-speaker-fee system work? Execs say yes; critics, not so much (fiercepharmamarketing.com)
GlaxoSmithKline has taken some bold steps to polish its image, tarnished by a Chinese bribery scandal and $3 billion settlement with the U.S. government. But naysayers blame its first big moves--nixing sales-rep quotas and pegging bonuses to "softer" measures instead--for disappointing roll-outs for several new meds...Now that it's dropping the time-tested tactic of paying doctors to promote its meds, critics are piling on there, too. But the U.K.-based drugmaker is sticking to its guns, recruiting its own doctors and other experts to tout its meds...Society now sees pharma-paid doctors as "hired guns,"...The only way to avoid that rap is for drugmakers to use their own employees instead...Since GSK first announced its no-payment policy--which went into effect Jan. 1--other Big Pharmas have addressed the question. Will they do the same? Several companies are on record with an unequivocal "No," saying that their doctor-speakers are important to their promotional efforts...Time will tell whether GSK's new policy will boost its credibility without cutting into revenue. More time will tell whether a buffed-up image would translate into bigger sales.
- Drug Diversion in the 340B Program (pharmacytimes.com)
Section 340B of the Public Health Service Act requires drug manufacturers participating in the Medicaid Drug Rebate Program to sign an agreement...This...limits the price that manufacturers may charge certain covered entities for covered outpatient drugs...Drug diversion in the program is defined as a 340B drug being provided to an individual who is not an eligible outpatient of that entity and/or dispensed in an area of a larger facility that is not eligible (eg, an inpatient service or a non-covered clinic)...in 2013 that 94 audits were underway, which included 700 outpatient facilities and 1930 contract pharmacies. During these audits, drug diversion, duplicate discounts, and ineligible sites/providers were the common areas of noncompliance... Noncompliance to 340B program impacts patients’ bottom line because the more diversion that occurs, the more drug manufacturers increase prices for both public and private insurers, leading to an increase in rates and charges to patients. If HRSA were able to enforce 340B regulations and audit all hospitals on a continual basis, there would be fewer cases surrounding duplicate discounting, drug diversion, and ineligible site/providers...The 340B Program is in desperate need of stronger controls and more audits. Through proactive monitoring of drug inventory and dispensing, 340B drug diversion would decrease, leading to a decrease in drug spending.
- Nevada Health Link records 77,411 enrollees, topping previous enrollment period (reviewjournal.com)
Two days ahead of a key signup deadline, the federal government released new enrollment numbers for Nevada's health insurance exchange...Nevada Health Link had 77,411 enrollees on Wednesday, the Department of Health and Human Services reported...That means the exchange has already bested the 72,000 enrollees that bought coverage through the marketplace in all of 2015's open-enrollment period...The department said the latest numbers include existing customers who were automatically re-enrolled, though officials didn't break down how many enrollees were new versus returning...Consumers who haven't bought coverage by Jan. 31 face a federal tax of 2.5 percent of household income, or $695 per adult and $347.50 per child up to a household maximum of $2,085 — whichever is higher.
- Nevada State Board of Pharmacy, Newsletter – January (bop.nv.gov)
- Flash News! -Governor Brian Sandoval appointed Darla Zarley to the Board...reappointed Kirk Wentworth and Leo Basch
- Senate Bill 459 - Effective October 1, 2015, this bill addresses three primary topics: (1) the mandatory use of the Nevada PMP ( Prescription Monitoring Program) by prescribers, (2) the Good Samaritan
Drug Overdose Act (GSDOA), and (3) next-day reporting to the PMP database. - Discontinue Use of Chen Shwezin Sterile Drug Products, FDA Warns
- Seven Persistent Safety Gaffes in Community/Ambulatory Settings That Need to Be Resolved! (final article of a three-part series): 6. Compounded Pain Creams: High Profit Margin and Danger; 7. Clear Care: Still Causing Severe Eye Injuries Five Years Later
- Risk of Dose Confusion and Medication Errors With Avycaz, FDA Cautions
- US Compounding, Inc, Recalls All Lots of Sterile Compounded Products
- FDA Investigates the Risks of Using PainMedicine Tramadol in Young Patients
- Decreased Potency Reported in Drugs Stored in Becton-Dickinson Syringes
- MediStat Pharmacy Issues Recall of Sterile Drug Products
- DEA Number for Residents Working at a Hospital
- 2015 Pharmacist Renewal Update! - Nevada had another successful year with pharmacist renewals.
- The Changing Landscape for Specialty Pharmacy Patients (specialtypharmacytimes.com)
Marc O'Connor, chief operating officer of Curant Health, discusses potential changes patients of specialty pharmacies may experience in the near future.
- State measures to slash drug prices face big hurdles (statnews.com)
When shopping for a good deal, people usually want the same bargain as the next guy. And a group of AIDS activists is taking this approach in hopes of containing the rising cost of prescription drugs...In California and Ohio, they’re pushing ballot measures that would require state programs — such as Medicaid or prison systems — to pay no more for medicines than the Department of Veteran Affairs... “The goal [ballot initiatives] is to obtain lower prices, as well as gain more transparency on industry pricing, which is opaque,” said Ged Kenslea of the AIDS Healthcare Foundation...“And we’re hoping these will serve as a catalyst to other organizations or state legislators to enact similar measures in other states.”...well-intentioned, the effort may be somewhat quixotic...“These state initiatives might save a fistful of money in the short term and the first states where they are passed will get most of the benefit,”...“But the companies will find a way to compensate. So it isn’t likely to solve the problem in the long run.”...Still, the state initiatives may yet lead to change, if only because a groundswell of citizen-driven ballot measures might just force lawmakers to pay attention. In the end, getting a better deal might only be a vote away.
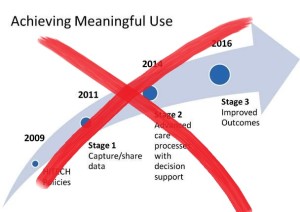
- CIOs celebrate end to meaningful use, want more details on future programs (healthcareitnews.com)Andy Slavitt puts meaningful use on ice; Read his J.P. Morgan speech transcript (healthcareitnews.com)
Some execs say easing off of the requirements will allow providers to focus more on innovation...Healthcare chief information officers breathed a sigh of relief on Tuesday when Andy Slavitt said the end of the meaningful use program was near. But many are waiting on the details before celebrating too much...The acting administrator of the Centers for Medicare and Medicaid Services Andy Slavitt said in a speech at the J.P. Morgan Healthcare Conference that meaningful use would be replaced with a more streamlined regulatory approach in line with the merit-based alternative payment models of the Medicare Access and CHIP Reauthorization Act of 2015...“The early stages of meaningful use took the country from ‘zero to 60' in five years, a remarkable achievement that would not have been possible without HITECH,”...“Now, it's time for the new payment model that rewards providers for achieving better health outcomes to be the driver of innovation, using the new electronic infrastructure that is now in place. That was the vision for ‘Phase 3’ from the very beginning.”..."It's truly unfortunate, but I'm not surprised,”...“The lack of alignment in Washington, varied interpretations by our industry and its vendors, and the resulting and inefficient ‘MU clicks,’ as termed and borne by our physicians have led down this path."...“The sad part of it all is that MU was designed with best intentions yet fated by regulation and political interests,”...