- Two serialization companies team up to tackle counterfeit pharmaceuticals (americanpharmacynews.com)Prohibition on proactive serialization for the EU FMD (securingindustry.com)
Systech International and Servicepoint recently joined forces to provide serialization and automation solutions to the pharmaceutical manufacturing industry...Internationalization, outsourcing, e-tailing and the expansion of international trade zones have created enormous complexities throughout the supply chain and product life cycle...This has resulted in a rapid escalation of global counterfeiting issues, threatening consumer safety like never before...Systech is known to have pioneered serialization and is becoming a model for the future of authentication. It unifies and protects the supply chain through...authentication and track and trace technologies that ensure regulatory compliance and reduce risks. These technologies have been used not only in the pharmaceutical industry, but also in industries, such as life science and consumer packaged goods...As part of a regulation adopted by the European Commission in October, the serialization of prescription medicine packages will be mandatory in all European Union countries by 2019. There are currently 15,000 prescription medicine production lines in Europe, and some may have to be either replaced or automated in order to put serialization into effect.
- How Practice Settings Will Be Affected by Changing Pharmacy Trends (pharmacytimes.com)
James Hoffman, PharmD, MS, FASHP, explains how pharmacy practice settings may change going forward.
- NOAC Dosing for Obese Patients vs. Non-Obese Patients (pharmacytimes.com)
Katie Greenlee, PharmD, BCPS-AQ Cardiology, of the Cleveland Clinic discusses how NOAC (novel oral anticoagulants) dosing for obese patients differs from NOAC dosing for non-obese patients.
- FDA launches Web-based precision medicine platform for next-generation sequencing (healthcareitnews.com)FDA Launches precisionFDA to Harness the Power of Scientific Collaboration (blogs.fda.gov)
Food and Drug Administration...launched the beta version of precisionFDA, its new collaborative platform for the exploration of next-generation gene sequencing...First announced in August, the platform features more than 20 public and private sector participants…Next-generation sequencing enables researchers to compile a vast amount of data on a person's exact order or sequence of DNA...scientists can look for meaningful differences in DNA that can be used to suggest a person's risk of disease, possible response to treatment and assess their current state of health. Ultimately, what we learn about these differences could be used to design a treatment tailored to a specific individual...The hope is to grow this community and improve the usability of precisionFDA in the coming months and years...One way we'll achieve that is by placing the code for the precisionFDA portal on the world's largest open source software repository, GitHub, so the community can further enhance precisionFDA's features...
- SEC, in complaint against Shkreli, alleges ‘widespread’ fraud over nearly 5 years (cnbc.com)Shkreli, CEO Reviled for Drug Price Gouging, Arrested on Securities Fraud Charges (bloomberg.com)
Martin Shkreli...was arrested by the FBI...after a federal investigation involving his former hedge fund and a pharmaceutical company he previously headed...The securities fraud probe of Shkreli, who is now chief executive officer of Turing Pharmaceuticals and KaloBios Pharmaceuticals, stems from his time as manager of hedge fund MSMB Capital Management and CEO of biopharmaceutical company Retrophin...SEC alleged that Shkreli engaged in "widespread fraudulent conduct" from at least October 2009 to March 2014... "made material misrepresentations and omissions to investors and prospective investors,"...once Shkreli took Retrophin public, he "fraudulently induced" the company to fund settlements with individuals who had claims against Shkreli from their investments in his hedge funds...
- Pharmacist Discusses Drug Abuse in HBO Documentary (pharmacytimes.com)
A Massachusetts pharmacist is speaking out about prescription drug abuse and heroin addiction in a new HBO documentary...Lauren Heroux-Camirand, RPh, conducted 2 interviews with the filmmakers of "Heroin: Cape Cod USA" at Country Square Pharmacy in Attleboro, Massachusetts...she thought it was important to talk openly about illicit drug use... “We are here, as pharmacists and humans, to be compassionate and help in any way we can,”...“That is why I agreed to participate and speak up.”...In her interviews with the filmmakers, Heroux-Camirand discussed both facts about drug abuse and her experiences seeing people struggle with addiction...The movie will premiere in Massachusetts before debuting on HBO on December 28, 2015
- Prevention Strategies Pharmacists Can Use to Reduce Errors (pharmacytimes.com)
Natasha Nicol, PharmD, FASHP, director of Global Patient Safety Affairs at Cardinal Health, provides prevention strategies to reduce medication errors.
- Mr. Mucus-repped cough meds get the most positive buzz online (fiercepharmamarketing.com)
Score one for Mr. Mucus. A new study found that the slimy spokes-character helped propel Reckitt Benckiser's family of Mucinex brands to three of the top four slots on a new list of preferred OTC cough-and-cold medicines. Treato, the online data-analysis company, created the list by analyzing findings from more than 5 million patient-written online posts and reviews...Mucinex D was No. 1, earning 4.6 (5-point satisfaction scale), followed by Delsym, which is also a Reckitt brand. Mucinex and Mucinex DM ranked Nos. 3 and 4, with Pfizer's Robitussin DM and Dimetapp brands tied at No. 5...Reckitt Benckiser's investment in DTC advertising is clearly paying off as consumers are disproportionately discussing their brands online...
- Under pressure, CDC delays release of opioid prescribing guidelines (statnews.com)Draft CDC Guideline for Prescribing Opioids for Chronic Pain, 2016: Summary of Constituent Comments and CDC Response (freepdfhosting.com)Painkiller politics: Effort to curb prescribing under fire (hosted.ap.org)
Under mounting criticism, the Centers for Disease Control and Prevention has delayed plans to...release controversial opioid prescribing guidelines for primary care physicians. Instead, the guidelines will now be issued sometime later in the year...The delay was first signaled yesterday when the CDC unexpectedly disclosed that public comments on its proposal would be accepted through Jan. 13. However, the agency will also tap the National Center for Injury Prevention and Control’s Board of Scientific Counselors, a federal advisory committee, to review the guidelines and public comments...The postponement follows complaints from representatives of other federal agencies and consumer advocacy groups that the CDC guidelines were based on weak evidence and would unfairly restrict some patients from obtaining needed pain relief. The agency was also chastised for using a “secretive” process to formulate the guidelines, which some critics argued had violated federal law...the CDC delay apparently reflects a desire to comply with the Federal Advisory Committee Act, which governs how expert groups are formed to provide official advice.
- The testing of new drugs is on the rise. Why that worries some people. (washingtonpost.com)
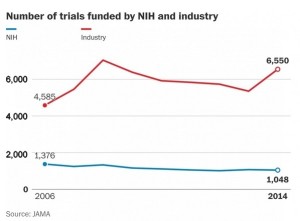
As the budget for the National Institutes of Health, the nation's largest funder of biomedical research, has flatlined over the past decade, the number of clinical trials funded by the federal agency has dramatically declined, a new study has found. At the same time, the number of trials funded by the pharmaceutical industry has exploded...Meanwhile, the number of NIH-sponsored trials fell from 1,376 registered on a website in 2006 to 1,048 by 2014, according to the study, which was published in the Journal of the American Medical Association. Industry-sponsored trials grew by about 2,000 over the same period, reaching 6,550 registered in 2014...More trials is generally a good thing...because it means that more new drugs, devices or other treatments are being tested. But the decline in federally funded trials concerns public health researchers, because federally funded trials are independent, often intended to compare drugs or other treatments instead of simply to show that a product works and should gain federal approval…there are many areas of deserving research...there may need to be a formal discussion of how research dollars are spent. A reasonable way to begin...is to look at the burden of a particular disease and allotting research dollars to those that have the greatest effect on mortality and disability...