- Pharmacy Week in Review: March 13 (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
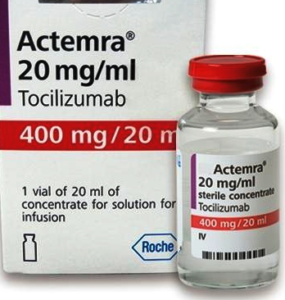
- China turns Roche arthritis drug Actemra against COVID-19 in new treatment guidelines (fiercepharma.com)
Some patients infected with the novel coronavirus can develop uncontrolled immune response, leading to potentially life-threatening damage to lung tissue. After seeing promising results in clinical practice, Chinese authorities now recommend a Roche arthritis drug to tackle that rampage...Roche’s blockbuster Actemra, first approved by the U.S. FDA in 2010 for rheumatoid arthritis, can now be used to treat serious coronavirus patients with lung damage, China’s National Health Commission said in its updated treatment guidelines for COVID-19...READ MORE
- Merck CEO Frazier finds fault in new studies suggesting pharma profits are too high (fiercepharma.com)
Is the pharma industry hiking up drug prices to boost profitability at the expense of hardworking Americans? That question—a favorite of politicians on both sides of the aisle—is at the heart of a special issue of the Journal of the American Medical Association, which features a collection of articles that aim to provide a well-rounded answer...Perhaps the most damning of the studies compares the profits of the 35 biggest pharmaceutical companies to the S&P 500 as a whole. The study...found that between 2000 and 2018 the median annual gross profit margin of Big Pharma was 76.5%, versus just 37.4% for the S&P 500 companies. Earnings before taxes and net income margins were also significantly higher among pharma companies, the authors reported...Merck CEO Kenneth Frazier had his own set of worries about the new JAMA studies, though he was more focused on fairness—or lack thereof...READ MORE
- Coronavirus spurs India to restrict exports of 2 dozen drugs (fiercepharma.com)India's restrictions on API exports only temporary, official says: report (fiercepharma.com)
While eyes have been on China for signs that COVID-19 might result in drug shortages, India has come up with a surprise of its own. The country, which accounts for about 40% of U.S. generic drugs, has halted exports of more than two dozen APIs and drugs...India’s Directorate General of Foreign Trade today announced it was restricting 26 APIs and formulations until further notice...The government gave no further explanation, but Dinesh Dua, chairman of the Pharmaceuticals Export Promotion Council of India, told Reuters, “Irrespective of the ban, some of these molecules may face shortages for the next couple of months.” If interruptions from the virus get worse, he said, some shortages may become “acute.”...READ MORE
- What we know about drugmakers’ response to the new coronavirus (biopharmadive.com)
The new coronavirus is moving quickly around the world. Since December, when it was first identified in Wuhan, China, the virus has spread to more than 100 countries. Some 105,000 cases have been confirmed, though the true number is surely higher, and at least 3,500 people have died. Already, the virus known as SARS-CoV-2 is straining healthcare systems in the hardest-hit regions, and threatens to do the same elsewhere as the infection total grows...BioPharma Dive compiled a roundup of our coronavirus coverage so far. There are many unanswered questions, and the drug industry's response is just beginning. We will update the roundup as we get more information...READ MORE
- This Week in Managed Care: March 6, 2020 (ajmc.com)
Christina Mattina, welcome to This Week in Managed Care from the Managed Markets News Network
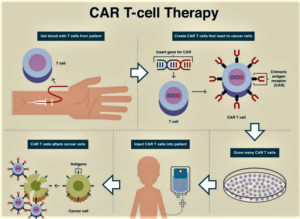
- CAR-T—the Future of Medical Progress Is Now (realclearhealth.com)
Personalized medicine is the future of medical progress...For instance, an immunotherapy treatment called chimeric antigen receptor T-cell therapy is accessible now, and the Trump administration has the opportunity to make it widely available to Medicare beneficiaries...over 70 Members of Congress—Democrats, Republicans, liberals, and conservatives—sent a letter to Seema Verma, administrator of the Centers for Medicare and Medicaid Services, commending the administration for “ensuring Medicare patients nationwide have access” to this life-saving treatment. The Congressional letter goes on to ask the administration to “ensure that hospitals are appropriately reimbursed so they may continue to provide” CAR-T therapy to America’s seniors...without appropriate reimbursement policy, a Medicare patient could be denied access to a treatment that would save his or her life. Without proper reimbursement by Medicare, providers simply will not be able to offer it as an option, especially in rural areas as patients must stay near a treatment center for four weeks to be monitored...READ MORE
- Dirty disposable socks? Cipla sterile injectables plant hit with FDA warning letter (fiercepharma.com)
When an inspection of Cipla’s sterile injectable plant in Goa, India, went badly last fall, it appeared pretty certain a warning letter would follow. The inspection report, after all, went on for 38 pages, detailing equipment layered in residue and bacterial contamination. The bad news has now arrived...The Indian drugmaker confirmed in a filing...that it had received a warning letter for the September inspection of the sterile products plant in Vasco Da Gama in Goa state. It said it “will work closely with the agency to comprehensively address all the observations” but didn't mention there were a dozen of them, many targeting shoddy cleaning methods and microbial contamination...READ MORE
- March 6 Week in Review (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- The Virus and the Supply Chain (city-journal.org)
The new coronavirus outbreak may be very bad for your health but not only for the reasons you imagined. This coronavirus is less likely to harm you directly than to injure you through its impact on your other medical needs...COVID-19 is more likely to harm Americans indirectly because the U.S. is increasingly reliant on drugs either directly sourced from China or made from intermediate chemicals...While 90 percent of the finished drugs Americans take are generics, most are manufactured overseas, primarily in India and China. Even India...relies on China for 80 percent of the APIs it uses in drug production...COVID-19 has resulted in massive disruption of Chinese manufacturing. It’s only a matter of time until this translates into supply disruptions for China-dependent customers...Coronavirus has created concerns about not only the quantity of Chinese medical products available but also about the virus’s effect on quality. China does not effectively regulate Chinese drug manufacturers. Multiple episodes have cast doubt on the safety and efficacy of their products...READ MORE