- Rite Aid launches genetic testing for Rx (chaindrugreview.com)Theranos isn’t the only diagnostics company exploiting regulatory loopholes (theverge.com)
Rite Aid Corp. has begun offering Harmonyx Diagnostics genetic testing for medications, making it the nation’s first drug chain to provide the service…Rite Aid said…that the self-administered oral swab test — available in all of its pharmacies…helps patients determine the efficacy of their prescription medication based on their genetic makeup…Patients can buy the test kit at any Rite Aid…In the pharmacy, patients then swab inside their cheek, and then a Rite Aid pharmacist takes the sample and returns it to Harmonyx for processing. Harmonyx provides a state-licensed physician to review each patient’s test, and detailed results are sent to the pharmacist and the treating doctor. The results are received as soon as 24 hours after the laboratory receives the test, according to Rite Aid.
- Germany, U.S. in hot pursuit of ‘messenger’ drug molecules (newsdaily.com)
A molecule that carries the recipe for making drugs inside body cells is exciting scientists and investors alike, attracting hundreds of millions of dollars in a scramble for the next promising area of biotechnology… synthetic messenger RNA, or mRNA technology, a new approach to tackling a range of hard-to-treat diseases... In theory, the promise of mRNA is enormous, ranging from cancer to infectious diseases to heart and kidney disorders, since it could be used to tackle the 80 percent of proteins that are difficult to affect with existing medicines…In effect, mRNA serves as software that can be injected into the body to instruct ribosomes, the “3D-printers” found inside cells, to churn out desired proteins…This is a radically different approach from conventional approaches, where therapeutic proteins are produced outside the human body and…then be inserted back into the human body at great complexity and cost…“The field is moving very rapidly,”...“I predict it will have a significant impact.”...
- Safeway to dissolve partnership with Theranos (pharmacist.com)
…code-named ‘T-Rex’...Safeway Inc. spent about $350 million to build clinics in more than 800 of its supermarkets to offer blood tests by startup Theranos Inc…But the tests never began, the clinics are now used largely for flu shots and travel-related vaccines, and the two companies have been negotiating to officially dissolve their partnership…Safeway executives said Theranos missed deadlines for the blood-testing rollout…article reported that the proprietary lab instrument developed by Theranos as the anchor of its growth strategy handled just a small fraction of the tests sold to consumers at the end of 2014…also…some of the startup’s former employees were leery about the machine’s accuracy.
- Known Knowns and Unknowns of U.S. Drug Pricing (blogs.plos.org)
Media scrutiny of high drug costs…spending on drugs represents 10% of overall health care costs. Together, hospital and physician expenditures account for 6 times the spending on drugs. Despite a recent uptick in the rate of drug cost growth, over the past 10 years the pace of hospital and physician expenditures has exceeded prescription drugs…these facts do not make for eye-catching headlines. By contrast, the recent surge in the price of drugs targeting hepatitis C, HIV, and various cancer and orphan diseases, is salient and the focal point of media attention…Here, we unravel several mysteries surrounding drug pricing and alignment of price and value.
- Why is pricing in the U.S. different?
- How are drugs priced in the US?
- What about R&D and its relation to price?
- Then what?
- But, does price = value?
- What are the alternatives?
- Summary
- More than 100 women sue over mispackaged birth control after becoming pregnant (mcall.com)
More than 100 women who became pregnant after allegedly taking mispackaged birthcontrol pills filed suit in Philadelphia last week against Qualitest Inc., a subsidiary of the Irish drug-maker Endo Pharmaceuticals Inc…The case seeks millions in damages, including in some cases the costs of delivering, raising, and educating the children borne of the unplanned pregnancies…The discovery of the mispackaged contraceptives prompted the FDA in 2011 to issue a recall notice for 3.2 million blister packs, according to court filings…. The FDA recall was triggered when a Kansas City woman returned a package to her pharmacist after noticing the blister pack had been rotated 180 degrees, reversing the weekly tablet orientation, according to the suit filed in Philadelphia Court of Common Pleas.
- Drug Prices, Manufacturer Rebates, and the Risk to Channel Economics (drugchannels.net) Prescription Medicines: Costs in Context (phrma.org)
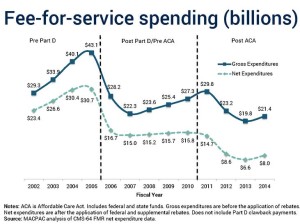
Gross Expenditures = payments to pharmacies Net Expenditures = payments to pharmacies minus federal and state supplemental rebates
When does a price increase not increase the price?...This riddle came to me while reviewing the excellent new Pharmaceutical Research and Manufacturers of America slide deck called Prescription Medicines: Costs in Context. The slides argue—...that pharmaceuticals have been undervalued as a source of health benefits and unfairly maligned as the key driver of costs… Despite the growth in drug list prices, manufacturers are experiencing limited growth in net revenues, which deduct payer rebates. As the data show, this difference has grown sharply over the past two years. Meanwhile, drug list price increases are boosting revenues at drug channels companies—pharmacies, wholesalers, and PBMs. These intermediaries are still compensated on gross (not net) revenues. I wonder: Will the growing gross-to-net disparity make current channel economic arrangements unsustainable for wholesalers and pharmacies?...
- Theranos isn’t the only diagnostics company exploiting regulatory loopholes (theverge.com)
Avoiding pre-market verification is downright easy…Theranos isn’t alone in avoiding regulation using an easily exploited loophole — in fact, it’s just one among many…Pathway Genomics, Admera Health, and Strand Life Sciences are diagnostics companies that offer cancer tests that impact people’s health care decisions. None of these companies have published data about their tests in peer-reviewed journals. Nor were any of these companies required to show regulators that their tests worked before they started marketing them to patients and physicians. That’s because each of these companies has been making use of what's known as the "laboratory developed test" loophole — which makes avoiding pre-market verification downright easy...Under the LDT loophole, any company that develops and conducts a diagnostic test in their own lab…can avoid submitting that test to the FDA before using it on patients. This get-out-of-regulation-free card exists because research hospitals often modify commercial tests. In the case of these hospitals, the academic researchers tend to publish their results anyway…The FDA knows this — even they’ve been calling LDTs a loophole — and the agency wants to change the way these tests are regulated…until that loophole closes, we’ll keep seeing companies using the LDTs as a dodge, because it’s a simpler and cheaper way to get to market. The real expense, of course, is patients’ health.
- Gene therapies offer dramatic promise but shocking costs (washingtonpost.com)
…gene therapy might soon find itself steeped in a new controversy: soaring drug prices…crucial questions about how much patients will pay directly…industry leaders are…talking about ways to get ahead of potentially massive one-time price tags that could make insurers and patients balk…A gene therapy approved in Europe in 2012 costs close to $1 million, and prices are expected to follow suit in the United States. The therapies in the pipeline are mostly for rare genetic diseases: sickle cell, hemophilia or immune deficiency. Their likely high prices stem from the expected value; unlike drugs that a person takes regularly, gene therapies are designed to be given once and have lasting effects…But everyone involved anticipates the potential backlash against a seven-figure price tag, which is leading to radical proposals. Instead of paying for a treatment all at once, insurers and patients could make installment payments as long as the therapy works…Some researchers are adding up the cost of the traditional treatments that a patient will be able to avoid each year to determine a price that, although high, could lead to savings for the health-care system.
- Pharmacy Week in Review: November 13, 2015 (pharmacytimes.com)
Mike Glaicar, Business Development: Pharmacy Times...(PTNN) This weekly video program highlights the latest in pharmacy news, product news, and more. (video)
- Drug makers kept many clinical trial results a secret: study (pharmalot.com)
When it comes to disclosing clinical trial data, some drug makers are still keeping secrets, according to a new study…Thirty-five percent of all trial results for 15 drugs that were approved in 2012 by the Food and Drug Administration were not publicly disclosed. And nearly 30 percent of the trials conducted for those drugs failed to meet legal disclosure requirements…“This confirms that pharmaceutical companies often fall below legal and ethical standards,”…The findings come amid growing clamor from academics and consumer groups to press drug and device makers to release trial data. If research is not published or reported in accessible registries, physicians and patients are prevented from having a complete picture of the risks and benefits of medicines…Such concerns have been heightened following various safety scandals that revealed trial data for some products was never fully published or disclosed. A few notable examples include…Vioxx.. and…Paxil…Over the past year, regulators in the US and Europe responded to concerns by releasing new rules designed to widen access. The World Health Organization has released a new position statement calling for companies to publish all research studies…Sense About Science, a UK nonprofit that launched the AllTrials campaign to widen access to data, is working with 85 asset managers and pension funds to assess steps taken by drug makers to provide trial information…







