- FTC raises anticompetitive concerns about FDA naming proposal for biosimilars (pharmalot.com)Nonproprietary Naming of Biological Products Guidance for Industry DRAFT GUIDANCE (fda.gov)In Response to a Request for Comments on Its Guidance for Industry on the “Nonproprietary Naming of Biological Products; Draft Guidance for Industry; Availability” (ftc.gov)
What’s in a name?..A contentious debate over identifying biosimilars is sparking concern from antitrust regulators. These drugs are designed to emulate expensive biologics and are forecast to save billions of dollars in US health care costs. But finding the best approach for naming biosimilars has confounded regulators and divided the pharmaceutical industry amid clashes over patient safety and the potential for big profits…At issue is whether biosimilars should be given the same name as biologics. Two months ago, the Food and Drug Administration issued draft guidelines that suggested both biologics and biosimilars can use the same name. But the agency also proposed that biosimilar names add a four-letter suffix that differs from the four-letter suffix that should follow a biologic brand name medicine…
- Cryotherapy under fire after death of spa worker (video.foxbusiness.com)Nevada agencies say cryotherapy death is not their investigation (reviewjournal.com)
KryoLife CEO Joanna Fryben on the reported health benefits of cryotherapy and concerns about the potential risks.
- Warner Chilcott Agrees to Plead Guilty to Felony Health Care Fraud Scheme and Pay $125 Million to Resolve Criminal Liability and False Claims Act Allegations (justice.gov)
Warner Chilcott U.S. Sales LLC, a subsidiary of pharmaceutical manufacturer Warner Chilcott PLC, has agreed to plead guilty to a felony charge of health care fraud, the Justice Department announced...The plea agreement is part of a global settlement with the United States in which Warner Chilcott has agreed to pay $125 million to resolve its criminal and civil liability arising from the company’s illegal marketing of the drugs Actonel, Asacol, Atelvia, Doryx, Enablex, Estrace and Loestrin…company committed a felony violation by paying kickbacks to physicians throughout the United States to induce them to prescribe its drugs, manipulating prior authorizations to induce insurance companies to pay for prescriptions of Atelvia that the insurers may not have otherwise paid for and making unsubstantiated marketing claims for the drug Actonel…As part of today’s resolution, the whistleblowers will receive approximately $22.9 million from the federal share of the civil recovery.
- 6 Southern Nevada hospitals included in paying $250M settlement (reviewjournal.com)
Six Southern Nevada hospitals are among hundreds of U.S. facilities (457 hospitals in 43 states) that will pay a total of more than $250 million stemming from allegations that they implanted cardiac devices in Medicare patients in violation of coverage requirements, the U.S. Justice Department said…Medicare guidelines provide that doctors should not implant ICDs (implantable cardioverter defibrillator) in patients who have recently suffered a heart attack or had other procedures, such as heart bypass surgery. Each of the hospitals…had implanted ICDs during 40 day waiting periods that Medicaid requires for heart attack patients and 90 day waiting periods for bypass patients…Las Vegas-area hospitals involved include Centennial Hills, Desert Springs, Summerlin, Valley, Sunrise, and North Vista...
- How Updated Beers Criteria Will Affect Pharmacists’ Practice (pharmacytimes.com)2015 AGS Beers Criteria and Opioid Warnings (pharmacytimes.com)6 Key Beers Criteria Updates Pharmacists Should Know (pharmacytimes.com)
Todd Semla, MS, PharmD, BCPS, FCCP, AGSF, of the Department of Veterans Affairs explains how the AGS 2015 Beers Criteria will affect pharmacists' practice. (video)
- Sherwin-Williams creates paint that can kill bacteria (reviewjournal.com)
Sherwin-Williams wants to paint a cleaner picture of hospitals…The company created a paint that it claims will kill bacteria -- a major cause of healthcare-associated infections in hospitals. Among the targeted bacteria are Staph, MRSA, E. coli, VRE, and Enterobacter aerogenes…"Paint Shield" claims to kill over 99.9% of these bacteria after they've been on a painted surface for two hours. It also reduces the growth of "common microbes."…It's one of the most significant technological breakthroughs in our nearly 150 year history of innovation…By killing infectious pathogens on painted surfaces, Paint Shield is a game-changing advancement in coatings technology...
- MD Labs solution could change, enhance pharmacists’ role (drugstorenews.com)
MD Labs (Reno based) is currently making the rounds to retail pharmacy operators with a new service solution that pharmacists are uniquely qualified to deliver. And it’s a solution that will significantly enhance the role pharmacists already play between providers and patients…That new service is pharmacogenetics, which uses the DNA from a cheek swab sample to help determine the best course of pharmacotherapy for an individual patient across 200 medicines — a number that’s growing…The new RxIGHT Pharmacogenetic Test...can eliminate trial-and-error prescribing of medicine, in which a doctor prescribes a medicine and measures the impact of that course of therapy after the fact, rather than quickly identifying the most appropriate course of therapy…It drives adherence and — perhaps most importantly — improves outcomes.
- Pharmacy Podcast – Restating the Importance of Pharmacy (pharmacypodcast.com)
Ron Lanton with True North Political Solutions interviews Kurt Proctor, Ph.D., RPh Senior Vice President, Strategic Initiatives with the National Community Pharmacist Association while at the NCPA 2015 Annual Conference in Washington D.C. (podcast 14:43m)
- The Current State of Electronic Prior Authorization in the US (ajmc.com)National adoption Scorecard Electronic Prior Authorization (ePA) (epascorecard.covermymeds.com)
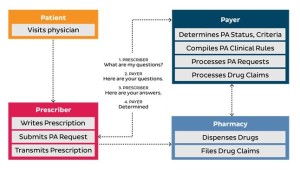
A majority of pharmacies have publicly announced that they are committed to implementing an electronic prior authorization solution that sends prior authorization requests electronically to the prescriber…70% of EHR systems…are committed to ePA. Eighty-seven percent of payers…are committed to ePA…CVS/pharmacy is the last major chain not yet committed to an ePA…many independent pharmacies have access to ePA functionality through pharmacy systems, most of which have live, integrated ePA capabilities…Prior authorization legislation has been considered, and in some states is in effect…Currently, 23 states have passed legislation pertaining to PA, and 4 additional states have pending legislation. The legislation seeks to make the PA submission process faster and easier for providers to prevent the delay of patient treatment…there is no federal direction on PA, so each state is on its own with respect to creating and enacting PA legislation...
- OPA announces opposition to marijuana oligopoly ballot initiative (ohiopharmacists.org)Ohio voters weigh legalizing recreational marijuana use (reuters.com)
Ohio Pharmacists Association, the state organization representing Ohio’s pharmacist practitioners, announced their opposition to the Ohio Marijuana Legalization Initiative, Issue 3, which if passed which would place legalized marijuana and a protected oligopoly of select commercial marijuana growers into the Ohio Constitution…OPA is unconvinced that marijuana is appropriate or beneficial as a form of treatment for illness or disease. Further, OPA rejects any medical marijuana proposal that removes the function, counsel, and expertise of the prescriber and pharmacist from the prescription process, as Issue 3 does…“If there is going to be a legitimate discussion about marijuana policy or the possible effectiveness of marijuana for medical purposes, then let’s have it. But ‘Responsible Ohio’ bypasses the discussion altogether in an effort to benefit to a few wealthy investors. Issue 3 is a slap in the face to Ohioans who genuinely care about sensible marijuana policy.”