- July 19 Pharmacy Week in Review: Study Finds Patients Attending Skin Cancer Screenings Likely to Be More Proactive with Sun Protection, Yogurt May Lower Risk of Developing Adenomas in Men (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- White House pulls drug rebate rule to relief of payers (biopharmadive.com)In win for pharma, Trump TV drug price rule struck down (biopharmadive.com)
The White House will withdraw a controversial plan to ban drugmaker rebates to pharmacy benefit managers in Medicare and Medicaid, stepping back from what the administration had once billed as "the most significant change in how Americans' drugs are priced at the pharmacy counter."..."Based on careful analysis and thorough consideration, the President has decided to withdraw the rebate rule...noting ongoing discussions about potential legislation around drug pricing...Drugmakers had supported the proposed rule, which fit with pharma's public relations campaign attacking the role played by PBMs in keeping drug costs high...READ MORE
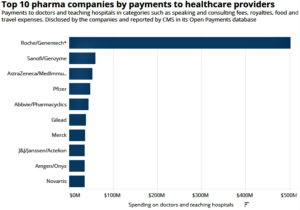
- Pharma shells out $3B to doctors and hospitals—with Roche, Sanofi leading: CMS (fiercepharma.com)The Facts About Open Payments Data (openpaymentsdata.cms.gov)
Drug and device makers shelled out $3 billion to doctors and teaching hospitals in 2018, a 3.5% increase from the previous year, according to the Open Payments database published...by the Centers for Medicare and Medicaid Services...Those payments include everything from royalties paid to teaching hospitals to physician speaking and consulting fees to free food and travel. The industry paid out an additional $4.93 billion for research and development...READ MORE
- This Week in Managed Care: July 5, 2019 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- JAMA: Withdraw This Flawed and Inaccurate Article About the 340B Program and Drug Prices (drugchannels.net)
The Journal of the American Medical Association recently published Estimated Changes in Manufacturer and Health Care Organization Revenue Following List Price Reductions for Hepatitis C Treatments by Sean Dickson and Ian Reynolds. The paper purports to show that manufacturers’ net revenues increased following a decrease in three drugs’ list prices...However, the paper contains a significant computational error: The authors do not properly calculate the 340B ceiling price. Consequently, the authors’ calculations are inaccurate and their conclusions are erroneous. As we might expect, a manufacturer’s revenue per-patient doesn’t increase when list prices decline and net prices remain constant...READ MORE
- Nevada State Board of Pharmacy July 2019 Newsletter (bop.nv.gov)
- CS: Reporting Theft or Loss
- National Pharmacy Compliance News July 2019
- FDA Changes Opioid Labeling to Give Providers Better Information on Tapering
- DEA Warns of Scam Calls Targeting Pharmacists and Other DEA-Registered Providers
- FDA Officials Outline 2019 Efforts to Improve Quality of Compounded Drugs
- China Agrees to Stricter Fentanyl Production Laws Following Pressure From US Lawmakers
- Two Lots of Transdermal Fentanyl Patches Recalled Due to Product Mislabeling
- FDA Releases Toolkit to Help Promote Safe Opioid Disposal
- This Week in Managed Care: July 12, 2019 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Billion-dollar settlement gets Reckitt out of federal opioid probes (biopharmadive.com)
...Reckitt Benckiser said it will pay up to $1.4 billion to settle all federal investigations related to the marketing of the opioid-dependence product Suboxone film by former subsidiary Indivior. The company denies all wrongdoing but said settling will avoid the "costs, uncertainty and distraction" of continued legal action... Reckitt's settlement is one of many now emerging from crackdowns on opioid marketing, with Purdue Pharma and Insys paying hundreds of millions to get out from under legal threats...The government claims Reckitt and Indivior deceptively marketed Suboxone Film as a safer, less divertable and less abusable form of opioid. Following the Reckitt settlement, these charges are still being made against Indivior, which was the target of a criminal grand jury indictment in the U.S. District Court for Western Virginia...READ MORE
- With test, Merck takes temperature-controlled drug delivery by drone from remote idea to remote locations (fiercepharma.com)
Drugmakers have speculated about delivering temperature-sensitive vaccines and drugs to remote locations using drones. Now, Merck & Co. is testing the method...Merck initiated the idea, and a collection of players coordinated by humanitarian aid organization Direct Relief have now moved to proof-of-concept missions. They developed and flew a drone with a temperature-controlled payload over the waters around the Bahamas..."This successful pilot demonstrates the potential of innovative unmanned aerial vehicles technology to aid in delivery of temperature-dependent medicines and vaccines to people who critically need them," Craig Kennedy, Merck's senior vice president of supply chain, said...Drone maker Volans-i built and operated the all-electric drones, while packaging expert Softbox developed the temperature-controlled payload box. Merck provided supply chain consulting...READ MORE
- July 12 Pharmacy Week in Review: Rate of Americans with History of Cancer Projected to increase; New Epinephrine Injections Available in Retail Pharmacies (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.