- Judge approves Teva’s $85 million opioid settlement with state of Oklahoma (cnbc.com)Suit: Generic drug makers used code to fix price increases (apnews.com)
An Oklahoma judge...approved Teva Pharmaceuticals’ revised $85 million settlement with the state over allegations the company helped fuel the nationwide opioid epidemic...decision came after state legislative leaders argued an earlier agreement did not comply with new state laws. Teva had reached an agreement with the state of Oklahoma last month, just two days before the drugmaker was set to face a trial alongside Johnson & Johnson...READ MORE
- New drug to boost women’s sex drive approved in US (apnews.com)AMAG wins FDA nod for Vyleesi. Could it be the ‘female Viagra’ that Addyi never was? (fiercepharma.com)
U.S. women will soon have another drug option designed to boost low sex drive: a shot they can give themselves in the thigh or abdomen that raises sexual interest for several hours...The medication OK’d...by the Food and Drug Administration is only the second approved to increase sexual desire in a women, a market drugmakers have been trying to cultivate since the blockbuster success of Viagra for men...The upside of the new drug “is that you only use it when you need it,”...“The downside is that it’s a shot — and some people are very squeamish.”...The FDA approved the new drug, Vyleesi (pronounced vie-LEE’-see), for premenopausal women with a disorder defined by a persistent lack of interest in sex, causing stress. The most common side effect in company studies was nausea...READ MORE
- June 21 Pharmacy Week in Review: Study Links BMI and Psoriatic Arthritis Severity, Survival Disparities Growing Among Young Patients with Cancer (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- NACDS applauds new Texas law to help curb opioid abuse (drugstorenews.com)
The National Association of Chain Drug Stores today praised Texas’ enactment of a bill (HB 2174) to address opioid abuse and addiction, while providing key safeguards to address the needs of those suffering from chronic pain...the new law will require electronic prescribing for controlled substances, to help prevent fraud and abuse. It also will limit the supply of a patient’s first opioid prescription to 10 days, when that prescription is for temporary, or acute, pain. It is important to note that this limit does not apply to prescriptions for ongoing, or chronic, pain. It also does not apply to cancer care, treatment of other illnesses, or end-of-life care...READ MORE
- GW Pharma takes docs on virtual trip to marijuana greenhouse (fiercepharma.com)
GW Pharma created a virtual reality voyage to allow doctors to see for themselves its pharma-quality growing operations and process…Looking to grow some trust, GW Pharma takes physicians on VR 'voyage' to marijuana greenhouse…GW Pharmaceuticals' strategy for promoting its new epilepsy drug Epidiolex (cannabidiol) to doctors sounded simple—earn their trust. But that's easier said than done, no thanks to Kim Kardashian…“A lot of people, especially in the medical community, were really skeptical about CBD. They associate it with pseudoscience and fluff…Epidiolex is the first and still only FDA-approved drug derived from the cannabis plant, and it's specifically indicated to treat two rare forms of epilepsy: Dravet syndrome and Lennox-Gastaut syndrome… The idea was to show doctors the grand scale of GW’s facilities as well as let them, from a more clinical standpoint, learn about the operation… If they only saw it, she told him, “they would understand that this is real medicine”…READ MORE
- This Week in Managed Care: June 21, 2019 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- EU industry loses €16.5bn in sales to counterfeit drugs (in-pharmatechnologist.com)2019 STATUS REPORT ON IPR INFRINGEMENT (euipo.europa.eu)
A report from the EUIPO demonstrated that a shift in the focus of counterfeited goods to include pharmaceutical products has caused significant loss of sales and employment...The European Union Intellectual Property Office has released a report this month regarding 11 analysed sectors in the region, including the pharmaceutical industry...The European Union organisation found that all analysed sectors lost sales as a result infringement of intellectual property rights for a total of €92 bn ($106bn) between 2013 and 2916...the pharma industry suffered the second biggest loss of sales, at €16bn…READ MORE
- New 340B Health Reports Confirms the Program’s Size—But Double-Dips on Hospitals’ (drugchannels.net)EVALUATING 340B HOSPITAL SAVINGS AND THEIR USE IN SUPPORTING CARE FOR RURAL AND LOW-INCOME PATIENTS (340bhealth.org)
340B Health, which lobbies for hospitals that participate in the 340B Drug Pricing Program, recently issued an eye-opening member survey. It found that 340B Health’s member hospitals reported total 340B discounts of $15.3 billion...The 340B Health report also claims to show evidence that hospitals are properly using 340B funds...In reality, the report merely affirms that hospitals are spending money to meet their fundamental legal and statutory community benefit obligations. Any money from 340B discounts shouldn’t be double-counted to meet hospitals’ fundamental spending requirements...This latest report therefore again highlights the need for transparency and accountability to ensure that billions in 340B discounts are shared with the patients whose prescriptions generated those funds...READ MORE
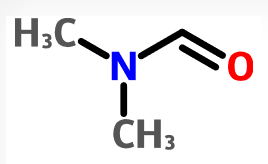
- Pharmacy takes FDA to task in citizen petition over tainted valsartan (fiercepharma.com)
The FDA has tied the contamination of blood pressure drugs by probable carcinogens to an approved switch in manufacturing to a process that uses certain solvents. Now an online pharmacy says it has discovered the solvents are as bad as the impurities they have been creating and has criticized the FDA for not taking steps to limit their use...In a citizen petition...Valisure says the FDA has established acceptable limits for impurities such as those commonly known as NDMA and NDEA after they were discovered in heart drugs last year, leading to massive global recalls...But it claims the FDA has not lowered the acceptable level for the solvent DMF, although it “has become apparent that the switch in the manufacturing industry to the use of the DMF solvent may be largely responsible” for the appearance of the impurities in the U.S. drug supply...The petition says the FDA needs to investigate the use of the solvent N,N-Dimethylformamide in drug manufacturing since it is classified as a Class 2 carcinogen...READ MORE
- Former FDA chief Scott Gottlieb says some claims about health benefits of CBD are ‘pretty hokey’ (cnbc.com)
Scott Gottlieb (Former FDA Commissioner) warns that health benefit claims about CBD have gotten well ahead of what science actually knows about the substance...“The idea that you can put it in dog food and it’s going to calm your dog ... I think that’s pretty hokey,” he says...CBD is not a “benign compound,” and regulations will be needed to protect people from possible unknown side effects, Gottlieb said...READ MORE