- May 3 Pharmacy Week in Review: Health Care Providers Urged to Encourage Parents to Vaccinate Their Children, Study Demonstrates Safety of Oral Immunotherapy for Peanut Allergy (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
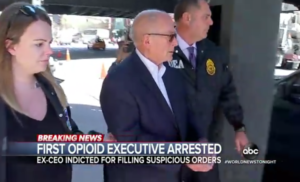
- Feds charge Rochester Drug Cooperative and CEO in first criminal case over opioids (abcnews.go.com)
Federal prosecutors charged drug distributor Rochester Drug Cooperative and its former CEO with drug trafficking charges...the first criminal charges for a pharmaceutical company and executives in the nation's ongoing opioid crisis... This prosecution is the first of its kind: executives of a pharmaceutical distributor and the distributor itself have been charged with drug trafficking...The U.S. Attorney's Office for the Southern District of New York charged Rochester Drug Cooperative...with "knowingly and intentionally" violating federal narcotics laws "by distributing dangerous, highly addictive opioids to pharmacy customers that it knew were being sold and used illicitly,"...RDC was also charged with failing to properly report thousands of suspicious orders of oxycodone, fentanyl and other controlled substances to the Drug Enforcement Agency...READ MORE
- Hospital-led generic drugmaker Civica Rx opens HQ in Utah (biopharmadive.com)
Civica Rx, a provider-led nonprofit generic drug company that aims to improve access to generic medication and lower drug costs, last week opened its headquarters in Lehi, Utah. The company plans to supply 14 generic drugs and have its first on the market by the end of the year...About two dozen healthcare companies, including Intermountain Healthcare, Mayo Clinic, Providence St. Joseph Health and Trinity Health are represented on Civica's governing board or are founding members in the nonprofit. Civica expects to partner with more than 900 U.S. hospitals...READ MORE
- FTC sues Surescripts, charges company with illegally monopolizing e-prescribing market (fiercehealthcare.com)
In its latest move to rein in what it views as anticompetitive tactics in the healthcare industry, the Federal Trade Commission filed a lawsuit against health information company Surescripts charging the company with illegally monopolizing the e-prescribing market...The FTC alleges that the company employed "illegal vertical and horizontal restraints in order to maintain its monopolies over two e-prescribing markets: routing and eligibility."... Surescripts has maintained at least a 95% share over many years...the FTC said it is seeking to undo and prevent Surescripts’s unfair methods of competition, restore competition, and provide monetary redress to consumers...READ MORE
- The obscure advisory committees at the heart of the U.S. drug pricing debate (reuters.com)
Expectations were high last year for three new migraine drugs hitting the market from Amgen Inc, Eli Lilly and Co and Teva Pharmaceutical Industries...But a small group of external medical experts who quietly advise U.S. health insurers on new drugs was not impressed, according to a private meeting held at UnitedHealth Group’s OptumRx offices...These relatively unknown expert committees have been involved in drug coverage decisions for decades. Their identities are kept secret due to federal regulations aimed at preventing pharmaceutical industry interference. The committees make their decisions based on a drug’s clinical value, independent of cost, pharmacy benefit managers say. But their power has grown more recently with the consolidation of most of the U.S. pharmacy benefits business under OptumRx, CVS and Express Scripts. Taken together, their three advisory committees now guide drug coverage for more than 90 million Americans...READ MORE
- This Week in Managed Care: April 26, 2019 (ajmc.com)
Jaime Rosenberg, welcome to This Week in Managed Care from the Managed Markets News Network
- Critics take aim at plan to allow prescription drug imports from Canada (pressherald.com)
Pharmacists and some health experts are opposing a proposal to permit the bulk importation of drugs from Canada to Maine, arguing that it could result in unsafe drugs being brought into the state and have unintended consequences, such as causing drug shortages in Canada...Proponents of Senate President Troy Jackson’s bill say those fears are unfounded, and believe it could be one of several proposals that would help to rein in prescription drug prices in Maine...If approved, the state would designate a wholesale purchaser of drugs from Canadian wholesalers...READ MORE
- Walgreens to raise tobacco buying age to 21 in September amid FDA pressure (cnbc.com)Rite Aid raises tobacco buying age to 21, following Walgreens’ lead (cnbc.com)
Walgreens is increasing the age to buy tobacco at its drugstores to 21 later this year as the retailer faces possible sanctions from the Food and Drug Administration for allegedly selling to minors...The FDA put Walgreens “on notice” in February, accusing the pharmacy chain of violating rules that prohibit selling cigarettes and other tobacco products to underage buyers. Walgreens, the FDA noted, is currently the top violator among pharmacies that sell tobacco products. Some 22 percent of Walgreens locations inspected by the agency caught employees illegally selling tobacco products to minors...READ MORE
- April 26 Pharmacy Week in Review: Comment Sparks Dialogue About Health Care Working Conditions, US Sees Second Viral Wave of Flu Season (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Popular heart drugs tainted with carcinogens face a wave of lawsuits (pressherald.com)
The FDA has been coordinating a recall of adulterated heart medications since last July...Dozens of lawsuits have been filed against drug makers and sellers over widely prescribed generic heart medications tainted with potential carcinogens, the first claims in what some lawyers expect to be a wave of litigation...the carcinogen NDMA was discovered in valsartan manufactured by Zhejiang Huahai Pharmaceutical Co. The contaminated valsartan was sold to a number of major drugmakers and used as an ingredient in other popular cardiovascular therapies...Zhejiang Huahai and its affiliates are the primary targets of the lawsuits. Other companies named in the complaints include generic-drug giants Teva Pharmaceutical Industries Ltd. and Mylan NV, as well as CVS Health Corp., which operates large pharmacy and drug-benefit management businesses. Almost 40 defendants have been sued so far...READ MORE