- Congressman aims to break pharma’s insulin ‘pricing monopoly’ by legalizing Canadian imports (fiercepharma.com)
U.S. legislators have been introducing bills left and right proposing a variety of methods for bringing down drug prices. Now, Vermont's congressman is zeroing in on a specific class of diabetes medicines that he says have become far too expensive: insulin...Rep. Peter Welch introduced a bill on Wednesday that would make it legal for patients, wholesalers and pharmacists to import insulin from Canada, and eventually from other countries with acceptable safety standards...Welch clearly wants to make an end run around Lilly and its fellow insulin makers. If his bill were to become law, patients with valid prescriptions would be able to import low-cost insulin and have it covered by their insurance plans...
- Expired drugs may remain effective, safe to use in a pinch (reuters.com)Expired Drugs in the Remote Environment (wemjournal.org)
Even medicines that are years past their expiration date and haven’t always been kept in strict climate-controlled conditions may still retain their original potency, a small study suggests...That is good news for people working in remote areas of the world where sometimes an expired medication is the only one available and the alternative is having no way to treat a serious illness...This date is not necessarily the point at which the drug becomes ineffective or dangerous, and for many medications, this window may be far longer than the usual two-to-three-year expiry date...The study team tested the stability of five expired drugs that had been returned from the British Antarctic Survey...They tested five types of drug, all one to four years past expiration, and compared these to fresh samples of the same medications to see if the expired versions were chemically stable and retained their active ingredient...Researchers found that all of the tested drugs were stable, and would, theoretically, have still been effective...
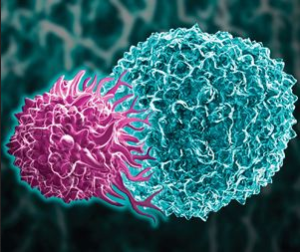
- CMS unveils CAR-T proposal, with emphasis on patient outcomes (biopharmadive.com)
Under a proposal...Medicare would cover CAR-T cell therapies through a Centers for Medicare and Medicaid Services pathway known as Coverage with Evidence Development...The proposal holds a mix of provisions, including that patients must be monitored for at least two years post-treatment. Hospitals administering CAR-T therapy, whether through inpatient or outpatient care, must participate in a CMS-approved registry that collects data on patient outcomes and characteristics and then compares that data to what's been seen in pivotal clinical trials of the therapy or standard of care treatment...Hospitals and clinicians would track certain clinical data elements at baseline, at treatment, and then at three-month, six-month, one-year and two-year follow ups following administration...
- FDA clamps down on illegal dietary supplements (pharmaceutical-technology.com)
The Food and Drug Administration has announced new measures to strengthen its regulation and oversight of dietary supplements, which have not been reviewed by the regulator...FDA Commissioner Scott Gottlieb said: “It’s clear that the US Food and Drug Administration plays an important role in helping consumers make use of safe, high-quality dietary supplements while also protecting Americans from the potential dangers of products that don’t meet the agency’s standards for marketing...the FDA is committing to new priorities when it comes to our oversight of dietary supplements at the same time that we carefully evaluate what more we can do to meet the challenge of effectively overseeing the dietary supplement market while still preserving the balance struck by Dietary Supplement Health and Education Act...the FDA posted 12 warning letters and five online advisory letters to US and international companies, which are illegally selling more than 58 products, primarily dietary supplements, as treatments for serious health conditions, such as Alzheimer’s disease, when their safety and efficacy is unknown...The companies contacted include Nutrition Coalition, TEK Naturals, Sovereign Laboratories and DK Vitamins...
- This Week in Managed Care: February 22, 2019 (ajmc.com)
Samantha DiGrande, Welcome to This Week in Managed Care from the Managed Markets News Network
- As medical costs mount, Japan to weigh cost-effectiveness in setting drug prices (reuters.com)
Japanese have easy access to new medicines, whose prices are decided by the government and subsidized by the country’s public health insurance system...But that may change. Japan, confronted with the ballooning cost of caring for an aging population, is introducing a cost-effectiveness test for drugs as a means of capping prices...There are no plans to deny care for patients of any age... Japan should carefully consider an impact on the industry when introducing such analysis to reduce drug prices...If Japan is going to cut prices so much, I think Japan will really run a risk of losing its current position...while drugmakers threaten to pull back from Japan, the government is prepared to call the industry’s bluff, saying Japan is too lucrative a market for companies to ignore... Unlike the United States, where insurers may deny claims, or the UK, where patients can be denied costly drugs, Japan is seen as a relatively predictable market because of its social insurance system...
- This Week in Managed Care: February 15, 2019 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- February 22 Pharmacy Week in Review: Blood Glucose Test Strips Prove Accurate, Chronic Inflammatory Diseases Linked to Cancer (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- FDA issues statement on new policy for pricing of generic drugs (chaindrugreview.com)
...In too many cases, branded drugs that are no longer protected by patents or other exclusivities do not face expected competition. In fact, there are several hundred of such branded drugs that do not have any generic competition. Instances like these may keep prices high and ultimately hurt American patients...We’ve been taking new policy steps at the FDA to support downward pressure on drug prices by helping to clear a path for more efficient generic development...We’re working to reduce barriers to generic development and to lower the cost of generic entry so that more of the generic medicines that the FDA approves are launched and reach patients...
- issue draft guidance with recommendations on establishing active ingredient sameness.
- development of new analytical tools and in vitro tests to provide additional accurate, sensitive and reproducible tools to support approval of complex generic drugs.
- laying out new, efficient guidelines for the use of a novel pathway that provides incentives for developing generic versions of drugs that currently face little or no competition - Competitive Generic Therapies
- February 15 Pharmacy Week in Review: Psoriasis Treatment May Help Prevent Heart Disease, Unproven Products Prompt FDA Warnings (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.