- This Week in Managed Care: December 7, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Bristol-Myers Squibb must face whistleblower suit claiming underpaid Medicaid rebates (fiercepharma.com)
Years after a former pharmacist sued Bristol-Myers Squibb and other companies for allegedly underpaying Medicaid rebates, a federal court rejected Bristol's attempt to escape the case...Pharmacist and lawyer Ronald Streck filed a False Claims Act whistleblower lawsuit against Bristol-Myers and other companies back in 2013, but later withdrew his claims against the other defendants. Now, his lawsuit alleges that BMS fraudulently manipulated its average manufacturer prices to underpay Medicaid rebates from 2007 to 2016...Pennsylvania federal judge Timothy Savage ruled last week that Streck "has alleged sufficient facts to state a false claims cause of action."...
- As state awaits data from diabetes drug manufacturers, initial report highlights price increases (thenevadaindependent.com)
Pharmaceutical companies are preparing to submit their initial reports detailing why rising prices of some diabetes drugs have outpaced medical inflation, giving state officials the first detailed look into the costs associated with a disease that affects about a tenth of Nevadans...Despite a protracted legal battle...pharmaceutical manufacturers are required to submit reports...to comply with a new diabetes drug transparency law… the state released a list of 175 so-called national drug codes...manufacturers...are required to submit reports to the state detailing the factors that contributed to the price increases...
- CVS Health Completes Acquisition of Aetna, Marking the Start of Transforming the Consumer Health Experience (cvshealth.com)CVS Completes $70 Billion Acquisition of Aetna (wsj.com)CVS-Aetna merger approved by New York regulators (nypost.com)
CVS Health...today announced that it has completed its acquisition of Aetna...“Today marks the start of a new day in health care and a transformative moment for our company and our industry,” declared CVS Health President and Chief Executive Officer Larry J. Merlo. “By delivering the combined capabilities of our two leading organizations, we will transform the consumer health experience and build healthier communities through a new innovative health care model that is local, easier to use, less expensive and puts consumers at the center of their care.”
- CVS offers ‘guaranteed net cost’ for pharmacy benefit clients (reuters.com)
Pharmacy chain and benefits manager CVS Health Corp...said...it will offer a new prescription benefit option guaranteeing its health plan clients 100 percent of any rebates, discounts or other fees paid by drugmakers...The new plan model is aimed at providing greater drug cost simplicity, predictability and transparency...Under its new option, CVS takes on the risk of drug price inflation and shifts in drug use - at least for the term of each contract...The new model guarantees average spending per prescription across each distribution channel – retail, mail order and specialty pharmacy...
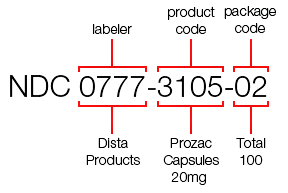
- The NDC Shortage: What the FDA Could (and Should) Do to Address It (drugchannels.net)An Open Letter To The FDA: New NDC Format Public Meeting (rxtrace.com)
...the U.S. Food & Drug Administration will run out of 5-digit National Drug Code codes within the next ten to fifteen years…The problem now facing U.S. healthcare is that, due to an explosion of new labelers entering the market, the FDA is running out of 5-digit labeler codes. The 5-digit format provides 90,000 potential combinations, and although that seems like a lot, the FDA anticipates running out of labeler codes within the next 10-15 years...the disruption and impact to the healthcare industry will be widespread and of a large magnitude, and will require retooling of major industry systems...
- This Week in Managed Care: November 30, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Antimicrobial Stewardship Programs Need Pharmacist Expertise (drugtopics.com)
As pharmacists continually build their scope of expertise, hospital anti-microbial stewardship programs are just one more avenue for putting their experience and knowledge to work...Anna Legreid Dopp, PharmD, director of clinical guidelines and quality improvement at ASHP, says pharmacists have a responsibility to take a prominent role in antimicrobial stewardship programs by
- Promoting the opial use of antimicrobial agents;
- Reducing the transmission of infections;
- Decreasing adverse events associated with antibiotic use; and
- Participating in formulary management and educating health professionals, patients, and the public.
- Walmart and Aurobindo sued as litigation mounts over contaminated blood pressure drugs (fiercepharma.com)
Litigation is growing over tainted blood pressure medications with a class-action lawsuit now filed in Florida against Walmart and three drug companies that span the supply chain...In addition to the retailer, the lawsuit names Indian drugmaker Aurobindo, U.S. drugmaker ScieGen Pharmaceuticals and distributor Westminster Pharmaceuticals. It was filed in federal court in Florida against the four over the sale of irbesartan because the drug contained the impurity, N-nitrosodiethylamine...Hauppauge...and its distributor Westminster initiated a nationwide voluntary recall to the consumer level of all lots of unexpired irbesartan tablets because they had been made with the Aurobindo API and then shipped to Walmart. The FDA learned this summer that the suspected carcinogen...can show up in “sartan”-based drugs as a result of certain manufacturing processes...
- November 30 Pharmacy Week in Review: Trial Evaluating Effectiveness and Safety of Drugs Used to Treat Patients with Ebola (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.