- GSK resumes some doctor payments, backtracking on blanket ban
GlaxoSmithKline, which five years ago stopped paying doctors for promoting its drugs, said...it would allow such payments once again in limited circumstances...The British group’s 2013 no-payment pledge marked a first for an industry battling past scandals over sales practices. But other drugmakers failed to follow suit, leaving it at a competitive disadvantage...Drugmakers have long used so-called key opinion leaders to promote the benefits of their products to other prescribing physicians and the decision to abandon this strategy was questioned by a number of analysts...GSK’s new updated policy...will now permit payments to global experts who speak about the science behind novel new medicines...“These changes are being made for a select number of innovative products in a limited number of countries and apply to restricted time periods in a product’s lifecycle,” the drugmaker said.
- Use of Evolution to Design Molecules Nets Nobel Prize in Chemistry for 3 Scientists (nytimes.com)The Nobel Prize in Chemistry 2018 (nobelprize.org)
Three scientists shared this year’s Nobel Prize in Chemistry for tapping the power of evolutionary biology to design molecules with a range of practical uses...Those include new drugs, more efficient and less toxic reactions in the manufacture of chemicals and plant-derived fuels to replace oil, gas and coal extracted from the ground...Half of the prize and the accompanying $1 million...Stockholm, went to Frances H. Arnold, a professor of chemical engineering at the California Institute of Technology. She is only the fifth woman to win a chemistry Nobel and the first since 2009...The other half of the prize is shared by George P. Smith, an emeritus professor of biological sciences at the University of Missouri, and Gregory P. Winter, a biochemist at the M.R.C. Laboratory of Molecular Biology in England...The prize highlights the narrowing of the gap between biology and some fields of chemistry as chemists turn to nature for inspiration...
- New Law To Force Payments Disclosure by Drug and Device Manufacturers to Non-Physician Providers (ptcommunity.com)
Sunshine Law Will be Expanded to Include Nurse Practitioners, Physician Assistants, Nurse-Midwives, and Others...Congress is expected to finalize within days a new law to expand transparency surrounding payment to non-physician providers. Beginning in 2020, pharmaceutical and medical device companies must disclose payments made to those providers for speaking or consulting, as they must do now with physicians...In many states, nurse practitioners, physician assistants, and other non-physician health professionals account for a substantial number of prescriptions—often including prescription opioids.” The law also applies to clinical nurse specialists, nurse-anesthetists, and nurse-midwives...Without full transparency, pharmaceutical companies can operate under the cover of darkness, possibly using gifts and payments to influence the prescribing practices of medical professionals like nurse practitioners and physician assistants...
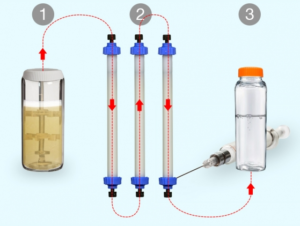
- A new way to manufacture small batches of biopharmaceuticals on demand (news.mit.edu)
Biopharmaceuticals, a class of drugs comprising proteins such as antibodies and hormones, represent a fast-growing sector of the pharmaceutical industry. They’re increasingly important for “precision medicine” — drugs tailored toward the genetic or molecular profiles of particular groups of patients...Such drugs are normally manufactured at large facilities dedicated to a single product, using processes that are difficult to reconfigure. This rigidity means that manufacturers tend to focus on drugs needed by many patients, while drugs that could help smaller populations of patients may not be made...To help make more of these drugs available, MIT researchers have developed a new way to rapidly manufacture biopharmaceuticals on demand. Their system can be easily reconfigured to produce different drugs, enabling flexible switching between products as they are needed...Traditional biomanufacturing relies on unique processes for each new molecule that is produced...We’ve demonstrated a single hardware configuration that can produce different recombinant proteins in a fully automated, hands-free manner...
- This Week in Managed Care: October 5, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- Impatient patients turn to online ‘buyers club’ for new drugs (reuters.com)
Frustrated by delays in new medicines reaching their own country, a small but growing number of patients are turning to an online broker that bills itself as a legal version of the Dallas Buyers Club...While regulators warn of the risk of buying drugs online, the Amsterdam-based Social Medwork sees its network of trusted suppliers as filling a gap in the market for the latest drugs against diseases such as cancer, migraine and multiple sclerosis...Now it is looking to raise its profile and expand, by signing up former EU Commissioner Neelie Kroes to its supervisory board and securing 1.5 million euros ($1.73 million) in new funding from the Social Impact Ventures capital fund...patients who cannot get the drugs they want through local healthcare systems are using the organization to self-import medicines from abroad...
- AmerisourceBergen to pay $625 million in U.S. civil fraud settlement (reuters.com)
AmerisourceBergen Corp...will pay $625 million to resolve civil fraud charges over the sale of syringes containing drugs for cancer patients, double billing, and providing kickbacks to doctors...The settlement...boosts AmerisourceBergen’s total payout to $885 million over its repackaging and distribution of pre-filled syringes that were not approved by the Food and Drug Administration...AmerisourceBergen admitted that from January 2001 to January 2014, its Medical Initiatives Inc pharmacy unit in Alabama shipped millions of syringes for patients undergoing chemotherapy that contained drugs prepared in an unsterile environment...Authorities said AmerisourceBergen would harvest “overfill” from the original vials of such drugs as Aloxi, Anzemet, Kytril and generic Kytril, Neupogen and Procrit...That enabled the company to create more doses than it bought, and generate at least $99.6 million of extra profit...
- October 5 Pharmacy Week in Review: 2018 Next Generation Pharmacist Awards, Vaccines for Health Care Professionals (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Banner Health, Ascension executives join hundreds of hospital CEOs defending 340B program (fiercehealthcare.com)
CEOs from more than 700 hospitals sent a letter to congressional leaders...calling on them to protect the 340B drug discount program, among them Banner Desert Medical Center...The 340B program requires drugmakers to provide discounted drugs to hospitals, health centers, and other healthcare providers that serve disproportionate numbers of low-income and rural patients...But the program has been under increased scrutiny after enjoying years of bipartisan popularity, including three expansions in Congress. Critics argue that hospitals are taking advantage of the discounts to line their wallets and are snapping up oncology providers to boost the number of pricey specialty drugs they can prescribe under the program...the letter said the group is concerned cuts to the 340B program would increase the prices of need and existing drugs...In our larger disproportionate share hospitals, the program enables us to provide a wider range of comprehensive services to people who are uninsured and traditionally underserved—services that would need to be scaled back along with any 340B reductions...For our more rural critical access hospitals, cuts to 340B would threaten the only places for people in the communities to get their care in the first place...
- FDA Warns of Dosing Errors With Compounded Injectables (raps.org)
The Food and Drug Administration last week warned that differences in how drugmakers and compounders label the strength of injectable drugs may lead to dangerous dosing errors..."Conventional manufacturers label their injectable products with the strength per total volume as the primary and prominent expression of strength on the label, whereas some compounders label their injectable products differently,"...For small volume parenteral drugs and biologics, FDA recommends that strength per total volume be the primary and most prominent expression of strength, followed by strength per milliliter enclosed by parentheses...two recent MedWatch reports cited medication errors that led to patients overdosing on compounded injectables that featured strength per milliliter more prominently on the labels than the strength per total volume...Such mix-ups could be avoided...if compounders were to follow the same labeling conventions as traditional drugmakers, though FDA does not review the labels of compounded drugs before they are sold.