- FDA OKs first generic under new approval pathway (drugstorenews.com)
This new approval pathway was created to expedite the development and review of a generic drug for products that lack competition...The FDA gave the nod for Apotex’s potassium chloride oral solution...“Today’s approval marks the successful implementation of a new program designed to encourage generic drug development for products with inadequate generic competition,” said FDA commissioner Scott Gottlieb, in a press statement...“The quick implementation of this new pathway is part of our broader effort to foster generic competition and help address the high cost of drugs. So are our efforts to narrow the time it takes for generic drugs to reach the market by reducing the number of review cycles that generic applications typically undergo. This new generic drug application was also approved in its first cycle of review. This approval demonstrates that the competitive generic therapy pathway is efficient and open for business. This pathway is a key step in making safe and effective generic drugs available to patients quickly and ensuring there’s adequate competition so patients have affordable access to the treatments they need,” said Gottlieb.
- Albertsons, Rite Aid terminate merger (drugstorenews.com)Rite Aid, Albertsons Call Off Merger Amid Investor Opposition (wsj.com)
Albertsons and Rite Aid on Wednesday evening announced that they had mutually decided to terminate the proposed merger...The termination came a day before Rite Aid was set to hold a special shareholders meeting at which the company has been urging its shareholders to vote in favor of the merger...“Albertsons believes that the strategic rationale of the Rite Aid combination was compelling, including the $375 million of cost synergies and $3.6 billion of identified revenue opportunities,” Albertsons said. “We disagree with the conclusion of certain Rite Aid stockholders and third-party advisory firms that although they acknowledged the strategic logic of the combination, did not believe that Albertsons was offering sufficient merger consideration to Rite Aid stockholders.”...Rite Aid chairman and CEO John Standley said that despite believing in the merits of the merger, it had listened to its stockholders and has committed to moving forward as a standalone company...
- Cashing in on DNA: race on to unlock value in genetic data (reuters.com)
How much is your DNA worth? As millions of people pay for home tests to check on ancestry or health risks, genetic data is becoming an increasingly valuable resource for drugmakers, triggering a race to create a DNA marketplace...GlaxoSmithKline’s decision to invest $300 million in 23andMe and forge an exclusive drug development deal...crystallizes the value locked up in genetic code...Firms like EncrypGen, Nebula Genomics, LunaDNA and Zenome are using blockchain...to secure sensitive DNA records and create a transaction ledger. The new players all have slightly different models, with most simply provide data platforms, where people are rewarded for providing data...For drugmakers...access to this data offers a way to accelerate drug development, since finding a drug target linked to a human genetic variant doubles the chance of producing a new medicine.
- Opioid Lawsuit Links Counties in Fight Against Big Pharma (bloomberg.com)
Mike Papantonio, senior partner at Levin Papantonio, discusses a class-action lawsuit against pharmaceutical companies over the U.S. opioid crisis. He speaks with Bloomberg's Kevin Cirilli on "Bloomberg Markets: Balance of Power." (Source: Bloomberg)
- This Week in Managed Care: August 10, 2018 (ajmc.com)
Laura Joszt, Managing Editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
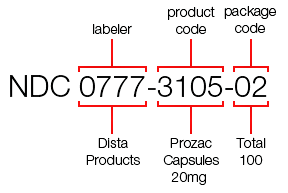
- FDA Anticipates Need for Expanding National Drug Code, Calls for Industry Input (raps.org)
The Food and Drug Administration announced...it plans to hold a public discussion on expanding the National Drug Code format and the impact on industry...FDA currently assigns 10-digit NDCs for the purposes of identifying drugs in the country and the format is comprised of three codes regarding the labeler, the product and the package...“FDA anticipates that it will run out of 5-digit labeler codes in approximately 15 years,” the agency wrote in the notice on the 5 November public hearing. “FDA will begin assigning 6-digit labeler codes at some point in the future due to exhaustion of 5-digit labeler codes.”...Prior to implementing the new formats for longer configurations, FDA officials will seek feedback from meeting participants on the potential impact on companies’ bottom line and drug safety, as well as “issues associated with the current lack of NDC uniformity in the marketplace.”
- China drug scandals highlight risks to global supply chain (cnbc.com)
The drug safety scandals...have underlined the risks to international consumers posed by weak oversight in China, the world's largest supplier of active pharmaceutical ingredients...The European Medicines Agency and the US Food and Drug Administration issued alerts over a cancer-causing ingredient used in a blood pressure medication, supplied by Chinese company Zhejiang Huahai, resulting in a recall of affected drugs...Then Beijing announced that hundreds of thousands of substandard vaccine doses had been sold in China, prompting a public outcry. Senior executives were arrested at the pharma company, Changsheng Biotech, which was also accused by authorities of forging data during the production of rabies vaccines...China is home to thousands of API producers, with exports worth $29bn last year...its producers supply ingredients for generic drugmakers such as Teva Pharmaceutical and multinationals including Johnson & Johnson and Novartis. About 80 percent of APIs used in the US come from China and India…Warnings to Chinese companies published by the FDA and EMA in recent years show that dozens have violated standards, mainly relating to record-keeping during the manufacturing process. In several cases the exporters shipped large volumes of product before the infractions were discovered...
- August 10 Pharmacy Week in Review: National Immunization Awareness Month, Bilingual Clinical Service for Diabetes Outcomes (pharmacytimes.com)
Nicole Grassano, PTNN, Pharmacy Week in Review, this weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Brexit Guidance: MHRA Outlines What to Expect (raps.org)
The UK’s Medicines & Healthcare products Regulatory Agency...explained what pharmaceutical and medical device companies can expect during the period that the Brexit agreement is implemented...During the implementation period, which will end in December 2020, pharmaceutical firms will be able to continue UK batch release testing and Qualified Person certification in the UK, which will be recognized by the EU and vice versa, MHRA said...But one of the setbacks is that for medicines, the UK will not have voting rights in EMA and EU committees during the implementation period, MHRA said...“Marketing authorisation holders and qualified persons for pharmacovigilance will continue to be able to be based in the UK and access EU markets. There will be continued mutual recognition of manufacturing and distribution licences, as well as associated inspections such as good manufacturing practice (GMP),” the regulator added.
- Antianxiety drugs — often more deadly than opioids — are fueling the next drug crisis in US (cnbc.com)
Today more than 40 million adults in the United States suffer from anxiety, and it is the most common mental illness in the United States...Overdose deaths involving benzodiazepines — such as Xanax, Librium, Valium and Ativan, drugs commonly used to treat anxiety, phobias, panic attacks, seizures and insomnia — have quadrupled between 2002 and 2015...The trend is being fueled by a 67 percent rise in prescriptions...The market for...(benzodiazepines)...is expected to reach $3.8 billion in the U.S. by 2020...many mental health experts are sounding the alarm, claiming that benzodiazepine addiction is an epidemic as frightening and serious as the opioid crisis...