- 3 Reasons Why USP Supports Pharmacist Health Care Provider Status Legislation (ncpanet.org)
For people living in rural areas of the U.S., access to health care providers can be a serious challenge. Pharmacists can be part of the solution. And in a welcome show of bipartisanship, Republicans and Democrats in Congress are co-sponsoring legislation that can help...The Pharmacy and Medically Underserved Areas Enhancement Act...would include pharmacists on the list of recognized health care providers that can be reimbursed by Medicare for providing much needed patient care in underserved areas. USP supports this legislation as a major step forward in improving health care. Here are three reasons why:
- Pharmacists are highly trained health care professionals
- Pharmacists are easily accessible to patients
- Pharmacists improve health care
- Nevada leaders want Heller to explain how key parts of ACA will be replaced if measure is repealed (reviewjournal.com)
The top two Democrats in the Nevada Legislature are calling on U.S. Sen. Dean Heller to explain how Republicans in Congress will replace key provisions of the Affordable Care Act if they repeal it...Aaron Ford and Jason Frierson...sent a letter...posing...questions to Nevada’s senior senator in Congress...the state leaders asked what steps will be taken to ensure people who purchased coverage through the state exchange and those who receive federal tax credits will still be able to buy affordable insurance...what will be done to ensure more than 217,000 Nevadans receiving coverage under expanded Medicaid eligibility will remain covered; guarantees for women’s health care; and continued mandates to cover people with pre-existing conditions and allow parents to keep children on their policies until age 26...“I would anticipate nobody’s going to lose their health care for the next two or three years until the replacement is put in place,” Heller said...
- Why the DEA just said ‘no’ to loosening marijuana restrictions
For the fourth consecutive time, the Drug Enforcement Administration has denied a petition to lessen federal restrictions on the use of marijuana...While recreational marijuana use is legal in four states and D.C., and medical applications of the drug have been approved in many more, under federal law, it remains a Schedule 1 controlled substance, which means it's considered to have "no currently accepted medical use" and a "high potential for abuse."...Just this week, the National Conference of State Legislatures, a group representing state lawmakers, called on the federal government to move marijuana from Schedule 1 to Schedule 2. The group criticized federal law for imposing "substantial administrative and operational burdens, compliance risk and regulatory risk that serve as a barrier to banks and credit unions providing banking services to businesses and individuals involved in the cannabis industry."...Despite this, the DEA says it cannot change the legal status of marijuana unless the FDA determines it has a medical use. The FDA cannot determine it has a medical use in part because of the highly restrictive legal status of the drug. It's a classic bureaucratic Catch-22...The only body that can truly resolve this conflict, now, is Congress — by amending the Controlled Substances Act to treat marijuana differently. Most federal lawmakers seem to agree that this needs to happen, but there's disagreement on how to do it...
- Panel Would Make Insurers Help Contain Rising Drug CostsPanel Would Make Insurers Help Contain Rising Drug Costs (nytimes.com)
An influential federal advisory panel is calling for Congress to force private insurers to rein in rapid increases in prescription drug costs — by cutting some Medicare payments to insurance companies while shielding older Americans from higher out-of-pocket expenses...The recommendations by the nonpartisan Medicare Payment Advisory Commission would squeeze private insurers and drug makers alike, creating strong new incentives for insurance companies to manage the use of prescription medicines by beneficiaries and negotiate larger price discounts with pharmaceutical manufacturers...The recommendations will be included in a June report to Congress, which often heeds the panel’s advice...More than 40 million of the 56 million Medicare beneficiaries have drug coverage through Part D, and national surveys indicate that most are satisfied with the coverage. But out-of-pocket costs for some beneficiaries have increased in recent years as insurers require them to pay 25 percent or more of the cost for expensive specialty drugs to treat chronic or complex illnesses, including cancer, rheumatoid arthritis, multiple sclerosis and hepatitis C...Pharmaceutical companies and advocates for beneficiaries said some of the panel’s recommendations could harm patients’ access to certain medicines and raise costs for some low-income people...
- Sandoval says overhaul of Affordable Care Act remains a top concern for governors (reviewjournal.com)
Nevada Gov. Brian Sandoval said Saturday he would take his concerns about the overhaul of the Affordable Care Act to Capitol Hill where lawmakers are mulling over various proposals to replace Obamacare...The nation’s governors, in town for their annual meeting, are expected to spend Monday talking with Republican leaders and members of Congress about repeal and replacement of the health care act...Many states, including Nevada, broadened Medicaid programs to take advantage of federal funds to help insure more elderly and poor. Officials in those states are concerned about how the federal government will continue its funding commitment for the expanded programs...Sandoval said congressional overhaul of the ACA is a top concern for governors and that the “conversation is ongoing.”...
- Trump should be good medicine for the pharmaceutical industry. Here’s why (statnews.com)
The holidays came early for the pharmaceutical industry this year...With Donald Trump elected to the White House and Republicans controlling Congress, the companies that make your medicines can expect a friendlier environment in which to operate. And investors quickly responded by boosting drug and biotech stocks…So what lies ahead? Here are a few predictions:
- FDA will stand for Faster Drug Approvals - Many Republicans would like to speed the Food and Drug Administration process for getting new medicines to patients.
- Drug makers will have more leeway on pricing - A big bet now is that the Trump administration will not be as aggressive in tackling prescription drug prices as a Clinton administration might have been.
- Friendly tax policies are on the way - Another key piece of the Trump plan is tax reform. Again, the Trump team is short on details, but one plan floating around is to allow companies to repatriate money parked overseas and pay taxes of only 10 percent or less, instead of 35 percent.
- Experts Decry Tying Medical Research Funds to FDA Standards Changes (wsj.com)
Moves in Congress to link billions of dollars in new medical research funding to revised standards for drug and medical-device approvals are troubling some public-health experts, who say the combination makes it too easy for lawmakers to support lower patient-safety standards...These safety advocates say legislation to beef up research funding for the National Institutes of Health should be separated from product-approval changes at the Food and Drug Administration..."This is the first time this has been done this way, and it’s a deal with the devil," said Dr. David A. Kessler, onetime FDA commissioner during the 1990s under presidents of both parties. "It’s time to uncouple the promise of research funding from the requirement that FDA standards be lowered."...To its supporters, linking NIH funding with FDA bills (21st Century Cures Act)—including changes to approvals of antibiotics and some medical devices—produces innovation in life-saving products and research. Politically, it is a classic Washington bargain that has something for everyone...The new FDA commissioner, Dr. Robert M. Califf, said in a recent talk that "this legislation, if not carefully crafted, could pose significant risks for FDA and American patients…Innovative therapies are not helpful to patients if they don’t work, or worse, cause harm," he said...
- Aetna, Humana terminate $37 billion merger (chaindrugreview.com)
After being blocked by a federal court, Aetna Inc. and Humana Inc. have decided to terminate their $37 billion merger deal...The health insurance giants said...that, with the move, Aetna will pay Humana a $1 billion breakup fee...In January...the...District Court for the District of Columbia ruled in favor of the Department of Justice’s request to enjoin the Aetna-Humana merger. The government claimed that the combination of the two health insurers would lessen competition, harming seniors who buy private Medicare coverage and some consumers who buy health insurance on public exchanges...The consolidation wave hitting the health insurance sector has been seen as a byproduct of the Affordable Care Act. The mega-deals would provide fast access to the millions of people who have gained health coverage under the ACA and would be a vehicle for capturing Medicaid business, which has expanded dramatically under the health reform law...the climate for mergers and acquisitions in the health care sector has become uncertain. Besides the DOJ’s opposition to the mega-mergers, the Trump administration and Republican leaders in Congress have begun efforts to dismantle the ACA. That has cast a cloud on the health insurance marketplace, since it remains unclear what would replace the ACA and how consumers who have obtained coverage via the exchanges would continue to receive benefits.
- Drug firms continue to abuse citizen petitions, FDA tells Congress (statnews.com)
...the US Food and Drug Administration filed an annual report to Congress about citizen's petitions that can be used to ask the agency to refrain from approving a generic drug or a biosimilar...FDA officials reiterated complaints that many petitions generally do not raise valid scientific concerns and appear to have been filed to delay approval of competing medicines...Congress...requires the FDA to respond to most petitions within 150 days. And as far as the FDA is concerned, this creates an unnecessary problem...agency officials are concerned that most petitions are merely a competitive ruse, and they wrote Congress that they are forced to redirect efforts at the expense of completing the other work of the agency...The concerns expressed by FDA officials largely mirror a forthcoming analysis...The analysis found that brand-name drug makers filed 92 percent of such citizen petitions between 2011 and 2015...Citizen petitions represent a hidden tool in (the brand-name drug maker's) toolkit of entry-delaying activity, all to the detriment of consumers forced to pay high drug prices...the analysis concludes.
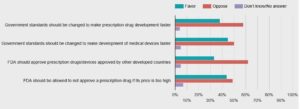
- Public wary of faster approvals of new drugs, STAT-Harvard poll finds (statnews.com)
A majority of Americans opposes federal regulatory changes to speed up the development and approval of new medical treatments, a new STAT-Harvard poll finds — suggesting the public has serious doubts about legislation now moving through Congress...both parties are pushing to change government regulatory standards that they blame for slowing the approval process to get new products to patients...nearly 6 out of 10 Americans said they oppose changing government safety and effectiveness standards to allow for faster approvals of new prescription drugs by the Food and Drug Administration, while 38 percent said they’re in favor of speedier FDA action...The poll sheds new light on how Americans balance their priorities between speed and safety in the approval of new medical treatments.