- Lab-Grown Mini Organs Could Speed Up Drug Discovery (forbes.com)
The thought of lab-grown organs conjures up Frankenstein-like imagery. The reality however, is somewhat less visually dramatic, with the term ‘organoids’ used to describe tiny 3D structures of human tissue, a millimeter or so in diameter...these tiny lumps of cells are creating a lot of excitement in the world of medical research...Cells in dishes and animal models have been used for preclinical testing of drugs for decades. Success in these experiments is a key hurdle for any new medicine to overcome before being given the green light for all-important human clinical trials...Organoids are most commonly made either from a small sample of tissue needled out of a person or from stem cells cultured in a cocktail of nutrients intent on pushing them towards becoming a particular tissue type. So far, organoids have been made resembling several tissues including lung, liver, brain, kidney and intestine...as a relatively new innovation they are being used to investigate dozens of conditions from infectious diseases to cancer.... A study published last year in Science Translational Medicine by scientists at the University Medical Centre, Utrecht generated organoids formed from the rectal tissue of 71 people with cystic fibrosis and exposed them to experimental drugs. By observing changes in the organoids, the scientists accurately predicted which patients would respond to the therapies in just one week at a cost of around $1200 per patient. The results were so convincing that a positive organoid test is now considered sufficient evidence for insurance companies to fund the new therapies in the Netherlands...
- 2015: The Year in Specialty Drugs (specialtypharmacytimes.com)
SPECIALTY PHARMACEUTICALS featured prominently in the FDA’s new drug approval and expanded indications list once again in 2015. Below is the first of a 2-part summary of specialty pharmacy–related FDA approvals and expanded indications that took place this year...Part 2, which will be featured in the next issue of Specialty Pharmacy Times, will include oncology drugs and late-breaking FDA actions...
Bleeding Disorders
- Ixinity (coagulation factor IX (recombinant), Emergent BioSolutions Inc)
- Nuwiq (coagulation factor VIII (recombinant), Octapharma)
- Coagadex (coagulation factor X (human), Bio Products Laboratory Ltd)
- Adynovate (antihemophilic factor [recombinant] pegylated, Baxalta Inc)
Inflammatory Conditions
- Cosentyx (secukinumab, Novartis Pharmaceutical Corp.)
- Humira (adalimumab, AbbVie)
Cystic Fibrosis
- Kalydeco (ivacaftor, Vertex Pharmaceuticals)
- Orkambi (lumacaftor/ivacaftor, Vertex Pharmaceuticals )
HIV
- Evotaz (atazanvir/cobicistat, Bristol-Myers Squibb Co)
- Prezcobix (darunavir/cobicistat, Janssen Therapeutics)
- Genvoya (cobicistat/elvitegravir/emtricitabine/tenofovir alafenamide, Gilead Sciences, Inc)
Hepatitis C
- Technivie (ombitasvir/paritaprevir/ritonavir, AbbVie Inc)
- Daklinza (daclatasvir, Bristol-Myers Squibb)
- Harvoni (ledipasvir/sofosbuvir, Gilead Sciences, Inc.)
Multiple Sclerosis- Glatopa (glatiramer acetate injection, Sandoz, Inc.)
- Betaconnect (Bayer HealthCare’s electronic auto injector - Betaseron (interferon beta-1b)
Specialty Ophthalmics
- Lucentis (ranibizumab, Genentech)
- Eylea (aflibercept, Regeneron Pharm)
Hypercholesterolemia
- Praluent (alirocumab, Sanofi-Aventis)
- Repatha (evolocumab, Amgen Inc)
Miscellaneous Specialty Approvals
- Natpara (parathyroid hormone, NPS Pharmaceuticals, Inc)
- Cresemba (isavuconazonium sulfate, Astellas Pharma US, Inc)
- Cholbam (cholic acid, Asklepion Pharmaceuticals LLC)
- Jadenu (deferasirox, Novartis Pharmaceuticals)
- Anthrasil (anthrax immune globulin intravenous [Human], Cangene Corp)
- Rapamune (sirolimus, Wyeth Pharmaceuticals)
- Promacta (eltrombopag, GlaxoSmithKline and Novartis Pharmaceuticals)
- Envarsus XR (tacrolimus extended-release tablets, Veloxis Pharmaceuticals A/S)
- Dysport (abobotulinumtoxinA, Medicis and Ipsen)
- Keveyis (dichlorphenamide, Taro Pharmaceutical Industries Ltd)
- Procysbi (cysteamine bitartrate, Raptor Pharmaceutical)
- Xuriden (uridine triacetate, Wellstat Therapeutics Corporation)
- Industry’s rare disease moves pay off (epvantage.com)
The pharmaceutical industry’s growing interest in orphan disease shows no sign of diminishing, and for good reason. The increased R&D investment in these indications in recent years has translated into a growing share of overall pharmaceutical sales...The drivers of this change include the favourable regulatory treatment of orphans, as well as market exclusivity and pricing advantages that make such drugs much more profitable than non-orphans. It is notable that the top 25 non-oncology orphan drugs in 2022 are expected to show a compounded growth of 18% over the next six years, almost three times the wider market average...Orphans...remain one of the brightest spots of pharmaceutical development, and the legislation framing them is widely claimed to have been a great success in public policy terms. However, there is a risk that before long orphans could face the same sort of pricing pressure as is common in non-orphan indications...
- 23andMe launches new consumer test service to check for genetic disorders (reuters.com)
23andMe announced the launch of a new consumer genetic test service…that will show whether an individual carries genes associated with 36 different disorders...The new test service…will allow healthy people to see if they carry a genetic variant related to 36 conditions that could be passed on to a child…cystic fibrosis...sickle cell anemia, Tay-Sachs disease and beta thalassemia…The service also provides non-medical details on traits like freckles or hair curliness as well as a person's lactose intolerance….23andMe, which is privately held, is backed by Google Inc and was valued at $1.1 billion in a recent round of venture capital funding.
- 2015: A banner year for personalized medicine (catalyst.phrma.org)
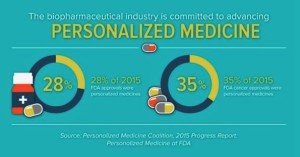
2015 was a record year for personalized medicine approvals, according to a new analysis from the Personalized Medicine Coalition. This news confirms the growing role of personalized medicine as an approach to treatment that can improve outcomes for patients and also create important efficiencies in the health care system. Personalized medicine is an emerging field of medicine that uses diagnostic tools to identify specific biological markers to help assess which medical treatments and procedures will be best for each patient. Personalized medicine also takes into account patients’ medical history, circumstances and values in developing targeted treatment and prevention plans...45 novel new drugs approved in 2015, the new analysis indicates that 28 percent of novel new drugs approved by the Food and Drug Administration...were personalized, or precision, medicines...Some of the personalized medicine highlights from 2015 include:
- Two new medicines for patients with different forms of non-small cell lung cancer;
- A new combination therapy for patients with cystic fibrosis;
- Two new medicines to help patients with a difficult-to-treat form of high cholesterol; and
- A new targeted therapy for melanoma.
- This Medical Charity Made $3.3 Billion From a Single Pill (bloomberg.com)
Cystic Fibrosis Foundation..had…given drugmaker Vertex, around $150 million in exchange for —a share of the royalties for any treatment Vertex’s research yielded…(the foundation) sold its royalty rights..For $3.3 billion.