- U.S. drug agency to ask Congress to classify illicit fentanyl like heroin (reuters.com)
Illicit chemical knock-offs of the extremely potent opioid fentanyl would be put permanently in the same legal class as heroin to boost prosecutions of traffickers and makers of the drugs, under a proposal to be unveiled...by the U.S. Drug Enforcement Administration...The new classification is meant to help fight a proliferation of chemical look-alikes of fentanyl, known as analogues...DEA Acting Chief Operations Officer Greg Cherundolo is set to go before a Senate committee...to propose that Congress make the measure permanent so that cases against various analogues will not be undercut when the temporary ban lapses...READ MORE
- Study links opioid epidemic to painkiller marketing (reuters.com)Association of Pharmaceutical Industry Marketing of Opioid Products With Mortality From Opioid-Related Overdoses (jamanetwork.com)
Researchers are reporting a link between doctor-targeted marketing of opioid products and the increase in U.S. deaths from overdoses...In a county-by-county analysis, they found that when drug companies increased their opioid marketing budgets by just $5.29 per 1,000 population, the number of opioid prescriptions written by doctors went up by 82 percent and the opioid death rate was 9 percent higher a year later...“It really doesn’t take much marketing to increase the number of deaths,” lead author Dr. Scott Hadland...Jordan Trecki of the U.S. Drug Enforcement Administration warns that the new analysis only addresses part of the problem...“As the opioid epidemic grows, it is evolving beyond prescription medications and heroin to involve illicitly produced fentanyl, fentanyl-related substances and other opioids, either alone or in combination,”...
- U.S. judge blocks DEA from suspending drug distributor over opioid sales (reuters.com)
A federal judge blocked the U.S. Drug Enforcement Administration from suspending a Louisiana drug distributor from selling controlled substances over allegations it failed to identify suspicious orders of opioids that were diverted for illicit uses...U.S. District Judge Elizabeth Foote in Shreveport, Louisiana, on Tuesday entered a temporary restraining order blocking the DEA from enforcing an order issued last week that immediately suspended Morris & Dickson Co’s registration...The DEA’s order marked the first time during President Donald Trump’s administration that it had moved to immediately block narcotic sales by a distributor as the agency attempts to combat a national opioid abuse epidemic...The DEA on Friday announced it was suspending the registration of privately-held Morris & Dickson, saying the distributor failed to properly identify large, suspicious orders of drugs sold to independent pharmacies.
- DEA proposes cutting production of some opioid painkillers (reuters.com)
The Drug Enforcement Administration...proposed a 20 percent reduction in the manufacture of certain commonly prescribed opioid painkillers as well as other controlled substances for next year...The proposal comes as U.S. regulators and lawmakers take steps to limit the supply of opioids - a class of drugs that include prescription painkillers and heroin - to combat the epidemic of abuse, overdose and addiction...The DEA's proposed production quotas for Schedule I and II substances reflect the amount needed to meet the United States' medical, scientific, industrial, export and reserve requirements...The DEA recommendation comes about two months after the Food and Drug Administration took the rare step of asking a drugmaker (Endo International) to withdraw its opioid painkiller from the market, citing the public health crisis.
- Big drug distributor pays $22 million to settle U.S. opioid charges (reuters.com)
Morris & Dickson Co, one of the largest U.S. wholesale drug distributors, agreed to pay $22 million in civil penalties to settle U.S. government charges that it failed to report thousands of suspicious orders of the opioids hydrocodone and oxycodone...The...Louisiana-based company will also spend millions of dollars to hire staff and upgrade oversight to help comply with federal regulations requiring that orders be properly reported...Drug Enforcement Administration...since...2014 uncovered more than 12,000 retail pharmacy orders for hydrocodone and oxycodone that Morris & Dickson should have flagged to that agency...READ MORE
- Congressional report: Drug companies, DEA, failed to stop flow of millions of opioid pills (washingtonpost.com)Committee Report Details Alleged Opioid-Dumping in West Virginia (energycommerce.house.gov)Red Flags and War ning Signs Ignored: Opioid Distribution and Enforcement Concerns in West Virginia (energycommerce.house.gov)
A report from the majority staff of the House Energy and Commerce Committee found that distributors, which fulfill orders for prescription drugs to pharmacies, failed to conduct proper oversight of their customers by not questioning suspicious activity and not properly monitoring the quantity of painkillers that were being shipped to individual pharmacies...The committee also found that the DEA did not properly use a database that aims to monitor the flow of powerful prescription painkillers from manufacturers to sellers, something that could have allowed federal agents — in real time —...The agency also curtailed enforcement of distributors...and infighting inside the agency affected the way cases were handled...
- DEA to share prescription drug data with 50 attorneys general, crack down on drugmakers (fiercehealthcare.com)
The Drug Enforcement Agency has reached an agreement with 50 attorneys general to share prescription drug data with one another to support ongoing investigations...from its Automation of Reports and Consolidated Orders System, which collects 80 million prescription drug transactions from manufacturers and distributors each year...Attorney General Jeff Sessions...said the data-sharing pact will “make both the DEA and our state partners more effective at finding evidence of crime.”...Exactly how that data sharing agreement would operate remains fuzzy...
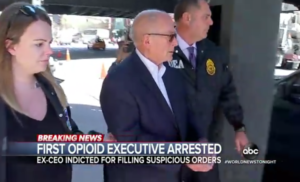
- Feds charge Rochester Drug Cooperative and CEO in first criminal case over opioids (abcnews.go.com)
Federal prosecutors charged drug distributor Rochester Drug Cooperative and its former CEO with drug trafficking charges...the first criminal charges for a pharmaceutical company and executives in the nation's ongoing opioid crisis... This prosecution is the first of its kind: executives of a pharmaceutical distributor and the distributor itself have been charged with drug trafficking...The U.S. Attorney's Office for the Southern District of New York charged Rochester Drug Cooperative...with "knowingly and intentionally" violating federal narcotics laws "by distributing dangerous, highly addictive opioids to pharmacy customers that it knew were being sold and used illicitly,"...RDC was also charged with failing to properly report thousands of suspicious orders of oxycodone, fentanyl and other controlled substances to the Drug Enforcement Agency...READ MORE
- HHS recommended that the DEA make kratom a Schedule I drug, like LSD or heroin (statnews.com)
The Department of Health and Human Services has recommended a ban on the chemicals in kratom that would make the popular herbal supplement as illegal as heroin or LSD…HHS asserted in a letter to the Drug Enforcement Administration that two chemicals in kratom should be classified as Schedule I substances... FDA...has said that kratom is “an opioid” and has been “associated” with dozens of deaths...Kratom should not be used to treat medical conditions, nor should it be used as an alternative to prescription opioids...Some states have already banned kratom, but it’s currently legal at the federal level. It’s sold in different forms, including dry powder and capsules. According to the American Kratom Association, millions of Americans use the substance.
- DEA, Dutch law enforcement continue attack on dark web drug sales (dea.gov)
The United States Drug Enforcement Administration and Dutch law enforcement officials...announced sustained action against drug trafficking on the dark web, following last summer’s significant market takedowns of AlphaBay and takeover and subsequent takedown of the Hansa market. DEA continued to partner with the National Police of the Netherlands...to identify individuals who purchase drugs on the dark web and to further disrupt dangerous drug trafficking. Further examination of the Hansa Market data revealed illicit drug purchase information identifying U.S. and Dutch individuals, resulting in numerous face-to-face doorstep visits by police to suspected opioid buyers throughout the U.S. and the Netherlands...AlphaBay operated as a hidden service on the “Tor” network, and utilized cryptocurrencies including Bitcoin, Monero and Ethereum in order to hide the locations of its underlying servers and the identities of its administrators, moderators, and users. Based on law enforcement’s investigation of AlphaBay, authorities believe the site was also used to launder hundreds of millions of dollars deriving from illegal transactions on the website...the Hansa Market, another prominent dark web market...was used to facilitate the sale of illegal drugs, toxic chemicals, malware, counterfeit identification documents, and illegal services. The administrators of Hansa Market, along with its thousands of vendors and users, also attempted to mask their identities to avoid prosecution through the use of Tor and digital currency.