- Experts Decry Tying Medical Research Funds to FDA Standards Changes (wsj.com)
Moves in Congress to link billions of dollars in new medical research funding to revised standards for drug and medical-device approvals are troubling some public-health experts, who say the combination makes it too easy for lawmakers to support lower patient-safety standards...These safety advocates say legislation to beef up research funding for the National Institutes of Health should be separated from product-approval changes at the Food and Drug Administration..."This is the first time this has been done this way, and it’s a deal with the devil," said Dr. David A. Kessler, onetime FDA commissioner during the 1990s under presidents of both parties. "It’s time to uncouple the promise of research funding from the requirement that FDA standards be lowered."...To its supporters, linking NIH funding with FDA bills (21st Century Cures Act)—including changes to approvals of antibiotics and some medical devices—produces innovation in life-saving products and research. Politically, it is a classic Washington bargain that has something for everyone...The new FDA commissioner, Dr. Robert M. Califf, said in a recent talk that "this legislation, if not carefully crafted, could pose significant risks for FDA and American patients…Innovative therapies are not helpful to patients if they don’t work, or worse, cause harm," he said...
- FDA Approves New Version of Oxycodone (painnewsnetwork.org)
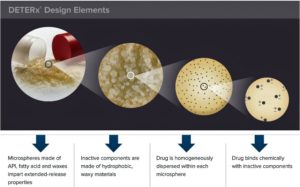
Food and Drug Administration has approved a new extended released version of the opioid painkiller oxycodone that has abuse deterrent properties unlike anything else on the market...Xtampza – can be ingested in capsule form, but users can also sprinkle the capsule contents on soft foods or into a cup, and then directly into the mouth...The medication, which can also be ingested through a feeding tube, is the sixth opioid pain medication with an abuse deterrent formula to be approved by the FDA...Xtampza is made by...Collegium Pharmaceutical with proprietary technology (DETERx technology platform) that combines oxycodone with fatty acid and waxes to form small spherical beads that are placed inside the capsule...The beads are designed to resist breaking, crushing, chewing, dissolving and melting, methods long used by drugs abusers to snort or inject opioids.
- The FDA Wants Pharma to Ditch its Archaic Drug Making Process (fortune.com)
The old process is slow and prone to errors...The process of biopharmaceutical drug manufacturing is stuck in the past. And the Food and Drug Administration is now openly calling for drugmakers to spring it forward into the 21st century..."batch" manufacturing technique used by the industry to an archaic relic...batch manufacturing in pharma involves regular breaks between spurts of production. Continuous manufacturing is usually a persistent, unbroken process wherein production plants keep humming... continuous manufacturing is more reliable...the (FDA) agency’s pioneering decision last week to approve Johnson & Johnson biotech arm Janssen’s request to switch over from batch to continuous manufacturing for the production of the HIV drug Prezista. And now, regulators are declaring outright that other biopharma players should "consider similar efforts."
- Drug makers inconsistently report side effects in the US and Europe (statnews.com)Variation in adverse drug reactions listed in product information for antidepressants and anticonvulsants, between the USA and Europe: a comparison review of paired regulatory documents (bmjopen.bmj.com)
Drug makers must report side effects to regulators, but the amount of useful information can vary depending upon which regulator gets the data, according to a new analysis...A team of researchers examined a dozen brand-name drugs used to treat depression and seizures and found that, on average, the manufacturers filed 77 more side effects reports with the US Food and Drug Administration than with the European Medicines Agency...On average, 71 percent of the side-effect terms listed in the reports submitted to the FDA were not filed with the EMA...86 percent of reports submitted to the FDA, there was more information on the source of the report — such as a spontaneous patient report during treatment or if it occurred during a trial — than what was provided to the EMA...On average, only 29 percent of the side effects were reported to both agencies. Moreover, most of the reports lacked information on duration and seriousness of the side effects, as well as the extent to which the reactions were reversible...that reporting needs to be improved...This study shows that the reporting of drug adverse effects on product information is fragmentary and inconsistent...neither the FDA nor the EMA versions provided much information about the onset, duration, and severity of drug side effects. Instead they provided just lists that were not necessarily in agreement...
- FDA Advises Against Fluoroquinolone Antibiotic Use for Uncomplicated Infections (pharmacytimes.com)
Serious side effects associated with fluoroquinolones generally outweigh the antibiotics’ benefits for patients with uncomplicated infections like sinusitis and bronchitis…the FDA determined that systemic use of fluoroquinolones is associated with "disabling and potentially permanent serious side effects that can occur together" involving the tendons, muscles, joints, nerves, and central nervous system...The alert comes a year after the FDA convened an advisory committee to review the risk-benefit balance for fluoroquinolones...the risk wasn’t worth the potential benefit for patients with sinusitis, bronchitis, and uncomplicated urinary tract infections, for which other treatment options exist...Labels and medication guides for all fluoroquinolones will be updated to reflect the FDA’s findings...
- FDA reconsiders training requirements for painkillers (hosted.ap.org)
Food and Drug Administration is reconsidering whether doctors who prescribe painkillers like OxyContin should be required to take safety training courses...The review comes as regulators disclosed that the number of doctors who completed voluntary training programs is less than half that targeted by the agency...Under the current risk-management programs, drugmakers fund voluntary training for physicians on how to safely prescribe their medications...many experts - including a previous panel of FDA advisers - said those measures don't go far enough and that physician training should be mandatory...The FDA's initial ideas to improve safety included mandatory certification for doctors and a national registry to track patients taking the drugs. But industry pushed back. Drugmakers and the pain specialty groups they fund argued that certification would be too burdensome for doctors, leaving many patients undertreated. And patient groups said that registries would unfairly stigmatize those who rely on painkillers to deal with long-term pain....
- Biosimilars gain momentum — and pharma leaders are noticing (biopharmadive.com)
Biosimilar development in the U.S. appears to be gathering momentum, following the approval of Celltrion and Pfizer’s Remicade copy by the Food and Drug Administration in early April...The drug, marketed as Inflectra, is only the second biosimilar to clear regulatory review in the U.S., after Sandoz’s Zarxio in March 2015. But there are a number of other biosims entering late-stage development or with applications filed at the FDA... And with several blockbuster biologics nearing patent expiry, competition is likely to increase...Biosimilars promise to increase competition and lower prices across a number of other blockbuster biologics. They also represent new avenues of growth for companies seeking to enter previously protected markets...There is still a long way to go before biosimilars have anywhere near the market impact of generic drugs in the U.S...Biosimilars are still very much in early stages domestically...biosimilars look set to present a number of important opportunities and challenges to the U.S. biopharma landscape...
- Ex-FDA head and Sanofi call for harmonized drug regulation (reuters.com)The need for global regulatory harmonization: A public health imperative (stm.sciencemag.org)
Drug regulation has failed to keep up with a globalized world and governments should harmonize oversight to improve patients' access to new and innovative medicines...That is the view of the former leader of the U.S. Food and Drug Administration, the world's top drug regulator, and the research head of French drugmaker Sanofi, who made a joint plea to governments for action...Margaret Hamburg, who led the FDA until 2015, and Elias Zerhouni said there was an urgent need to harmonize a "mosaic of regulations" in different countries, and they called for the issue to be taken up at the G8 or G20 groups of nations..."Essentially, it is a hidden bureaucratic inefficiency tax on the whole effort of finding new and valuable therapies," he said in a telephone interview...Drug development is global and we need to have safety and efficacy data globally, so we should have a global system, just like with airplanes...Sanofi was spending around 20 percent of its research and development budget on ensuring convergence between different systems, often involving duplication of efforts.
- New implant set to join fight against U.S. painkiller epidemic (reuters.com)
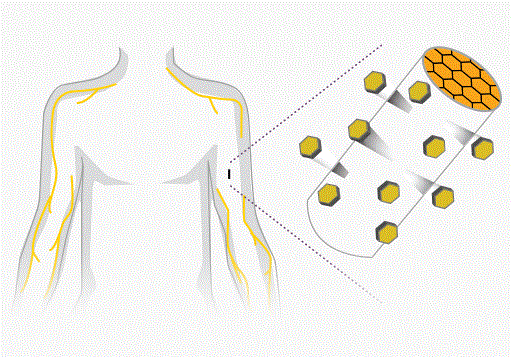
Two companies are on the cusp of taking a new treatment for opioid addiction to the U.S. market at a time when lawmakers are seeking ways to arrest an epidemic of heroin and painkiller abuse...Titan Pharmaceuticals Inc and...Braeburn Pharmaceuticals have together developed a matchstick-sized implant that analysts expect will be approved next month, despite mixed reviews...Implanted into the arm, the treatment is designed to be less vulnerable to abuse or illicit resale than the oral drugs that are currently used to treat opioid addiction...Two drugs are predominantly used to treat opioid addiction: methadone, which is dispensed only in government-endorsed clinics, and the less-addictive buprenorphine, which exists as a pill or strip of film...The implant, known as Probuphine, offers an alternative by administering buprenorphine for up to six months after users have first been stabilized on the oral form of the drug...Food and Drug Administration have raised reservations about possible complications from the insertion and removal of the 26mm long implant...
- Drug firm to pay $4m to settle investor fraud charges (statnews.com)
Aveo Pharmaceuticals agreed to pay $4 million to settle charges that it misled investors about efforts to obtain regulatory approval for its flagship drug, a kidney cancer treatment called Tivozanib, the US Securities and Exchange Commission said today...the agency is still pursuing a case against three former executives...The biotech allegedly concealed concerns that the Food and Drug Administration had about the medicine in public statements to investors. In particular...failed to disclose that FDA staff had recommended...that the company should run a second clinical trial to address issues concerning patient death rates that were seen during an earlier clinical trial...A spokesman for Aveo..."we hope to have this matter behind us and to be able to pursue our new strategy without being distracted by these claims" The new strategy...includes running a second Phase III study for the...drug...