- FDA announces enhanced warnings for immediate-release opioid pain medications related to risks of misuse, abuse, addiction, overdose and death (fda.gov)
New safety warnings also added to all prescription opioid medications to inform prescribers and patients of additional risks related to opioid use...Food and Drug Administration...announced required class-wide safety labeling changes for immediate-release opioid pain medications...the FDA is requiring a new boxed warning about the serious risks of misuse, abuse, addiction, overdose and death...actions are among a number of steps the agency recently outlined in a plan to reassess its approach to opioid medications. The plan is focused on policies aimed at reversing the epidemic, while still providing patients in pain access to effective relief.
- VA expands hepatitis C treatment to all patients with the virus (militarytimes.com)
Veterans Affairs Department will begin providing hepatitis C treatment to all veterans in its health system who have the virus, regardless of their disease stage...Having received a boost in funding from Congress late last year for the costly medications needed to cure hepatitis C, the VA is now able to treat the 174,000 veterans in its health system who have the disease…The Food and Drug Administration in January approved Zepatier (elbasvir and grazoprevir), made by Merck, to treat the disease...executives said they priced the medication to broaden and accelerate access to treatment for patients covered in commercial or public plans, including our country’s veterans...This is a good example of how government and industry can work together toward a shared goal in the best interests of public health — particularly for our veterans who are so deserving...Merck spokeswoman said it was too early to tell whether Zepatier will become the favored treatment within VA...but that the company priced it appropriately to ensure that it could be accessed by all veterans.
- FDA still struggling with backlog of generic drug applications (statnews.com)
Under pressure to speed approval of generic medicines, the Food and Drug Administration...released data to defend its progress...the statistics indicate the agency is making headway, there are also clear signs the FDA continues to struggle with the workload...the number of full and tentative drug approvals has been rising each month since last April and reached 99 this past December...the agency also appears to be doing a better job of communicating with generic drug makers about their applications...Generic drug approval is gaining more attention thanks to the intensifying national debate over the rising cost of prescription medicines. Although prices have also risen for some of these copycat medicines, generics remain...lower-cost alternatives to brand-name drugs. And generics now account for 88 percent of all prescriptions written...The FDA is being a little disingenuous saying its backlog is almost cleared...The FDA faces...the increasingly large number of applications that drug makers are submitting...more than 4,000 have been filed in the past four years...the FDA workload will not abate...The upshot is that the rate at which new generics will find their way to pharmacy shelves is unclear — and that adds further uncertainty for health care budgets...
- Health Officials Urge FDA To Add Black Box Warning On Opioids, Benzos (forbes.com)
City and health directors from across the country are urging the Food and Drug Administration to adopt new labels to "explicitly warn about a dangerous combination of medications" that have been fueling the nation’s prescription drug overdose epidemic over the years...Available online, the petition, which has been signed by academics, researchers and physicians, requests that the FDA amend black box warnings on all opioid analgesic and benzodiazepine class medications...to require medication guides that specifically warn patients of concurrently using both drug classes...We believe that this black box warning is a critical first step in raising awareness around the danger of co-prescribing these medications…The warning educates doctors so that they can provide the highest quality of care to patients...
- FDA changes policy to prevent the next Martin Shkreli (statnews.com)
...the Food and Drug Administration...made a policy change that may prevent companies from pulling a Martin Shkreli...The agency plans to expedite reviews of applications for generic drugs where only one treatment is currently sold. The shift was prompted by public outrage that erupted last fall when Turing Pharmaceuticals...bought a life-saving drug called Daraprim and promptly jacked up the price by 5,000 percent...We identified a gap and were able to identify a path forward...The change being made (allows the agency) to capture circumstances when the only approved product on the market is a generic drug...Even though Turing does not hold a patent on the medicine...company was able to increase the price as it did because there was no generic competition. The drug maker runs a closed distribution system, and as a result, Turing has a monopoly on Daraprim...This should provide a faster way to inject competition in the marketplace, so that the price gougers can’t get away with what they’re doing...the policy change is retroactive, which means the agency will review pending applications to see if any merit an expedited review.
- FDA Settles With Drugmaker in Fish-Oil Drug Marketing Case (abcnews.go.com)FDA deal with Amarin is unlikely to spark more off-label promotion (statnews.com)
The maker of a prescription fish-oil drug says it has reached a legal settlement that will allow it to promote unapproved uses of its drug for lowering fat levels...The closely watched case between Amarin and the Food and Drug Administration could strengthen the drug industry's hand in the ongoing debate over promoting drugs for uses that have not been declared safe and effective by regulators...FDA said...the settlement is "specific to this particular case and situation," and did not mark a new legal precedent...pharmaceutical experts said companies would likely pursue more aggressive legal action against FDA, in light of the settlement...We would expect companies throughout the country to ask courts to provide the same legal reasoning...Amarin won a surprise victory over the FDA when a U.S. District Court judge ruled that the company had a First Amendment right to distribute journal articles about unapproved indications for Vascepa (icosapent)...Drugmakers are not allowed to advertise drugs for "off-label" uses, or those that have not been cleared by the FDA as safe and effective. But companies' ability to distribute independent materials about their drugs — such as medical journal articles — has been subject to years of legal debate centering around the limits of "commercial speech."
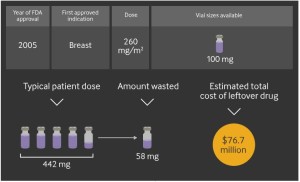
- Study: $3B will be wasted on unused portion of cancer drugs (cnbc.com)Overspending driven by oversized single dose vials of cancer drugs (bmj.com)
High prices for cancer medicines aren't the only reason they cost insurers and patients so much...Waste pads the bill…because infused cancer drugs are distributed in...vials that usually contain more medicine than most patients need. Most of the time that excess is thrown out, even though it's perfectly good — and worth hundreds or thousands of dollars...Researchers...estimate that wasted cancer medicine in the U.S. this year will add up to nearly $3 billion in excess costs...cancer medicines are one of the highest-priced, fastest-growing drug categories...study...in BMJ...details how drugmakers, hospitals and cancer doctors make money on unused cancer medicine. It focuses on the top 20 drugs for multiple cancer types packaged in single-dose vials and for which the dose depends on the patient's weight, finding that 1 percent to 33 percent of those 20 cancer drugs, on average, remains in vials after each dose is administered...The researchers say regulators could require manufacturers to supply multiple vial-size options, and the FDA and other federal health agencies need to reconcile their disparate guidelines on vial size and when leftover medicine from one vial can be given to another patient. Currently, sharing generally is only allowed within six hours of when the vial is opened to ensure the medicine's sterility.
- FDA warns India’s Emcure Pharma, cites repeated data fudging at plant (reuters.com)
Food and Drug Administration has warned Indian generic drugmaker Emcure Pharmaceuticals, saying it repeatedly fudged test records at its plant in western India, in another case of a pharmaceutical firm in the country facing such action...the FDA...found "significant violations" of standard manufacturing practices...The agency had already banned imports from the plant...except for some drugs...It is one of 42 drug-making factories in India that the FDA has banned in recent years as it stepped up inspections of foreign suppliers. The increased scrutiny has hit growth at Indian companies the hardest, as the country supplies nearly 40 percent of the medicines sold in the United Stares...We observed multiple examples of incomplete, inaccurate, or falsified laboratory records...The fabricated records were of tests that Emcure was required to conduct to ensure proper environmental control was maintained while aseptic filling of drug batches, so that the products wouldn't become contaminated...The company has 15 days to respond to the FDA's letter on the corrective actions it will take on the concerns raised.
- US FDA backlog holding up revamp of inactive ingredients database (in-pharmatechnologist.com)
The process of sorting out problems with the US FDA's controversial Inactive Ingredients Database is shaping up to be a huge task, particularly as the agency is working through a backlog of missing updates extending back several years...The IID is a listing of inactive ingredients found in FDA-approved drug products, and is meant to be updated in a consistent manner as new medicines and their excipients are given the go-ahead by the regulator...it emerged recently that - from around 2005 - FDA resource constraints meant that the IID was not being updated at all with new excipient listings, and was left in hiatus for around a decade...If the database is not reliable, the review times for new medicines can be extended and may lead to applications being refused, according to excipient trade body IPEC-Americas...The agency is now working hard to work through the backlog of applications...There are still major concerns about the quality of the data in the IID...the IID has become peppered with inaccurate information that has compromised its integrity, including inaccurate ingredient names and potencies and ingredients listed as a percentage with no indication of basis units...Overall, the FDA says it is aiming to transform the IID into a complete, fully-searchable database that will link into other databases on nomenclature, toxicology etc and accommodate electronic submissions, to eliminate data entry errors and potentially allow it to be updated in real time...
- State oversight of compounding pharmacies varies dramatically (statnews.com)
Amid ongoing safety concerns over compounding pharmacies, a new analysis finds state oversight is a spotty patchwork of regulations, raising questions about the ability to protect the public health.
- Only about half of the states require compounding pharmacies that make sterile medicines, which are injected or infused into the body, to fully comply with recognized quality standards.
- 60 percent of the states do not require compounding pharmacies to report serious adverse events and reactions related to sterile compounding...
- 16 states reported they do not track compounding pharmacy activity at all.
- 28 states allow traditional compounding pharmacies as opposed to those that make large amounts of medicines for hospitals to provide drugs without prescriptions for individual patients. This practice conflicts with??federal law.
- nine states do not plan on taking any action, such as requiring compounders to register with the FDA or disciplining these businesses.
- 53 percent of the states conduct annual routine inspections for in-state pharmacies that perform sterile compounding.
- any issues discovered during an inspection, 79 percent of the states requires a written response from the pharmacy describing how the issues were addressed,
- 67 percent also need an additional on-site inspection to verify compliance.
- one-third of the states do not perform on-site follow-up inspections to verify needed changes.
- Most of the states...have the authority to issue cease-and-desist orders, but 70 percent also said they were unable to mandate a recall of compounded drugs or were unsure of their explicit authority.