- FDA advisory panel strongly backs biosimilar Remicade (reuters.com)FDA Briefing Document Arthritis Advisory Committee Meeting February 09, 2016 (fda.gov)
A medical advisory panel to the Food and Drug Administration...recommended approval of a cheaper biosimilar form of Johnson & Johnson's Remicade (infliximab) arthritis drug that could eventually batter sales of the branded product...The panel, by a vote of 21-3, supported use of the biosimilar from Celltrion Inc and Pfizer Inc, called Remsima. The FDA usually, but not always, follows the advice of its advisory panels...The independent panel determined that clinical trials of Remsima showed no clinically significant differences with Remicade in treatment of rheumatoid arthritis and a related condition called ankylosing spondylitis...Moreover, the panel agreed Remsima is likely as safe and effective for other conditions Remicade treats, including psoriasis and inflammatory bowel conditions like Crohn's disease and ulcerative colitis, even though Remsima was not tested against those conditions.
- Payers see price leverage with entry of Merck hepatitis C drug (reuters.com)
...pharmacy benefit managers expect the launch of Merck & Co Inc's new hepatitis C pill to improve their leverage in price negotiations with drugmakers...The Food and Drug Administration...approved Merck's Zepatier (elbasvir and grazoprevir) for treatment of patients infected with the most common form of the liver-destroying virus, genotype 1, as well as the less common genotype 4...The list price for the new drug is $54,600 for a 12-week regimen - compared with $94,500 for Gilead Sciences Inc's Harvoni. A multi-pill regimen, Viekira Pak, from AbbVie Inc has a list price near $83,000..."We look forward to working with Merck," Express Scripts said..."Having multiple, clinically effective options allows us to again leverage competition and make medicine more affordable for our clients while ensuring appropriate patient access."..."Given Merck's interest in participating in such a large market, we model and fully expect increased price competition and we also view Merck's list price as a rational way to stay out of the drug pricing spotlight,"...
- Deficiencies Found at Theranos Lab (wsj.com)
Federal inspectors will soon release details on problems at blood-testing facility...U.S. health inspectors have found serious deficiencies at Theranos Inc.’s laboratory in Northern California...The problems were found during an inspection by the Centers for Medicare and Medicaid Services, the chief federal regulator of clinical labs, at the blood-testing company’s facility in Newark, Calif. Failing to fix the problems could put the Theranos lab at risk of suspension from the Medicare program...inspection results are expected to be publicly released soon...
- Express Scripts sued by compounding pharmacies for alleged antitrust practices (statnews.com)
For the second time since Express Scripts began blocking coverage of hundreds of ingredients used to make compounded medicines, several compounding pharmacies have filed a lawsuit accusing the pharmacy benefits manager of using illegal tactics...In the latest instance, a half-dozen compounding pharmacies have charged Express Scripts with violating antitrust laws and is attempting to force them out of business...Express Scripts has taken “a series of unreasonable restrictions and rules that would make it impossible for [the compounding pharmacies] to fill prescriptions” for patients “and obtain reimbursements that would cover their costs,”...The company and the other benefits managers “employed tactics designed to ensure that the compounding pharmacy industry …cannot survive.”...The move to cut back on covered ingredients has riled compounding pharmacies...the Food and Drug Administration has cracked down on compounding pharmacies by increasing the number of inspections, and, in rare cases, taking legal action to halt allegedly unsafe practices. The justifiable emphasis on safety has forced many compounding pharmacies to enhance operations...The compounding pharmacies are striking back. In November 2014, three others filed a lawsuit claiming Express Scripts illegally blocked legitimate prescriptions and unfairly forced patients to seek more expensive medicines or simply not seek treatment. The pharmacies maintained the benefits manager violated federal law because it lacks authority to essentially alter terms of health plans.
- Washington’s Prescription For Cheaper, And Unregulated, Medicines (forbes.com)
In 2011, in an effort to bring a lower-cost drug to the market, the Food and Drug Administration was forced to accept the widespread compounding of a specialty drugs...The agency’s submission involved an old, and previously cheap, generic medicine that had secured some renewed exclusivity, and was being sold as a higher-priced specialty drug. But the FDA’s move deliberately undermined the agency’s own approval requirements...It sent an unambiguous message that FDA wouldn’t try to enforce its already weakened authority over compounding–so long as the price of the knock-off drug was right...The compounding of drugs grew precipitously over the next few years...the passage of the 2013 Drug Quality and Security Act...closed loopholes that allowed compounding outfits to operate beyond the bounds envisioned when the local practice of pharmacy was carved out from typical FDA oversight...some are seeking to co-opt some key provisions in that law as a way to once again expand compounding well beyond the practices’ historical concept. Once again, these efforts are being insincerely waged in the name of increasing drug competition and lowering costs…The co-opting of the practice of pharmacy compounding in this manner also puts the entire practice of pharmacy at risk. Compounding remains an important pharmacy activity...But each time outfits try to exploit the practice simply as a way to evade traditional regulation of drug manufacturers; they create risks and bad outcomes that ultimately lead to new restrictions. That was the legacy of the 2013 passage of the DQSA. Now some who instigated that law want to do it all over again.
- FDA finds Indian drug maker Wockhardt hid failed tests (reuters.com)
Indian drugmaker Wockhardt hid the results of failed tests and deleted data from its systems at a plant in western India, according to a report by the U.S. Food and Drug Administration...Issues around "data integrity", maintaining accurate and consistent databases, are key to the U.S. watchdog, which regulates the world's largest market for generics producers...Wockhardt is the latest of several major players in the $15 billion Indian drugs industry to be hit by U.S. regulatory action over the past few months...It makes around a fifth of its $670 million in annual revenues from the United States...FDA inspectors also reported finding pharmaceutical ingredients that were not stored or labeled properly. A rejected drug batch was stored in the "approved material" area, and some batches did not carry expiry dates, the report said.
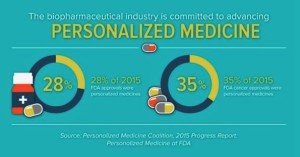
- 2015: A banner year for personalized medicine (catalyst.phrma.org)
2015 was a record year for personalized medicine approvals, according to a new analysis from the Personalized Medicine Coalition. This news confirms the growing role of personalized medicine as an approach to treatment that can improve outcomes for patients and also create important efficiencies in the health care system. Personalized medicine is an emerging field of medicine that uses diagnostic tools to identify specific biological markers to help assess which medical treatments and procedures will be best for each patient. Personalized medicine also takes into account patients’ medical history, circumstances and values in developing targeted treatment and prevention plans...45 novel new drugs approved in 2015, the new analysis indicates that 28 percent of novel new drugs approved by the Food and Drug Administration...were personalized, or precision, medicines...Some of the personalized medicine highlights from 2015 include:
- Two new medicines for patients with different forms of non-small cell lung cancer;
- A new combination therapy for patients with cystic fibrosis;
- Two new medicines to help patients with a difficult-to-treat form of high cholesterol; and
- A new targeted therapy for melanoma.
- We Can Beat Zika And Malaria–If The FDA Allows (forbes.com)The Emerging Zika PandemicEnhancing Preparedness (jama.jamanetwork.com)
Zika virus infection, the scary new disease for which there is no vaccine or treatment, is “spreading explosively” from Africa and Southeast Asia...The United States and 20 other countries...have reported cases of the virus since Brazil reported the first cases of local transmission last May. Delivered by varieties of mosquitoes...it has boosted interest in mosquito-borne diseases...What’s needed is...modern genetic engineering techniques to more effectively prevent mosquitoes from delivering the viruses and parasites that cause disease….The FDA has long delayed the approval of a November 2011 application for a field trial to test a new biological control agent for the mosquito species Aedes aegypti. Although that field trial is concerned specifically with dengue fever, A. aegypti...also transmits Zika...Oxitec has created a new way to control Aedes aegypti. Male mosquitoes are bred in the laboratory with a specific genetic mutation that, in the absence of a certain chemical, causes their offspring to die before reaching maturity...This safe and effective control technique has been approved in Brazil and open field trials of these mosquitoes have been conducted in Brazil, the Cayman Islands, Panama and Malaysia...Eight months have passed since FDA promised last May to publish for public comment a routine environmental assessment of the Oxitec field trial in Florida. Only after FDA reviews the comments will FDA consider whether to grant approval. This delay is unnecessary and unconscionable.
- Ed Markey blocks vote on FDA chief over opiate approvals (bostonglobe.com)Three senators now holding up FDA nominee — for three different reasons (washingtonpost.com)
Senator Edward Markey is using Senate rules to block the nomination of a proposed new leader of the Food and Drug Administration in an attempt to force the agency to rescind its approval of a prescription opioid for children and change its regulatory practices...Using parliamentary procedures, the Massachusetts Democrat has put a "hold" on the nomination of Dr. Robert Califf, which prevents the Senate taking a confirmation vote...Markey, who has made the growing wave of opioid addiction one of his legislative priorities, is demanding that the FDA agree to reverse its 2015 decision allowing the pediatric use of the prescription painkiller OxyContin. He also wants the agency to commit to convening expert advisory panels to provide advice whenever considering the approval of an opioid drug, and to ensure that the risks of drug addiction and abuse are taken into account whenever the agency considers approving a prescription opioid...
- Licorice Coughing Liquid Recall, Presence of Morphine (infozine.com)Master Herbs, Inc. Issues Voluntary Nationwide Recall of All Lots of Licorice Coughing Liquid Due to the Presence of Morphine (fda.gov)
Master Herbs, Inc. (Ma Ying Long Pharmaceutical Group) is voluntarily recalling ALL LOTS of Licorice Coughing Liquid, cough syrup in 100 ml bottles to the consumer level. This product has been found to contain morphine, which is an opioid, and it is not declared on the label. Opioid is an ingredient of Compound Camphor. Compound Camphor is declared on the label of the product, but not its ingredients...The product is used for the temporary relief of cough due to cold, minor throat and bronchial irritations...identified by the Chinese Product Name: Licorice Coughing Liquid The product was distributed to Chinese grocery stores in various cities in California, New Jersey, Hawaii, Illinois, Ohio and Nevada.