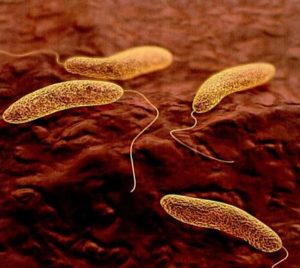
- Cholera Vaccine OK’d (medpagetoday.com)FDA Approves Vaxchora, PaxVax’s Single-Dose Oral Cholera Vaccine (paxvax.com)
First vaccine against lethal infection was 80%-90% effective in challenge study...A vaccine against cholera has, for the first time, received FDA approval...To be sold as Vaxchora, the product contains live but attenuated Vibrio cholerae bacteria. It is taken as a single oral liquid dose... the vaccine is indicated primarily for people traveling to areas where the infection is endemic...Vaxchora is manufactured by PaxVax Bermuda...
- Zika update: Vaccine race swells, PaxVax CEO on how to stop ‘chasing epidemics’
As the Zika virus continues to spread, more biotechs are announcing their Zika vaccine programs. Meriden,.. Protein Sciences,..GeoVax Labs and.. PaxVax are the latest...Getting caught off-guard by epidemics like this has happened time and time again. And "chasing" outbreaks instead of anticipating them rarely results in a vaccine being developed in time. Witness the most recent Ebola epidemic: Merck's experimental vaccine, the furthest along in a crowded field, won't be submitted for regulatory approval until 2017, more than two years after the outbreak started...we had known about Ebola for decades...Companies got a head start in 2014 from partly developed candidates that had been shelved away. It is not so with Zika. "Almost everyone is pretty much starting from scratch...To avoid this and have programs in place before an outbreak hits...governments and nongovernmental organizations...should create economic incentives for companies to make vaccines for neglected diseases like Zika...the FDA's priority review voucher system, in which a company developing a vaccine for a neglected tropical disease receives a transferable voucher for expedited FDA review. Malaria and dengue have been on the list of neglected diseases for years, but Zika is not yet on the list