- This Week in Managed Care: January 13, 2017 (ajmc.com)
Laura Joszt, assistant managing editor at The American Journal of Managed Care. Welcome to This Week in Managed Care from the Managed Markets News Network
- IBM Watson and FDA to develop secure ‘blockchain’ patient data sharing (pharmaphorum.com)
IBM Watson is to work with the FDA to investigate the use of blockchain technology, seen as one of the most secure ways of sharing patient data...(it) allows each separate patient data source to be a ‘block’ part of a complete, unalterable patient data profile which can then be shared securely with healthcare providers or research organisations...Initially focused on oncology-related data, the collaboration will look at how best to exchange data gathered from multiple owner mediated data sources such as Electronic Medical Records, clinical trials, genomic data, mobile devices, wearables, and Internet of Things technologies...The deal aims to overcome the limitations of large scale sharing of health data seen in the past, namely data security and patient privacy concerns during the data exchange process.
- FDA warning letter tells Clorox to clean up Aplicare plant (fiercepharma.com)
The FDA says the unit of Clorox that manufactures povidone-iodine drug products needs to clean a big mess at a plant in Meriden, Connecticut, which failed to follow steps to insure the sterility of its wound products—products that the FDA also said are “unapproved.”...The agency...posted a warning letter for the Aplicare plant…(it) said...the plant failed to implement adequate microbial controls...some of its products are unapproved because they do not comply with the FDA’s OTC Final Monograph for Topical Antifungal Drug Products.
- The FDA to study whether people can recognize misleading drug ads (mmm-online.com)
The FDA has proposed two studies that would seek to determine how well consumers and healthcare providers can identify deceptive prescription drug claims in branded marketing materials...The first study would investigate the ability of consumers and HCPs to detect false or misleading promotions when showed two mock pharmaceutical websites, one site oriented toward providers and a separate one for consumers...The second study would focus on examining whether the participants can identify the difference between implicit and explicit deceptive claims...is the claim overtly wrong? Or is it misleading because it understates certain information?...and whether those claims affect their attitudes and intentions toward the promoted drug...The notice comes as the agency grapples with an industry pushing for the sharing of truthful, non-misleading information with more stakeholders outside of a drug's approved label
- Pharmacy Week in Review: January 13, 2017 (pharmacytimes.com)
Kelly Walsh, PTNN. This weekly video program provides our readers with an in-depth review of the latest news, product approvals, FDA rulings and more.
- Non-Proprietary Naming of Biologics and Biosimilars: FDA Finalizes Guidance (raps.org)
In a departure from the way the WHO and Europe name biologics, the US Food and Drug Administration...finalized long-awaited guidance on how biosimilars and their biologic reference products’ names should include a four-letter, FDA-designated meaningless suffix attached at the end of the nonproprietary name...The decision to finalize this guidance follows...but large opposition to the idea of using meaningless rather than meaningful suffixes that could make it easier to distinguish the manufacturers of the products...For example...Sandoz’s Zarxio, which includes a non-proprietary name with a meaningful suffix (Sndz for Sandoz): filgrastim-sndz...But FDA has said it will change Zarxio’s nonproprietary name from filgrastim-sndz to “filgrastim-bflm." And Amgen's Neupogen (filgrastim) would be changed to "filgrastim-jcwp."...But these suffix rules do not necessarily apply to all related biologics...some instances it has designated a proper name that includes an identifier attached as a prefix to distinguish the products from previously licensed biologics. For example, with ado-trastuzumab emtansine, FDA includes a unique prefix, which it says was necessary to minimize certain medication errors and to facilitate pharmacovigilance.
- Nevada State Board of Pharmacy – Newsletter January 2017 (bop.nv.gov)
- Changing Faces
- Bowl of Hygeia Awarded to Adam Porath, PharmD
- No Prescription Needed!
- FDA Issues Final Rule Amending List of Drug Products That May Not Be Compounded
- Selected Medication Risks to Manage in 2017
- Environmental Factors, Workflow, and Staffing Patterns – Poor Quality Lighting
- DEA to Decrease Manufacturing Amount of Opioid Controlled Substances in 2017
- New CDC Brochure Offers Pharmacists Tips for Addressing Prescription Opioid Abuse and Overdose
- FDA Requires Boxed Warnings and Patient Focused Medication Guides Indicating Serious Risks Related to Combined Use of Certain Opioid Medications and Benzodiazepines
- FDA’s Division of Drug Information Offers CE Webinars for Students and Clinicians
- FDA Approves Labeling Changes for All Prescription Testosterone Products
- Latest FDA Drug Info Rounds Training Videos Available
- Inspector's Corner
- Ghost Towns and Medicines
- NCPA suggests any changes to ACA maintain Rx coverage and pharmacy access (drugstorenews.com)
The National Community Pharmacists Associatio...reached out to Republican congressional leaders to provide the community pharmacist perspective on repeal and replacement of the Affordable Care Act and potential Medicaid changes...Specifically, NCPA recommended any repeal and replacement of ACA include the following provisions:
- Preserve prescription drug coverage as an essential benefit to help reduce overall health costs;
- Require a robust pharmacy network that includes meaningful access to retail pharmacies, including independent pharmacies, for patients to fill their prescriptions and promote proper adherence;
- Maintain current definition for Average Manufacturer Price that is used to determine Medicaid reimbursements;
- Retain sections of the ACA that were dedicated to improving Medicare Part D medication therapy management programs, including an annual comprehensive medications review, and build upon it to further integrate pharmacists into the health care delivery system; and
- Keep transparency provisions requiring pharmacy benefit managers serving exchange plans and Medicare Part D to disclose to HHS the generic dispensing rate for retail and mail order pharmacies, the amount of the rebates collected, the amount passed onto the health plan and the total difference between what the PBM charged the plan and what it paid out to pharmacies
- Federal judge orders state to provide Mumia Abu-Jamal with hepatitis C treatment (philly.com)
A federal judge...ruled that Mumia Abu-Jamal should be provided new medications by the state to treat his hepatitis C infection...U.S. District Judge...ordered that Abu-Jamal, who is serving life in prison for the 1981 killing of Philadelphia Police Officer Daniel Faulkner, must be seen by a doctor within 14 days to determine if there is a medical reason he should not get the expensive drugs...If...is medically cleared, the state must provide him with recently developed direct-acting antiviral medication, also known as DDA (DAA, Direct-Acting Antivirals)... Department of Corrections...has argued in court filings that Abu-Jamal has not met the criteria for treatment...The state has about 7,000 inmates with hepatitis C, and treating them - at a cost of $84,000 to $90,000 per person - would cost $600 million... DOC will no doubt appeal this ruling...
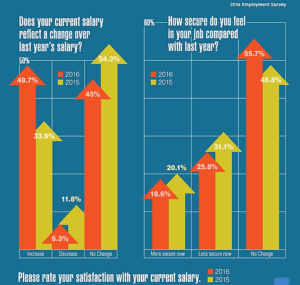
- Career Consensus: 2016 Employment Survey Europe-based bio/pharma employees are unified on satisfaction with employment conditions—and dissatisfaction with salaries (images2.advanstar.com)
While the pending exit of the United Kingdom from the European Union has generated uncertainty for industries and employees, including the bio/pharma markets across Europe, bio/pharma employees also expressed concerns about compensation, job security, and career development in the 2016 Pharmaceutical Technology/Pharmaceutical Technology Europe annual employment survey. Overall, the 2016 survey responses were similar to opinions about the employment market as the 2015 survey, but respondents also expressed stronger desires to seek better pay and career opportunities. Nearly three-quarters agreed or strongly agreed that their job was secure despite changes at their companies. More than half of the respondents reported an organizational change in their companies; 26.7% via merger or acquisition and 24.8% by downsizing/restructuring.